Tips & Tricks
DIL/TMA – Even Special Samples Can Be Handled!
The measurement of solid samples is generally no particular challenge for the methods of dilatometry (DIL) and thermomechanical analysis (TMA).
It gets more difficult, however, if the samples are powders, pastes or liquids or if the sample changes from the solid to the liquid state during the measurement. In the following, techniques will be presented enabling measurement on such samples with a dilatometer or TMA. Additionally, the points to be considered during preparation of such samples will be discussed.

Container for Powders and Pastes
Various sample containers are available for the measurement of powdery or pasty samples with the DIL and TMA (see figure 1). They are made of SiO2, Al2O3, sapphire or graphite. The choice of container material depends primarily on the temperature range of the measurement. Furthermore, possible interactions between the sample and container material must be taken into account.
Prior to preparation, the container including the pistons is first weighed empty and the lengths of the pistons and cylinder are determined. Afterwards, a piston is pushed into the cylinder so that it is flush with the lower edge. The container is then placed on this side. Now, it is filled from the side that is open at the top until the material is heaped slightly over the edge. For powders, it is recommended that the container be tapped occasionally during filling in order to prevent the occurrence of any cavities within the sample material. The excess is then removed with a straight edge (e.g., razor blade) and the second piston is pushed into the cylinder from above until the lower piston pokes out slightly. The lower piston is then also the one in contact with the pushrod. Now, the total length and mass of the cylinder with sample and pistons are determined. The sample length is then obtained by subtracting the lengths of the two pistons. Care must be taken that samples are prepared so as to have similar mass and length. This ensures that the samples will have a similar bulk DensityThe mass density is defined as the ratio between mass and volume. density. In general, however, it is recommended – if the sample and the measuring task both allow for it – to press the powder into a pellet.
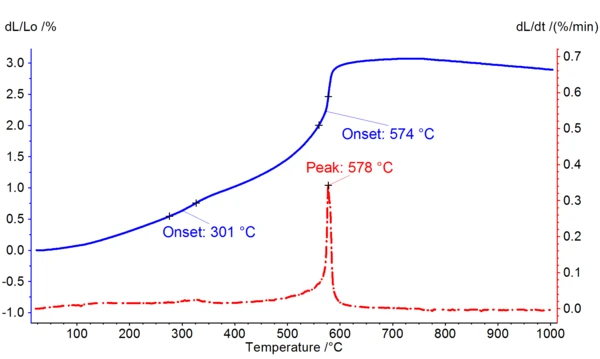
Figure 2 shows an example of a measurement on a powder sample in an Al2O3 container. Here, a foundry sand sample was heated from room temperature to 1000°C at 5 K/min. At 301°C (extrapolated onset), a slight bend in the thermal expansion can be observed, which might be due to the burnout of organic components. The α-β transition of quartz can clearly be seen, with a maximum in the expansion rate at 578°C.
For the filling of pasty samples, such techniques as injection can be used. To prevent the formation of air bubbles in the sample during filling, the tip of the injection nozzle is initially positioned at the bottom of the container and then slowly pulled upward as it empties. Here also, the container is filled until it is slightly heaped over the top, and the excess is then removed with a straight edge. The remainder of the procedure is then the same as described for the powder samples. Due to its vertical design, the TMA also offers another measurement option. Here, the powder can be filled into a crucible and covered with a disc (see figure 3). Here, too, the same masses and filling levels should be set, if at all possible.


Waxes
For the measurement of the thermal expansion of waxes into the melting range, NETZSCH offers a steel container. This consists of a steel cylinder, a cap and a diaphragm. First, the depth of the steel cylinder is determined since this will be equal to the later sample length. To fill the container, smaller pieces of wax will be melted within the container little by little, until it is over-filled (above the edge) after the wax has cooled (see figure 4). Then, the excess is cut off with a sharp razor blade. Melting Temperatures and EnthalpiesThe enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state. The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt).Melting can be carried out with a hot-air gun or a hot plate. This method, however, is only suitable for samples that do not undergo change as a result of the procedure described above.
Liquids
Liquids must be enclosed in a container to determine the thermal expansion. There are two possibilities here, and the appropriate choice depends heavily on the viscosity of the sample to be measured. For viscous samples (similar to honey), the same container can be employed as described beforehand for powdery and pasty samples. Care must be taken, however, to verify that the sample will not become significantly less viscous during the course of the measurement, since the sample could then leak out of the container. In order to prevent the occurrence of any damage to the sample holder, such substances should be tested beforehand in a separate furnace when in doubt.
For samples with low viscosity, the same steel container as is used for measurements on waxes can be utilized. In this case, however, only comparative measurements are possible, and no absolute determination of the thermal expansion may be carried out, since it is not possible to correct for the expansion behavior of the container. This, however, is rather insignificant in consideration of the heavy expansion of waxes during melting.