Glossary
Reaction Temperature and Reaction Enthalpy
Reaction temperature and reaction enthalpy can be determined, for example, with the help of differential scanning calorimetry (DSC). This method is described in DIN EN ISO 11357-5, for example.
Reactions such as Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.crosslinking reactions or polymerization reactions can generally be observed as ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal effects in the DSC measurement curves.
Figure 1 shows a DSC curve at a constant heating rate with an ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal effect. This ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal effect can be described by different characteristic temperatures.
The Onset temperature is the extrapolated beginning temperature of the reaction; the End temperature is the extrapolated end temperature of the reaction
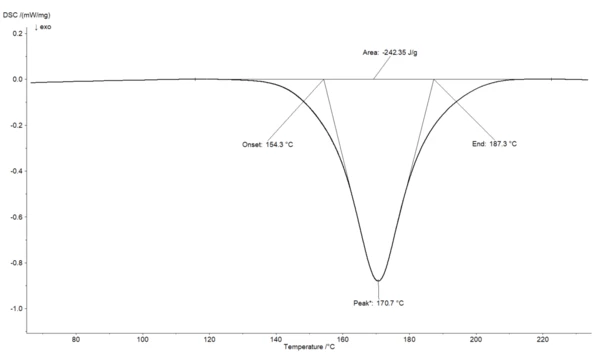
The reaction enthalpy is the change in enthalpy in the course of a reaction; i.e., the energy conversion for a reaction carried out at a constant pressure. The reaction enthalpy is the difference between the enthalpies of formation of the products and educts.
The reaction enthalpy can be obtained by DSC measurement by integrating the area of the reaction peak and the interpolated baseline between the beginning and end of the reaction. In the example shown in Figure 1, an enthalpy of 242.35 J/g is determined.

