03.06.2020 by Milena Riedl
Novel Technique for Preparing Samples for Biomaterial Investigations
Alucent Biomedical Inc. is a biomedical company dedicated to developing its breakthrough Natural Vascular Scaffolding (NVS) technology for the treatment of vascular disease. Multiple sample preparation techniques were evaluated to investigate a method that could alleviate signal artifacts attributed to sample movement during heating. Learn how the sample is prepared to achieve accurate and reproducible thermograms for biological tissue.
by Justin Barrett and Kevin Warner, PhD, Alucent Biomedical, and Michael Hsu, NETZSCH Instruments North America
Alucent Biomedical, Inc. is a biomedical company dedicated to developing its breakthrough Natural Vascular Scaffolding (NVS) technology for the treatment of vascular disease. NVS uses photoactivated protein linking of the vessel’s native structural proteins (i.e., collagen) to open vessels during angioplasty and keep them open – without the use of permanent implants.
Using DSC in Product Development Research
One aspect of the product development strategy is to identify and develop methods to characterize the photoactivated molecular changes in the vessel wall. Changes to the extracellular matrix proteins can be detected by a change in the denaturation temperature. In principle, higher denaturation temperature is indicative of an increased density of covalently linked extracellular matrix caused by the NVS treatment. Differential Scanning Calorimetry (DSC) is being investigated as a technique to determine the differences in the denaturation temperatures of the structural proteins of blood vessels in native versus treated conditions. DSC is not routinely used for biomaterial investigations, but carries value for biological tissue characterization to aid product development. A review of the relevant literature had limited discussion on sample preparation and method development to ensure reliable and reproducible DSC results.
How to Alleviate Signal Artifacts
Multiple sample preparation techniques were evaluated to investigate a method that could alleviate signal artifacts attributed to sample movement during heating. The techniques explored included tight packing of diced sample, crucible configuration (crimped lid, pierced lid, un-crimped lid, no lid), crucible size, using a disk of the sample matching the internal diameter of the crucible, and stacking multiple said sample disks. All of these tests yielded inconsistent results as the sample still had freedom to move within the crucible during heating. To combat this phenomenon, Alucent Biomedical Inc. came upon the idea that, instead of stacking multiple sample disks, an inert object placed between the hermetically sealed lid and the sample could better keep the sample pressed down and minimize movement. The size and morphology of both the sample and inert object were chosen to be a disk that matched the internal diameter of the crucible which would maximize the surface area contact of the sample with the bottom of the crucible pan to allow optimal heat transfer. Lastly, it was imperative that the inert object did not produce any extraneous DSC energetics that could interfere with the underlying sample signals. It was for these reasons that sapphire was chosen, a relatively cheap, readily available sensitivity calibration standard with no energetics in the area of interest of the samples.
Two Critical Factors for the Sample Preparation
1) Sample sizing to match the internal diameter of the DSC pan 2) Filling the DSC pan headspace with an inert material to prevent heat-induced movement of the sample during measurement The figure shows the thermograms from sections of the same biological tissue prepared using two different sample preparation techniques. Thermogram 1 in the figure represents biological tissue prepared by cutting the sample into pieces and placing in the sample pan. Thermogram 2 in the figure represents biological tissue prepared as described above to ensure sample movement is minimized.
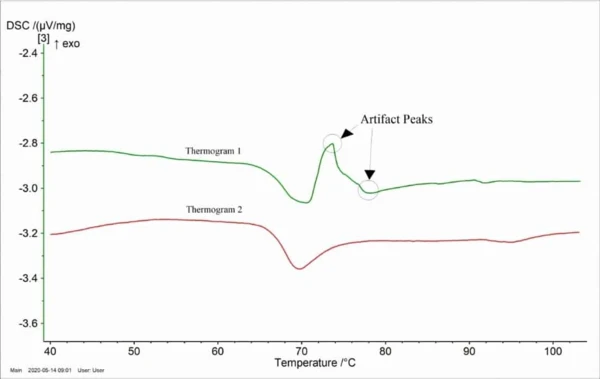
The results demonstrate that a critical parameter in generating accurate, reproducible thermograms for biological tissue is a sample preparation methodology that minimizes heat-induced sample movement. Matching tissue size to the internal diameter of the DSC pan and filling the headspace with an inert material can help prevent artificial signals caused by sample movement in the DSC pan. The Application Note is available here!
