Glossary
Solid-Gas Reactions
Solid-gas reactions are a type of heterogeneous solid-state reaction occurring when a reactive solid is exposed to a stream of reactive gas. Typical examples of solid-gas reactions are Sorption ProcessSorption is a physical and chemical process by which a substance (typically a gas or liquid) accumulates within another phase or on the phase boundary of two phases. Depending on the place of accumulation, a differentiation is made between absorption (accumulation in a phase) and adsorption (accumulation at the phase boundary).sorption and corrosion of metals.
Solid-gas reactions can be investigated by means of Thermogravimetric Analysis (TGA), Differential Scanning Calorimetry (DSC) and Evolved Gas Analysis (EGA).
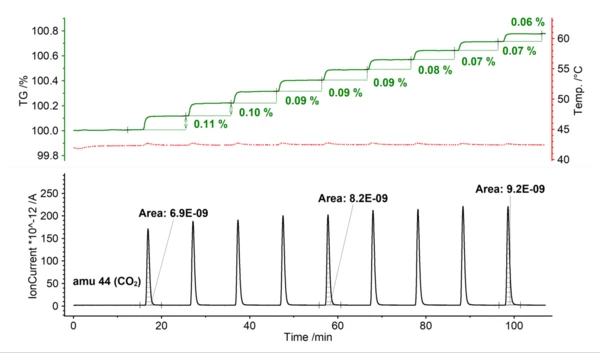
In the example below, calcium oxide (CaO, burned lime) was treated by PulseTA with defined injections of CO2 at 43°C. The volume of each injected pulse amounted to 250 μl. Every gas injection causes a reaction of solid CaO with the reactive CO2 gas, which can be seen via the stepwise increase of the sample mass. With each consecutive gas uptake, however, the height of the new step is reduced. This reaction may be continued until a plateau in the TGA signal is reached. Simultaneously, the peak area below the MS signal for m/z 44 (referring to CO2) can be evaluated. The peak area increases as the amount of CaO conversion decreases. The higher the level of saturation with CO2, the lower the consumption of the pulse gas.