Введение
Спортивные товары и игрушки для детей и домашних животных часто изготавливаются из гибкого пластика. В качестве примера можно привести сенсорные жевательные игрушки, фигурки, мягкие элементы для захвата, а также мячи любого вида. Чаще всего используется ПВХ (поливинилхлорид), который можно сделать более мягким и гибким, добавив в него пластификаторы. Эти соединения не имеют ковалентной связи с полимерной цепью, поэтому они могут испаряться или вымываться слюной или потом. Выделение пластификаторов, таких как фталаты, может быть вредным. В некоторых случаях это можно распознать даже по неприятному запаху.
Семейство фталатов, как известно, вызывает ряд опасностей для здоровья. Они действуют подобно гормонам и, как было доказано, вызывают повреждения печени, бесплодие, диабет, рак и многое другое. Поэтому Европейский союз с 2007 года запретил использование ряда фталатов в продуктах, контактирующих с пищей, в игрушках, в детских вещах и медицинских препаратах.
Поведение при разложении и идентификация пластификаторов
Термический анализ помогает обнаружить пластификаторы в полимерах. С помощью ТГА-ФТ-ИК анализа можно анализировать продукты на содержание пластификаторов и определять тип используемого пластификатора.
В следующем примере поверхностный слой различных игрушечных мячей был разрезан на small частей и измерен с помощью PERSEUS® TG 209 F1 Libra® в соответствии с условиями измерения, приведенными в таблице 1.
Шарик №. 1 демонстрирует несколько ступеней потери массы во время пиролиза, см. рисунок 1. Эти этапы потери массы происходят в результате испарения пластификатора или других органических добавок и пиролиза полимера в диапазоне температур от 200°C до 500°C. Разложение неорганических наполнителей было обнаружено между 500 и 700°C. Пики на кривой DTG (скорость потери массы) представляют собой температуры максимальных скоростей потери массы. Кривая Грамма-Шмидта отображает общую интенсивность ИК-излучения и является зеркальным отражением кривой ДТГ и также показывает максимальную интенсивность во время этапов потери массы. Это доказывает взаимодействие эволюционировавших соединений с ИК-лучами.
Таблица 1: Условия измерения
| Образец | Шарик № 1 | Шар № 2 |
|---|---|---|
| Масса образца | 9.08 мг | 10.38 мг |
| Температурная программа | От RT до 850°C | |
| Скорость нагрева | 10 K/мин | |
| Газовая атмосфера | Азот | |
| Расход газа | 40 мл/мин | |
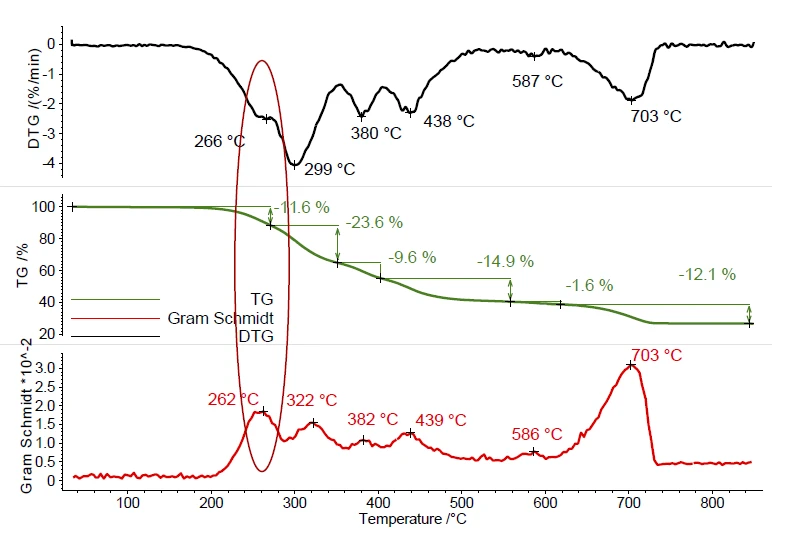
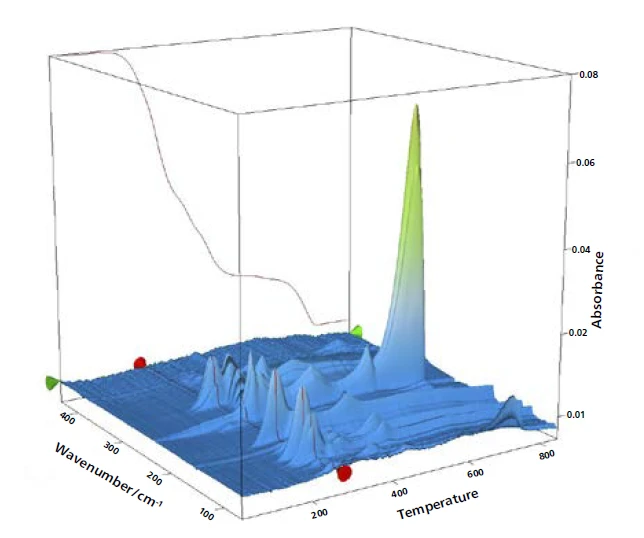
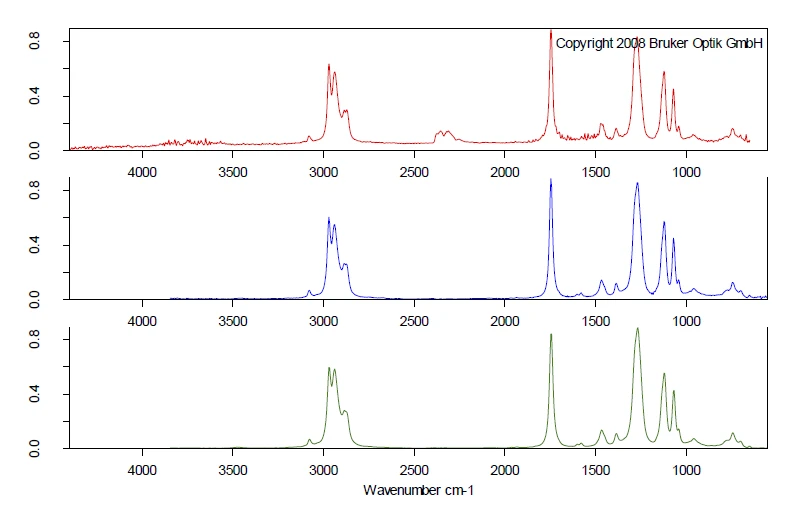
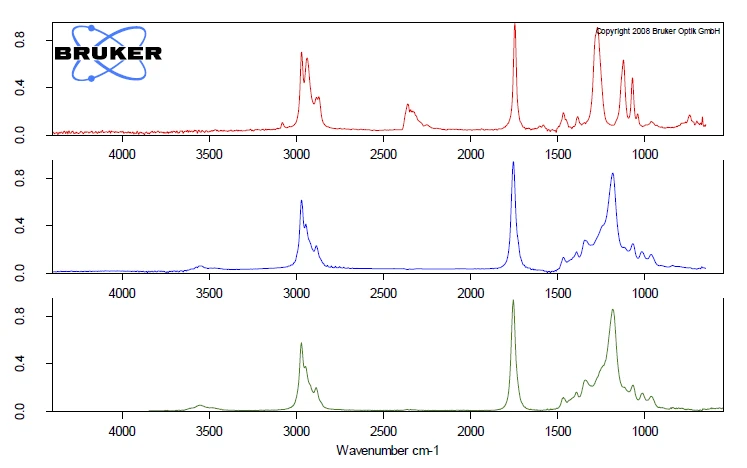
Полные ИК-данные представлены на рис. 2 в виде трехмерной диаграммы, зависящей от температуры и числа волн. Кривая ТГА нанесена красным цветом сзади и показывает зависимость потери массы от увеличения интенсивности ИК-излучения. В этом примере более точно исследован только первый этап потери массы. Для детального анализа содержащегося пластификатора был получен двумерный ИК-Фурье спектр, который сравнили с газофазным libraдля идентификации выделившихся соединений. Было обнаружено высокое сходство спектра при 266°C со спектрами libraди-н-октилфталата (DOP, синий) и бис(2-этилгексил)фталата (DEHP, зеленый). Можно предположить, что было выброшено одно соединение или смесь различных фталатов. Однако это сравнение ясно показывает, что шар No. 1 содержит вредные фталаты. Поскольку следующий этап потери массы немного совпадает с выделением фталатов, с помощью ИК-Фурье при 266°C также было обнаружено некоторое количествоCO2 small.
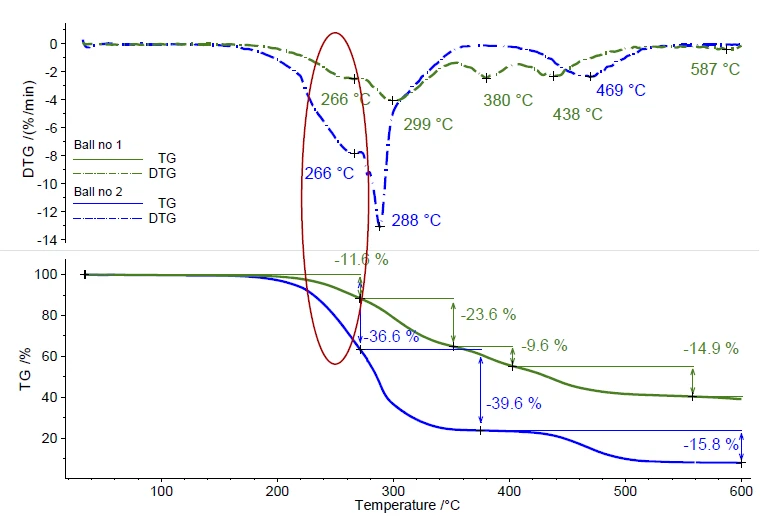
Второй шар был исследован в тех же условиях. Сравнение результатов обоих измерений ТГА показано на рисунке 4. В поведении пиролиза видна явная разница. Однако и для шара № 2 первый этап потери массы был обнаружен в диапазоне температур между 200 и 280 °C, а также с пиком на кривой ДТГ при 266 °C. Только ИК-Фурье может дать подробную информацию о содержащемся пластификаторе.
Сравнение ИК-Фурье спектров двух образцов шариков, полученных при 266°C, показывает совершенно разную картину колебаний, см. рисунок 5. Сравнение спектров при 266°C шарика № 2 (синий) со спектром газовой фазы library дает четкое соответствие со спектром трибутилцитрата (зеленый). В шарике № 2 токсичные фталатные пластификаторы были заменены на нетоксичный лимонный эфир, который также выступает в качестве пластификатора.
Резюме
Процессы газовыделения и разложения полимеров можно исследовать с помощью термического анализа. Термогравиметрия указывает на выделение газов уже при температуре ниже 300°C. Идентифицировать выделяющиеся газы можно только с помощью расширенного газового анализа, например ИК-Фурье. В данном примере удалось идентифицировать различные используемые пластификаторы и, следовательно, отличить токсичные добавки от нетоксичных. Прибор PERSEUS® TG 209 F1 Libra® идеально подходит для решения этой задачи.