소개
지르칼로이 BCR-276(Zirc-4)은 유럽위원회 인증 기준 재료입니다. 지르칼로이는 열 중성자 흡수 단면적이 낮고 열적 및 기계적 특성이 우수하여 원자로의 피복재로 널리 사용됩니다. 일본 후쿠시마 제1 원자력 발전소의 지진 및 쓰나미 관련 사고 당시, 건물 지붕 아래에 수소가 축적되어 발화되었습니다. 이러한 특수한 조건에서 수소를 생산하는 한 가지 방법은 비교적 간단한 화학 반응(1)을 따르는 것입니다:
Zr + 2 H2O--> ZrO2 + 2 H2 + ΔH
이 반응을 확인하기 위해 몇 가지 예비 실험을 수행했으며 아래에 설명되어 있습니다.
실험적
NETZSCH STA 449 F3 Jupiter® 에는 수증기 용광로와 QMS 질량 분석기 QMS 403 Aeolos가 장착되어 있었습니다. BCR-276의 실린더 3개(시료 무게 약 600mg)를 TG 시료 캐리어의 알루미나 플레이트 위에 놓았습니다. 샘플은 N2와 수증기 아래에서 5 및 10 K/min으로 1050°C까지 가열되었습니다. 질량 분석기로 물과 수소의 강도를 모니터링했습니다.
결과 및 토론
그림 1은 10 K/min 측정에 대한 TG 곡선(질량 변화)과 온도 대비 수소 및 물의 강도를 보여줍니다. 산화로 인해 무게가 증가하기 시작한 후 수소 수준도 증가합니다.
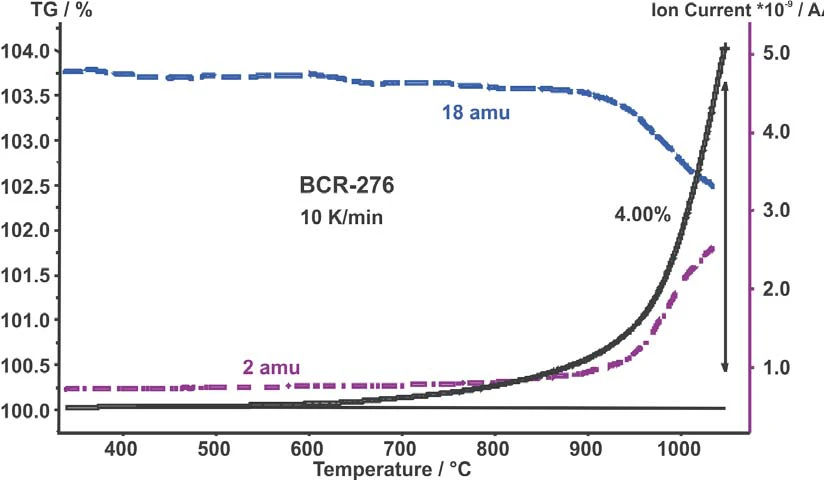
수소 증가와 동시에 물의 강도는 감소합니다. 1050°C까지의 무게 증가는 4wt%였습니다.
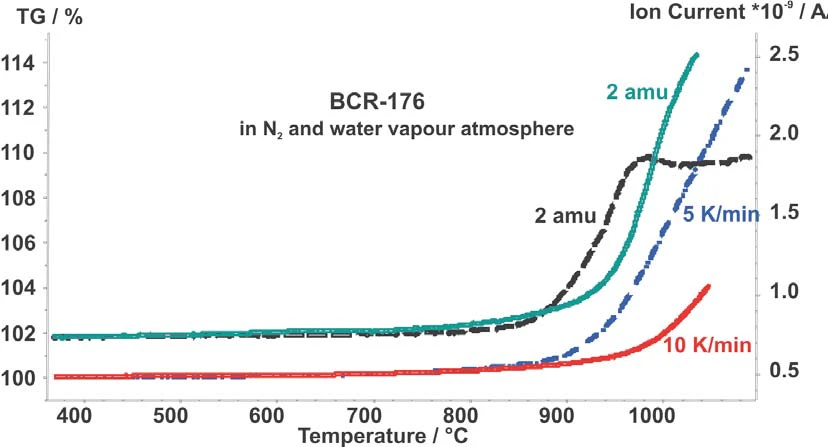
그림 2에서는 5 K/min과 10 K/min에서 두 측정값에 대한 TG 곡선과 수소의 강도를 비교했습니다. 5 K/min의 가열 속도에서 산화와 수소 진화는 10 K/min보다 일찍 시작됩니다. 약 950°C에서 수소 진화는 안정된 포화 상태(일정한 수준)로 들어갑니다.
그림 3은 측정 전과 후의 샘플을 보여줍니다.
