Glossary
Degree of Cure
The degree of Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.curing describes the conversion achieved during Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.crosslinking reactions (Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.curing).
In macromolecular chemistry, crosslinking refers to reactions in which a large number of individual molecules are linked to form a three-dimensional network. Linkage can be achieved either by direct setup of macromolecules or reaction to the already existing polymers.
This can easily be measured by means of differential scanning calorimetry (DSC).
The degree of Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.curing, α, is the amount of heat converted divided by the reaction enthalpy.
α = H/ΔHR
The degree of cure of an already (partially) crosslinked sample can be determined via the residual enthalpy of post-crosslinking, HPC (post-cure). Then, the degree of cure can be calculated as
α = 1-(HPC/ΔHR)
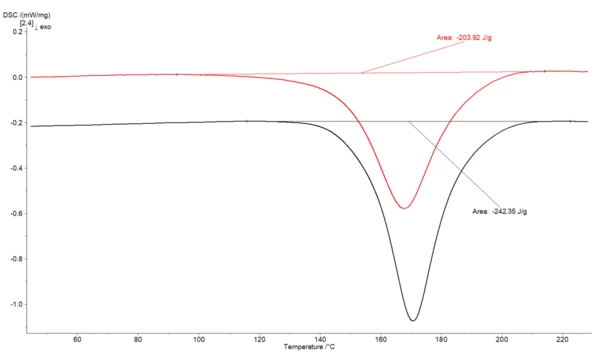
Figure 1 depicts an example for the calculation of the degree of cure for a partially cured adhesive. The uncured adhesive shows a Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.curing enthalpy of 242.35 J/g, whereas the partially cured sample shows an enthalpy of just 203.92 J/g for the post Curing (Crosslinking Reactions)Literally translated, the term “crosslinking“ means “cross networking”. In the chemical context, it is used for reactions in which molecules are linked together by introducing covalent bonds and forming three-dimensional networks.curing reaction. Therefore, a degree of cure of 15.9 % for the partially cured sample can be calculated.