applications
Metals & Alloys
Specific heat capacities, coefficients of thermal expansion, melting and solidification temperatures or enthalpies, and characteristic thermal effects – also under corrosive conditions – are some of the areas of high interest in metallurgical applications.
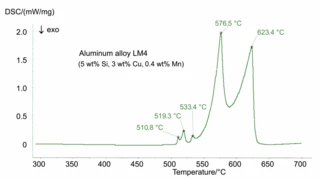
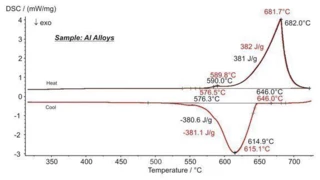
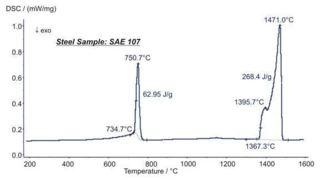
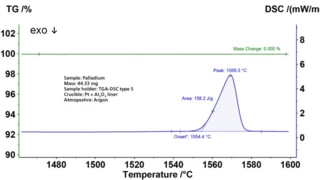
Melting Temperatures and EnthalpiesThe enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state. The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt).Melting, CrystallizationCrystallization is the physical process of hardening during the formation and growth of crystals. During this process, heat of crystallization is released.crystallization, Phase TransitionsThe term phase transition (or phase change) is most commonly used to describe transitions between the solid, liquid and gaseous states.phase transitions and specific heat capacities are important chemical properties in metals and alloys that are measured by means of DSC or STA (simultaneous thermal analysis). Besides these, the influence of corrosion, OxidationOxidation can describe different processes in the context of thermal analysis.oxidation or reduction as well as magnetic transitions and the Thermal StabilityA material is thermally stable if it does not decompose under the influence of temperature. One way to determine the thermal stability of a substance is to use a TGA (thermogravimetric analyzer). thermal stability of materials can also be analyzed.
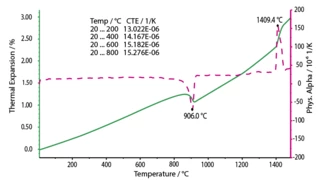
With dilatometers, the coefficient of thermal expansion can be precisely measured; DMA determines the modulus of elasticity and the damping values that are relevant in practice for components.
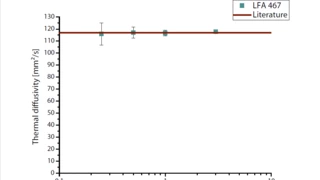
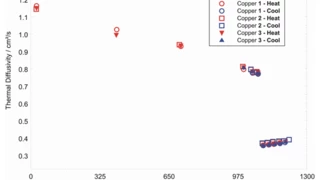
With LFA , the Thermal DiffusivityThermal diffusivity (a with the unit mm2/s) is a material-specific property for characterizing unsteady heat conduction. This value describes how quickly a material reacts to a change in temperature.thermal diffusivity and conductivity – even of molten metals – can be investigated.
Application Literature