Glossary
Melting Temperatures and Enthalpies
The melting point as well as the enthalpy of fusion are important and useful thermodynamic properties.
The enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state.
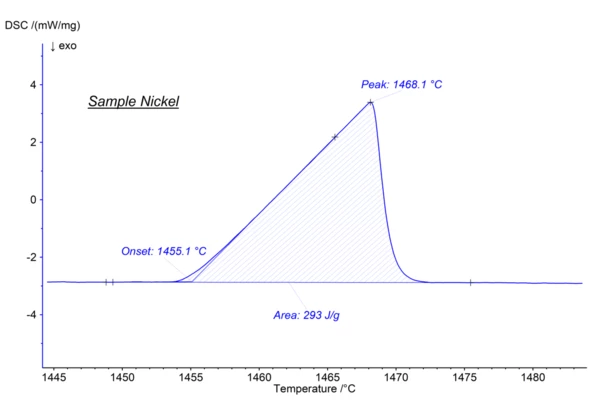
The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt). As an example the melting point of Nickel is plotted below, where the melting starts at an extrapolated onset temperature of 1455°C.
The detected peak temperature at 1468°C is depending of some influences factors like for instance sample mass or heating rate.
At the melting point, the solid and liquid phases exist in equilibrium. The melting point of a substance depends on pressure and is usually specified at standard pressure.
Melting effects can easily be determined as endothermal processes with a high degree of both reliability and accuracy by differential scanning calorimetry (DSC).
Further, dilatometry is also a feasible method for such procedures as determination of the melting point.