Glossary
Pyrolysis
Pyrolysis is the thermal Decomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition of organic compounds in an inert atmosphere.
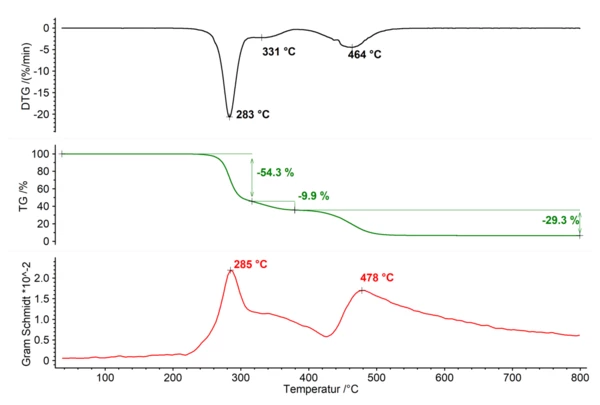
During pyrolysis, solid, liquid or gaseous products can be generated. If gases are released from the sample during pyrolysis, the changes in mass can be detected by TGA (thermogravimetry). The evolved gases can be identified by EGA (evolved gas analysis). Pyrolysis studies are often carried out on polymers, coal, biomass or organic compounds. Residual masses can be indicative of additional components.
The figure below shows the pyrolysis of PVC in a nitrogen atmosphere. The Gram Schmidt signal depicts the change in intensity caused within the FT-IR by the infrared-active gases released. Individual gaseous products can then be identified by database comparison. Measurement conditions: RT–800°C, 10 K/min, 40 ml/min nitrogen, sample mass 10 mg