Glossary
Thermal Conductivity
Definition of Thermal Conductivity
Thermal conductivity (λ with the unit W/(m•K)) describes the transport of energy – in the form of heat – through a body of mass as the result of a temperature gradient (see fig. 1). According to the second law of thermodynamics, heat always flows in the direction of the lower temperature.

Do you have any questions?
Suitable products for your measurement
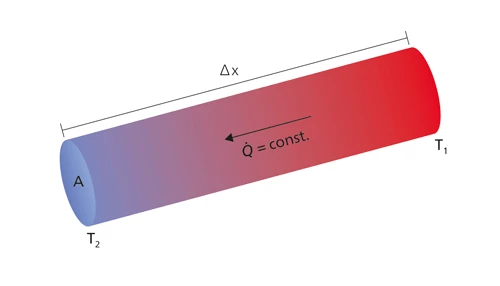
The relationship between transported heat per unit of time (dQ/dt or heat flow Q) and the temperature gradient (ΔT/Δx) through Area A (the area through which the heat is flowing perpendicularly at a steady rate) is described by the thermal conductivity equation.
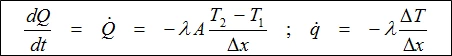
Thermal conductivity is thus a material-specific property used for characterizing steady heat transport. It can be calculated using the following equation:

Where
a: Thermal DiffusivityThermal diffusivity (a with the unit mm2/s) is a material-specific property for characterizing unsteady heat conduction. This value describes how quickly a material reacts to a change in temperature.Thermal diffusivity
cp: Specific Heat Capacity (cp)Heat capacity is a material-specific physical quantity, determined by the amount of heat supplied to specimen, divided by the resulting temperature increase. The specific heat capacity is related to a unit mass of the specimen.Specific heat capacity
ρ: DensityThe mass density is defined as the ratio between mass and volume. Density
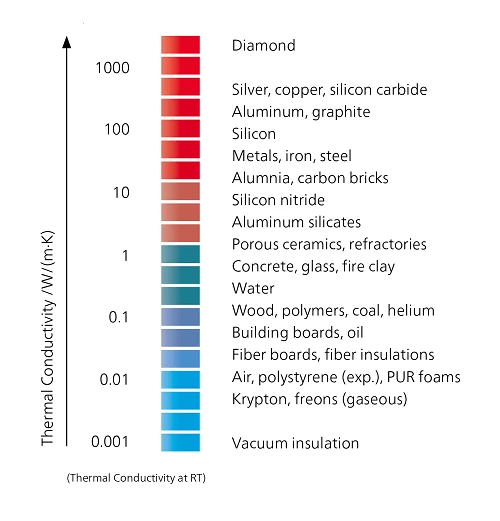
An overview of the thermal conductivity for various materials is shown in figure 2. Depending on the material the thermal conductivity can be measured by LFA, HFM or GHP.