
14.02.2023 by Aileen Sammler
How Rheology Optimizes Your Processes
Nowadays, steadily growing energy prices for gas and electricity result in highly expensive polymeric raw materials and rising processing costs. Therefore, resource-, cost-, energy- and eco-efficiency is more important than ever. These objectives cannot be achieved if the effectiveness of the manufacturing processes is not (re)considered, i.e., if the process is not optimized and tailored to the processed polymeric material or vice versa. Process optimization needs to start with understanding the flow behavior of the polymer employed. That’s where rheology comes into play.
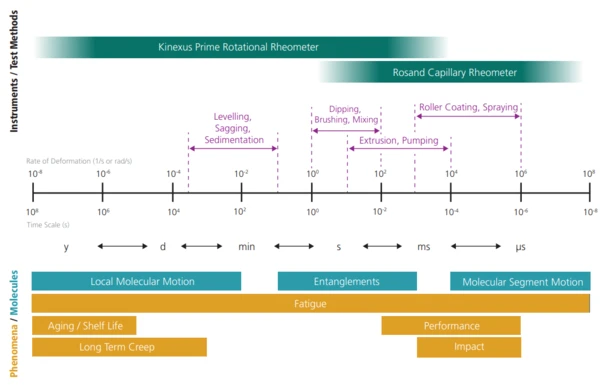
Rheology is the study of the deformation and flow of matter, unveiling the flow behavior of complex fluids such as polymers, food, slurries, suspensions and other compounds. Rheometers such as rotational and/or high-pressure capillary rheometers contribute to better understanding the rheological properties of the materials being analyzed. However, in order to optimize the processing conditions, the materials must be investigated at comparably high shear rates and temperatures.

Process Optimization by Rheology
The new Rosand high-pressure capillary rheometer line allows for the examination of the materials at process-relevant shear rate ranges up to extremely high shear rates (~108 s-1). Besides the shear dependency of the viscosity (shear thinning, shear thickening, Newtonian behavior), the rheological behavior under extensional flow can also be analyzed. Moreover, by means of pVT measurements, simulation-relevant parameters such as pressure-dependent specific volume, CrystallizationCrystallization is the physical process of hardening during the formation and growth of crystals. During this process, heat of crystallization is released.crystallization or Melting Temperatures and EnthalpiesThe enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state. The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt).melting temperature are accessible as well.
Read the full article and learn how to investigate the rheological properties of materials by means of the new NETZSCH high-pressure capillary rheometer:



