Введение
Влажность может влиять на свойства широкого спектра активных ингредиентов и вспомогательных веществ с точки зрения их стабильности, кристалличности, биодоступности и т.д. Одним из методов определения влияния влажности на поведение вещества является динамическая сорбция паров (DVS), при которой измеряется изменение массы образца при различных количествах паров растворителя, например, водяного пара. [1]
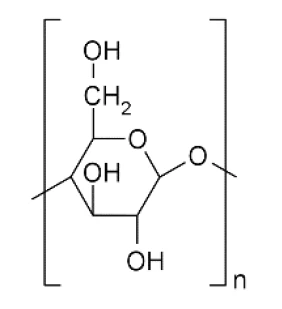
Такие измерения можно проводить с помощью STA (одновременного теплового анализатора), подключенного к модульному генератору влажности (рис. 1). Далее измерение динамической сорбции воды было проведено на микрокристаллической целлюлозе (МКЦ, химическая структура на рис. 2). Это вещество используется в таблетированных препаратах в качестве наполнителя и связующего. [2]


Условия измерения
Условия проведения экспериментов приведены в таблице 1.
Таблица 1: Условия проведения испытаний
| Устройство | STA 449 F3 Nevio , соединенный с генератором влажности |
|---|---|
| Образец | Микрокристаллическая целлюлоза |
| Масса образца | 41.22 мг |
| Держатель образца | Пластина из глинозема, Ø 17 мм |
| Температурная программа | Изотермическая температура 44°C, атмосфера азота, относительная влажность (RH) увеличивается от 0 до 80% |
Результаты измерений
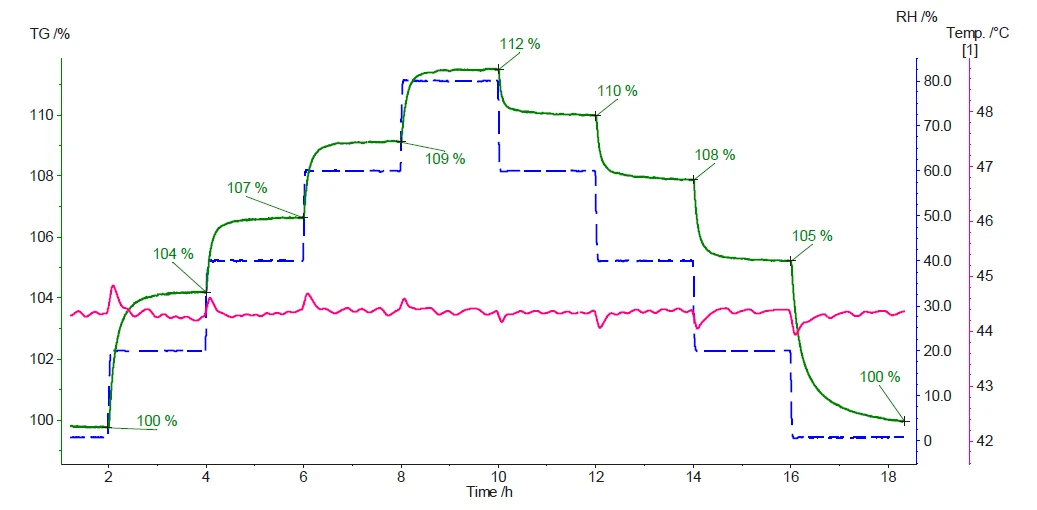
На рисунке 3 показаны измеренные масса и температура образца во время эксперимента.
Результаты демонстрируют сильную гигроскопичность микрокристаллической целлюлозы. Первое увеличение относительной влажности с 0% до 20% (синяя пунктирная кривая) вызывает увеличение массы на 4% (зеленая кривая). Последующие этапы показывают, что чем выше относительная влажность, тем больше прирост массы. Как только уровень влажности снижается, поглощенная и/или адсорбированная вода высвобождается, что приводит к потере массы. Когда в конце измерения будет достигнута абсолютно сухая атмосфера, количество поглощенной и/или адсорбированной воды будет количественно высвобождено. Это можно подтвердить, вернувшись к исходной массе образца (100%).
Каждое изменение уровня относительной влажности сопровождается пиком на кривой температуры образца (розовая кривая). Это связано с экзотермической и эндотермической природой сорбции и десорбции воды, соответственно.
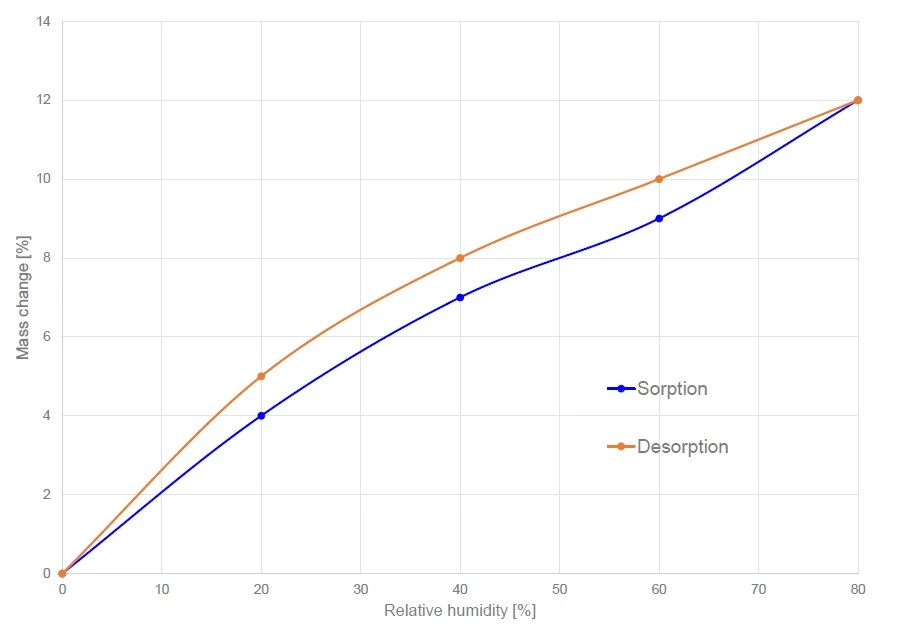
Прирост и потеря массы после достижения равновесия приведены на рисунке 4 для всех измеренных уровней относительной влажности от 0% до 80%. Максимальное увеличение массы составило 12% при относительной влажности 80%. Микрокристаллическая целлюлоза демонстрирует гистерезис сорбции, т.е. количество воды в образце во время десорбции выше, чем во время сорбции (см. рисунок 4), но в конечном итоге начальная и конечная точки цикла сорбции/десорбции идентичны.
Это явление гистерезиса характерно для многих пористых материалов. Чен и другие [3] показали, что связи вода-целлюлоза, образующиеся при набухании целлюлозы, не разрываются при десорбции при том же химическом потенциале.
Заключение
STA, подключенный к генератору влажности, позволяет проводить измерения динамической сорбции и десорбции воды. Измерения на микрокристаллической целлюлозе подчеркивают гистерезис этого процесса: Содержание влаги при десорбции выше, чем при сорбции. Это явление характерно для многих пористых материалов.