Introduzione
L'umidità può influenzare le proprietà di un'ampia gamma di principi attivi ed eccipienti in termini di stabilità, cristallinità, biodisponibilità, ecc. Un metodo per determinare l'influenza dell'umidità sul comportamento di una sostanza è il dynamic vapor sorption (DVS), in cui si misurano le variazioni di massa nel campione per diverse quantità di vapore solvente, ad esempio vapore acqueo. [1]
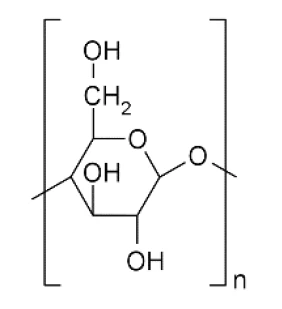
Tali misure possono essere effettuate con uno STA (analizzatore termico simultaneo) collegato a un generatore di umidità modulare (figura 1). Di seguito, è stata effettuata una misurazione dinamica dell'assorbimento dell'acqua sulla cellulosa microcristallina (MCC, struttura chimica in figura 2). Questa sostanza viene utilizzata nelle formulazioni di compresse come riempitivo e legante. [2]


Condizioni di misura
Le condizioni sperimentali sono riassunte nella tabella 1.
Tabella 1: Condizioni del test
| Dispositivo | STA 449 F3 Nevio collegato al generatore di umidità |
|---|---|
| Campione | Cellulosa microcristallina |
| Massa del campione | 41.22 mg |
| Supporto del campione | Piastra in allumina, Ø 17 mm |
| Programma di temperatura | Isoterma 44°C, atmosfera di azoto, umidità relativa (RH) aumentata da 0 a 80% |
Risultati della misurazione
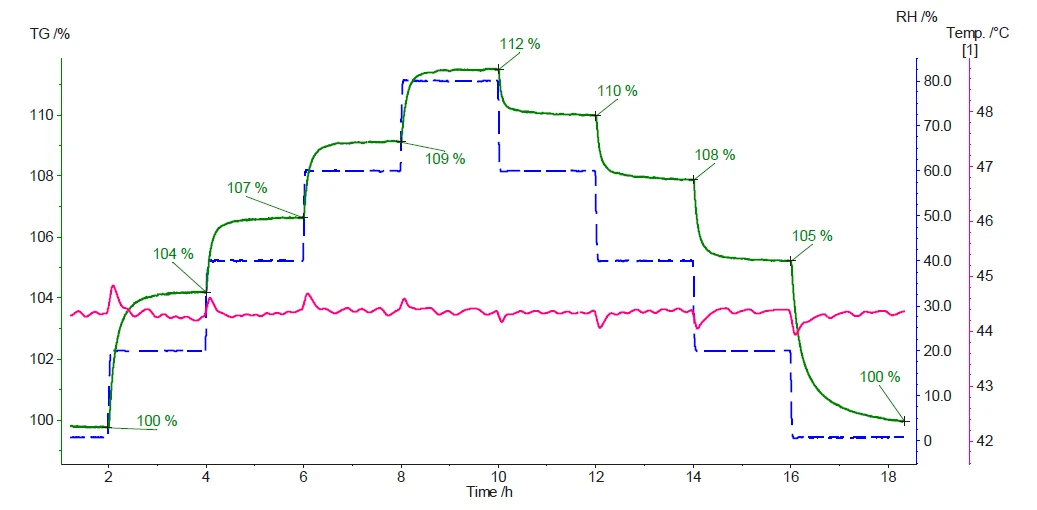
La Figura 3 mostra la massa del campione e la temperatura misurata durante l'esperimento.
I risultati dimostrano la forte natura igroscopica della cellulosa microcristallina. Il primo aumento dell'umidità relativa dallo 0% al 20% (curva blu tratteggiata) induce un aumento di massa del 4% (curva verde). I passaggi successivi mostrano che più alta è l'umidità relativa, maggiore è l'aumento di massa. Non appena il livello di umidità diminuisce, l'acqua assorbita e/o adsorbita viene rilasciata, con conseguente perdita di massa. Quando alla fine della misurazione si raggiunge un'atmosfera completamente asciutta, la quantità di acqua assorbita e/o adsorbita sarà stata quantitativamente rilasciata. Ciò può essere confermato dal raggiungimento della massa iniziale del campione (100%).
Ogni variazione del livello di umidità relativa è associata a un picco nella curva della temperatura del campione (curva rosa). Ciò è dovuto alla natura esotermica ed endotermica dell'assorbimento e del desorbimento dell'acqua, rispettivamente.
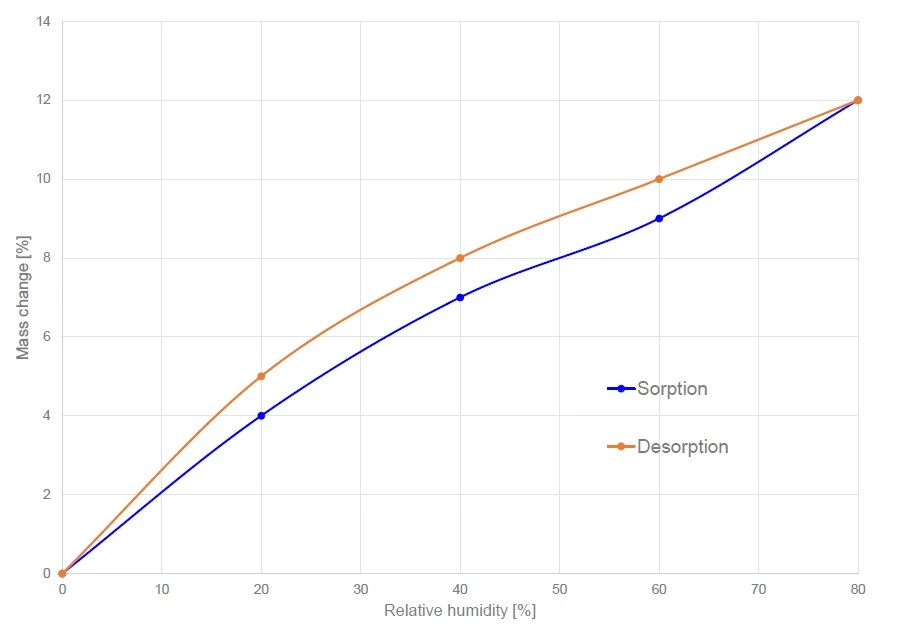
L'aumento e la perdita di massa dopo il raggiungimento dell'equilibrio sono riportati nella figura 4 per tutti i livelli di umidità relativa misurati tra lo 0% e l'80%. L'aumento massimo di massa è pari al 12% per un'umidità relativa dell'80%. La cellulosa microcristallina presenta un'isteresi di assorbimento, vale a dire che la quantità di acqua nel campione è maggiore durante il desorbimento che durante l'assorbimento (vedi figura 4), ma, in definitiva, il punto di partenza e il punto di arrivo del ciclo di assorbimento/desorbimento sono identici.
Questo fenomeno di isteresi è tipico di molti materiali porosi. Chen et al. [3] hanno dimostrato che i legami acqua-cellulosa formati durante il rigonfiamento della cellulosa non si rompono al momento del desorbimento allo stesso potenziale chimico.
Conclusione
Lo STA collegato a un generatore di umidità consente di misurare l'assorbimento e il desorbimento dinamico dell'acqua. Le misure sulla cellulosa microcristallina evidenziano l'isteresi del processo: Il contenuto di umidità è più alto durante il desorbimento che durante l'assorbimento. Questo fenomeno è tipico di molti materiali porosi.