
20.07.2023 by Prof. Dr. Ing. Sascha Englich
Rheokinetics – The Process-Specific Flow-Curing Behavior of Thermosetting Molding Compounds
The term rheokinetics is used to describe the reaction-dependent rheological behavior of thermosetting materials, which is of great importance in, for example, the processing of thermoset molding compounds (injection molding, transfer molding).
Prof. Dr. Ing. Sascha Englich is a Professor for Plastics Engineering at the Steinbeis University of Berlin. As part of the new blog series for optimization of epoxy resin injection molding by means of differential scanning calorimetry and rheology, he has already presented reports on: Thermoset Injection Molding in E-Mobility, Epoxy Resins – Reactive Polymers as a Basis for Injection-Moldable Compounds, and DSC Analysis on Thermosets. The fourth part of our blog series is all about rheokinetics.
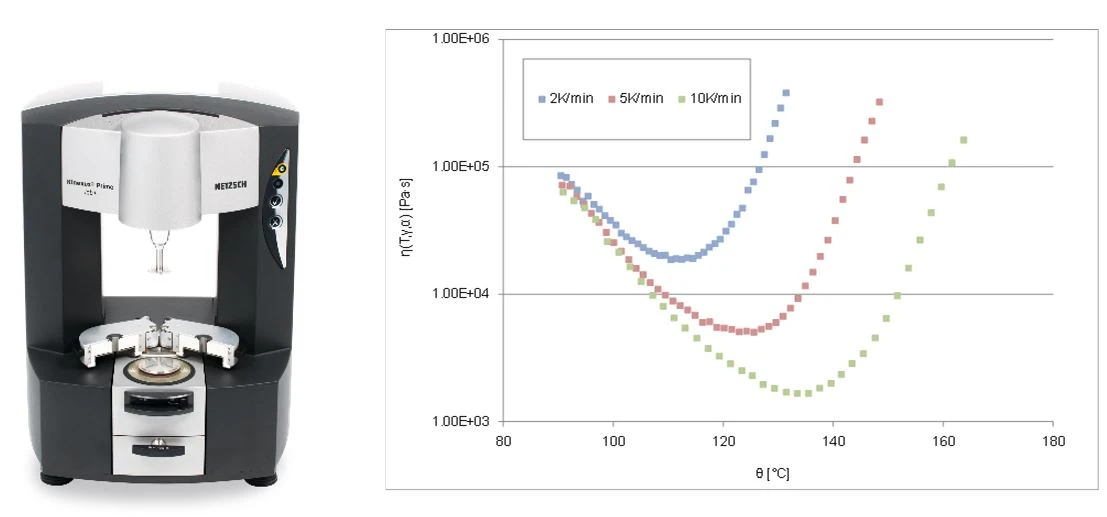
The term rheokinetics is used to describe the reaction-dependent rheological behavior of thermosetting materials, which is of great importance in, for example, the processing of thermoset molding compounds (injection molding, transfer molding). Classic simplified monitoring of the flow-curing behavior is usually based on a so-called “U” curve of viscosity/flowability, which is intended to represent the relationship between the initial decrease in viscosity due to a temperature increase and the subsequent increase in viscosity due to progressive molecular cross-linking (Figure 1).
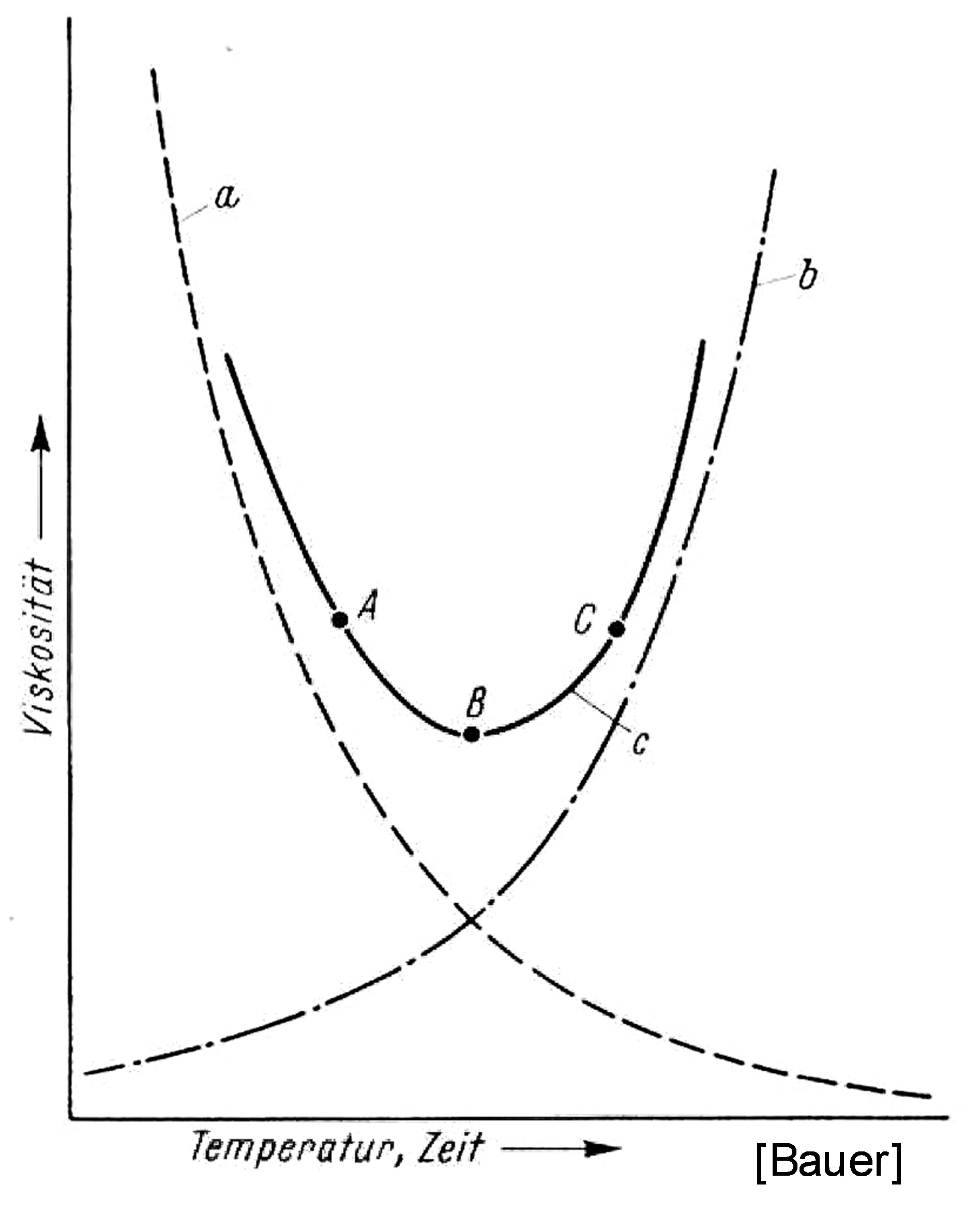
About the Flow-Curing Behavior of a Thermosetting Molding Compound
Applied to a real manufacturing process, this chemical-physical interaction leads to a much more complex process. Figure 2 depicts the schematic of the flow-curing behavior of a thermosetting molding compound (e.g., epoxy resin or phenolic resin) by means of an injection molding cycle. The material stiffness (corresponding to the viscosity in the molten state) shows characteristic curves in the individual process phases. During plasticizing, the mass temperature is systematically increased by the barrel heater and friction (screw rotation) until the Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition range of the amorphous resin is exceeded (Figure 2, yellow graph). The material is now in the molten state. Ideally, the temperature is selected/adjusted to a viscosity as low as possible, whilst also avoiding curing (cf. blog article “DSC Analysis on Thermosets – Application of the Appropriate Measurement Methodology for Different Resin Types”).
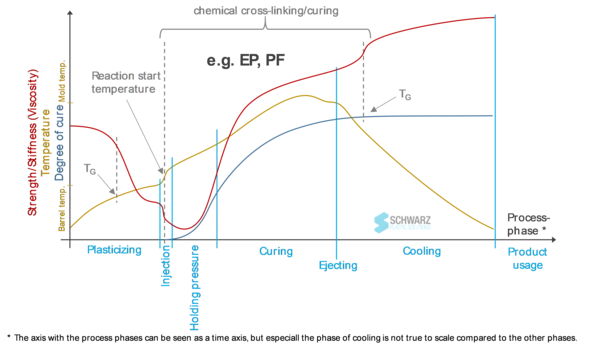
Process States During Injection Molding
During the injection phase, as the material flows through the machine nozzle, friction heats the material as closely to a relevant curing temperature in a short time. Therefore, the molecular crosslinking occurs at an accelerated rate from this point onward (Figure 2, blue curve). At the same time, this temperature rise leads to a significant reduction in viscosity. This relatively short process phase is important for process efficiency and quality since it defines mold filling (rheology) in addition to curing time (reaction dynamics). During the post-pressure phase, viscosity initially continues to decrease due to the temperature increase (mold heating including an ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermic crosslinking reaction), and finally increases as crosslinking progresses, with the material returning to the solid state (curing phase). The material/component is ejected from the mold in a hot, solid state. The threshold for the crosslinking-specific Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition temperature is first reached during cooling.

Insight into the material-specific flow-curing behavior is of fundamental importance for component and process design. For example, encapsulation of electronic components requires pressure-reduced mold filling in order to not damage sensitive electronic parts. At the same time, very narrow gaps often have to be completely filled (Figure 4). This requires materials with particularly low viscosity during mold filling (mostly special epoxy resin compounds) in connection with the appropriate mold and process design.

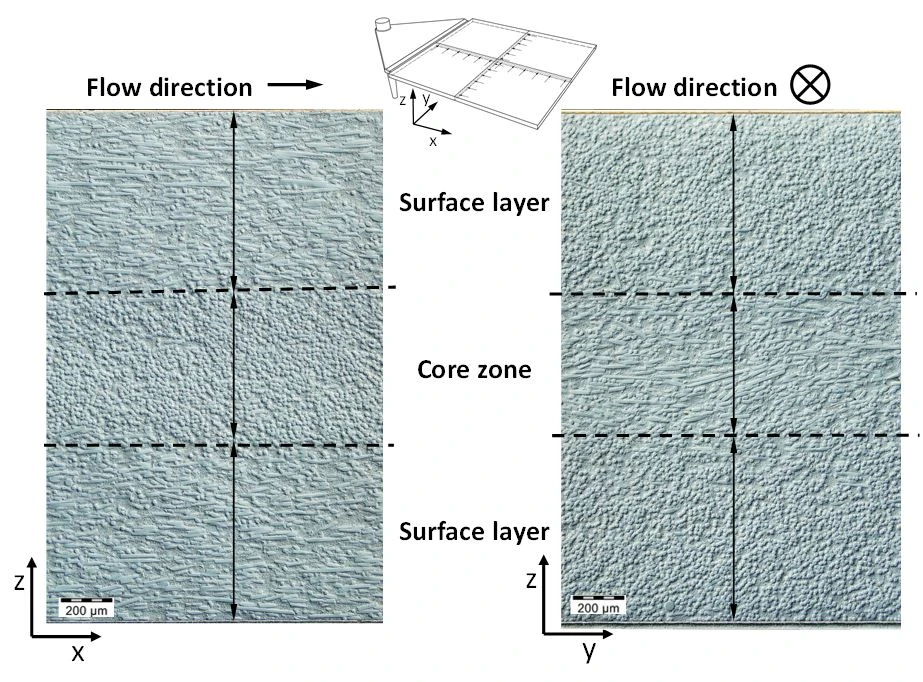
Another important point in which the rheokinetic properties play an important role is the formation of filler structure, for example fiber orientation. Two different orientations of layers are formed (Figure 5). There are two edge layers, in which the fibers are mainly oriented parallel to the flow direction (shear zones), and one core layer in which the fibers are mainly oriented perpendicular to the flow direction (lateral extension zone). The corresponding proportions of the oriented layers above the cross-section significantly influence the mechanical properties in different loading directions. These are influenced by the rheokinetic material behavior in addition to the mold and process design.
Processing Simulation Methods
Both the aforementioned application scenarios demonstrate the importance of having insight into the rheokinetic behavior of thermosetting molding compounds in order to carry out optimal material selection, mold and process design. The use of processing simulation methods (Figure 6) is also important to avoid experimental trial-and-error tests. Therefore, the rheokinetic material behavior must be determined by a combination of DSC and rheometry, and made available through mathematical models in processing simulation software as a “calculation basis”.
One option in a rheological measurement is the use of rotation/oscillation, the special feature, which will be presented in detail during analysis of thermosetting molding compounds in the next article.
One option in a rheological measurement is the use of rotation/oscillation, the special feature, which will be presented in detail during analysis of thermosetting molding compounds in the next article. Stay tuned!


