CERAMICS & GLASS
Gypsum and Quartz Sand — Phase Transitions
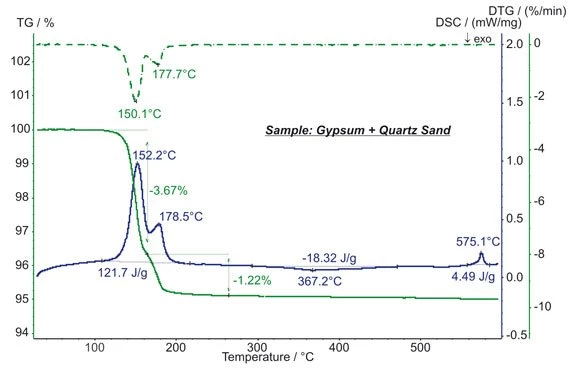
Gypsum and quartz sand are, for example, used in plaster and mortar.
The gypsum content of the sample shows a two-step release of H2O from CaSO4*2H2O (dihydrate) into CaSO4*1/2H2O (halfhydrate) and finally into CaSO4 (anhydrite). This requires an entire energy of 122 J/g. Quantitative analysis reveals that the sample contained 23.4% of pure dihydrate. Between approx. 300°C and 450°C, the ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermic formation of β-CaSO4 with a released energy of 18.3 J/g occurred. The EndothermicA sample transition or a reaction is endothermic if heat is needed for the conversion.endothermic effect at an extrapolated onset temperature of 573°C is due to the structural α→β transition of quartz (crystalline SiO2). (measurement with STA 449 F1 Jupiter®®)