CERAMICS & GLASS
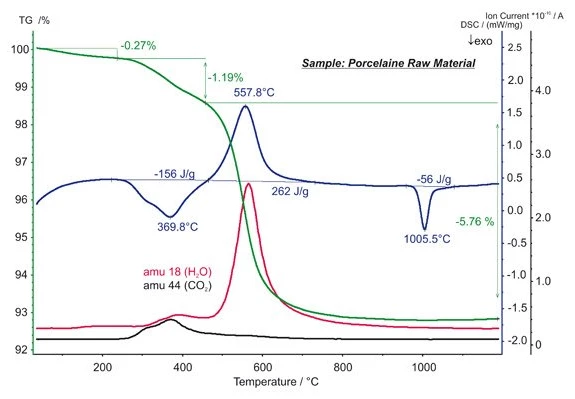
Porcelain Raw Material — Mass-Loss Steps
This STA measurement on porcelain raw material shows three mass-loss steps.
Below approx. 250°C, the evaporation of humidity occurred. At temperatures between 250°C and 450°C, the burn-up of organic content was observed, during which 156 J/g of energy was released. The dehydration of kaolin occurred above 450°C and required 262 J/g. The mass spectrometer signals for mass numbers 18 and 44 reflect the corresponding release of H2O and CO2. The ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermic DSC peak at 1006°C with an enthalpy of -56 J/g is due to an solid-solid transition. (measurement with STA 449 F3 Jupiter®®)