Glossary
Thermal Corrosion Studies
Thermal corrosion or high-temperature corrosion is a chemical interaction of a material (typically a metal) with the surrounding gas atmosphere as a result of heating.
This non-galvanic form of corrosion can occur when a material is subjected to a hot atmosphere containing oxygen, sulfur, water or other compounds capable of oxidizing (or assisting in the OxidationOxidation can describe different processes in the context of thermal analysis.oxidation of) the material examined.
The products of high-temperature corrosion can potentially be turned to the advantage of the source material. The formation of oxides on metals can provide a protective layer preventing further atmospheric attack, thus allowing a material to be used for sustained periods at both room and high temperatures under hostile conditions.
Thermal corrosion studies are typically conducted via Thermogravimetric Analysis (TGA).
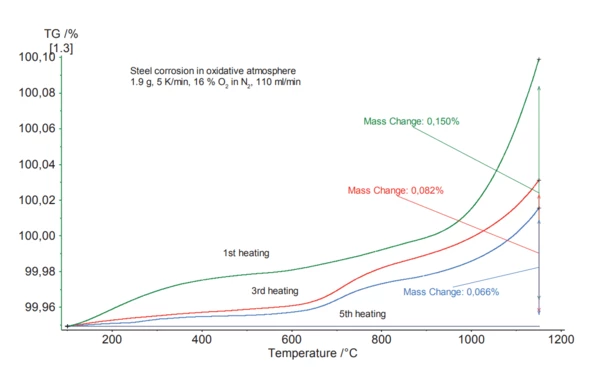
The figure below shows several heating cycles on a hanging steel sample. The steel sheet was heated at a rate of 5 K/min in a nitrogen atmosphere with 16% oxygen. OxidationOxidation can describe different processes in the context of thermal analysis.Oxidation (mass increase) decreases with each subsequent heating cycle. At the beginning of the test, OxidationOxidation can describe different processes in the context of thermal analysis.oxidation of the sheet surface occurs. This can be observed in the early onset and rapid mass increase for the first heating. After a couple of heating cycles, inner OxidationOxidation can describe different processes in the context of thermal analysis.oxidation occurs, which is indicated by a slower, diffusion-dependent mass increase.
This figure shows several heating cycles on a hanging steel sample. The steel sheet was heated at a rate of 5 K/min in a nitrogen atmosphere with 16% oxygen. OxidationOxidation can describe different processes in the context of thermal analysis.Oxidation (mass increase) decreases with each subsequent heating cycle. At the beginning of the test, OxidationOxidation can describe different processes in the context of thermal analysis.oxidation of the sheet surface occurs. This can be observed in the early onset and rapid mass increase for the first heating. After a couple of heating cycles, inner OxidationOxidation can describe different processes in the context of thermal analysis.oxidation occurs, which is indicated by a slower, diffusion-dependent mass increase.