Photovoltaïques
Uncured EVA — Determination of Glass Transition Temperature
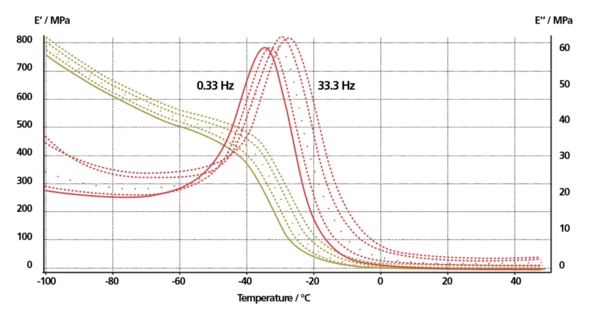
These DMA tests were carried out at the Federal Institute for Materials Research and Testing ("BAM"), Germany*. The multi-frequency measurement (0.33 Hz, 1 Hz, 3.33 Hz, 10 Hz and 33.3 Hz) was performed in the dual cantilever sample holder at a heating rate of 2 K/min and an amplitude of 40 μm.
The observed behavior of the Température de Transition VitreuseThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition is typical. The storage modulus E’ strongly decreases at -40°C while E’’ shows a clear peak. The glass transition is a function of frequency: the higher the frequency, the higher the glass transition temperature.
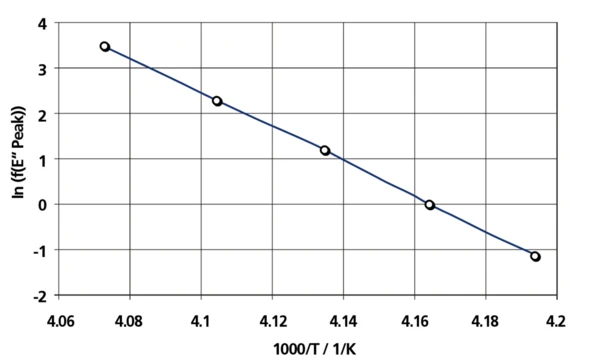
These values are used to determine the activation energy of the glass transition temperature. A linear correlation is found between ln(f) and 1/T. From the slope of the straight line, an apparent activation energy can be calculated. This activation energy amounts to 328 kJ/mol, a value in the range typical for a glass transition.
*Our thanks to Dr. W. Stark and M. Jaunich from the Federal Institute for Materials Research and Testing ("BAM") in Berlin for the measurements and discussion. The results are published in Polymer Testing 30 (2011) 236-242.