소개
다양한 물질을 열처리하면 암모니아가 방출될 수 있는데, 암모니아는 자극적인 냄새가 나며 기관지를 공격할 수 있습니다. 암모니아 방출은 다양한 공정에 의해 발생할 수 있습니다. 여기에는 소금의 열분해부터 담배를 태울 때 발생하는 연기, 폴리아미드(PA)와 같은 폴리머의 열분해, 발포제가 필요한 플라스틱 폼 제조에 이르기까지 다양합니다. 후자의 대표적인 제품으로는 요가 매트가 있습니다. 암모니아가 방출되면 염분이 형성되면 황산 및 질산과 반응하여 미세먼지를 생성할 수 있습니다. 환경에서 암모니아의 방출은 토양의 산성화로 이어질 수 있습니다. 환경에서 암모니아의 주요 공급원 중 하나는 농업, 특히 배설물과 질소 함유 광물질 비료를 사용한 시비입니다.
이러한 이유로 방출된 암모니아의 정량화는 많은 응용 분야에서 중요하게 여겨져 왔습니다. 암모니아의 온도 의존적 방출은 TGA-FT-IR 결합을 통해 쉽게 감지할 수 있습니다. 방출된 암모니아 부분을 정량화하려면 알려진 암모니아 농도를 가진 보정 곡선이 필요합니다. 중탄산암모늄은 물과 이산화탄소의 방출과 더불어 암모니아를 화학량 론적 비율로 방출하기 때문에 이에 적합한 화합물입니다(방정식 (1) 참조). 기체 화합물만 생성됩니다:
(1) NH4HCO3->NH3 + H2O+ CO2
보정 커브 생성 방법
NETZSCH PERSEUS® TG 209 F1 Libra® 이 연구를 수행하기 위해 사용되었습니다. 중탄산암모늄을 가열하면 200°C까지 완전히 분해되었으며, 질량 손실률은 127°C에서 정점을 찍었습니다(표 1의 측정 조건 기준).
표 1: 캘리브레이션 곡선 생성을 위한 측정 조건
| 파라미터 | NH4HCO3 | |||
| 온도 프로그램 | RT - 200°C, 5 K/min | |||
| 유량 | 40 ml/min | |||
| 시료 홀더 | 표준 시료 캐리어 | |||
| 가스 분위기 | 질소 | |||
| 도가니 | Al2O3 (85μl) 개방 | |||
| 시료 질량 | 5.31 mg | 10.16 mg | 15.01 mg | 20.50 mg |
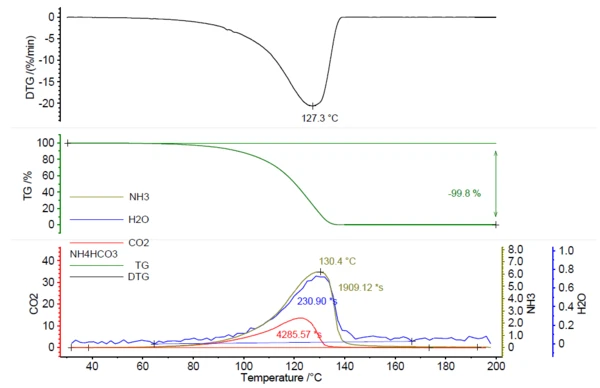
질량 손실과 상관관계가 있는 IR 활성 가스의 방출이 감지되었습니다(그림 1 참조).
그림 2는 암모니아(올리브), 물(파란색), 이산화탄소(빨간색)의 라이브러리 스펙트럼과 연계하여 130°C(녹색)에서 측정된 FT-IR 스펙트럼을 보여줍니다. 이러한 화합물의 온도 의존적 방출은 그림 1에 트레이스 형태로 표시되어 있습니다. 이러한 흔적은 전체 온도 범위에 걸쳐 각 화합물에 대한 FT-IR 스펙트럼의 색상 영역(그림 2 참조)을 통합하여 생성되었습니다. FT-IR 스펙트럼의 이러한 영역은 NH3 (898 cm-1 - 981 cm-1),CO2 (2200 cm-1 - 2450 cm-1) 및 H2O(3793 cm-1 - 4001 cm-1)에 대해 분리되어 있으며 다른 화합물의 영역과 겹치지 않았습니다.
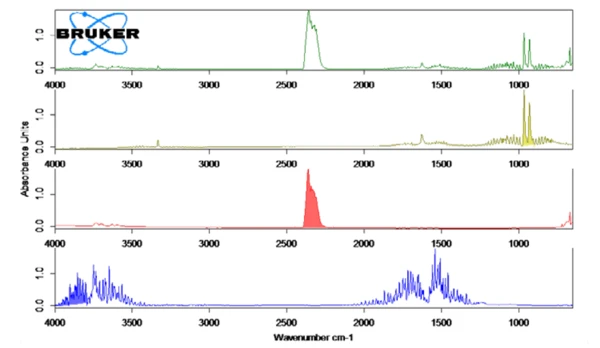
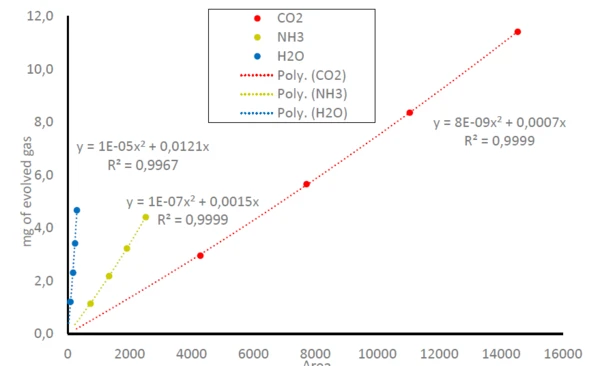
표 2는 중탄산암모늄의 시료 질량과 관련하여 화학량 론적으로 계산된 방출된 기체의 양을 보여줍니다.
따라서 그림 1에서 NH3,CO2 및 H2O의미량 아래 영역은 방출된 가스의 양과 관련이 있을 수 있으며, 방정식 (1)을 비교하세요. 이는 NH3,CO2 및 H2O에대한 보정 곡선으로 이어지며, 추적 아래 감지된 영역은 방출된 가스의 양과 관련이 있습니다(그림 3 참조). FT-IR은 선형 범위가 small 밖에 없기 때문에 세 가지 기체 분자 모두에 대해 측정 계수(R2)가 1에 매우 가까운 다항식 방정식이 도출됩니다. 이 연구에서는 각 샘플 질량을 한 번만 측정했습니다. 반복 측정 또는 더 많은 데이터 포인트를 사용하면 추세선의 정확도가 훨씬 더 높아질 것입니다.
표 2: 시료 질량과 그에 따른 화학량 론적 기체 양
| m (NH4HCO3) [mg] [mg] | m (CO2) [mg] | m (NH3) [mg] | m (H2O) [mg] |
| 5.31 | 2.96 | 1.14 | 1.21 |
| 10.16 | 5.66 | 2.19 | 2.31 |
| 15.01 | 8.36 | 3.23 | 3.42 |
| 20.50 | 11.42 | 4.41 | 4.67 |
보정 곡선의 정확도를 테스트하는 방법
보정 곡선의 정확도는 시료 질량이 15.22mg으로 정의된 NH4HCO3에 대한 또 다른 측정으로 확인했습니다. NH3,CO2 및 H2O의이론적 양을 보정 곡선을 사용하여 계산된 NH3,CO2 및 H2O의값과 비교했습니다. 그 결과 NH3의 경우 0.8%, H2O의경우 4.9%의 오차 값이 산출되었습니다(표 3 참조).
블로잉 에이전트 연구 - 이론을 실제에 적용하기
다음 단계에서는 획득하고 검증한 보정 곡선을 사용하여 보정된 가스의 미량 방출을 정량화할 수 있습니다.
아조디카보나미드는 폴리머 폼을 생산하기 위한 발포제로 사용됩니다(구조는 그림 4 참조). PVC 폼과 EVA-PE 폼 제조에 사용되며, 가공 온도에서 분해될 때 N2, CO,CO2 및 NH3를 방출하면서 기포를 형성합니다. 비닐 폼은 쉽게 압축되고 높고 빠른 회복력을 나타내므로 흔히 '탄성'이라고 불립니다. 또한 매끄러운 표면에도 잘 달라붙습니다. 이러한 이유로 카펫 언더레이, 바닥 매트 및 요가 매트 등에 사용됩니다.
이 발포제가 사용된 폴리머는 물과 접촉할 수 없습니다. NH3와 물은 NH4OH를형성하여 주변을 부식시킬 수 있습니다. 이러한 이유로 이 발포제에서 나오는 암모니아의 정량화는 큰 관심사입니다.
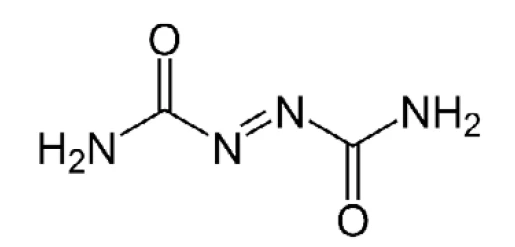
표 3: 오차 결정, 이론적 금액과 계산된 금액의 비교
이론적(mg) | 계산된 양(mg) | 오차(%) | |
| m (NH4HCO3) | 15.22 | ||
| m (NH3) | 3.28 | 3.30 | 0.801 |
| m (CO2) | 8.48 | 8.76 | 3.28 |
| m (H2O) | 3.47 | 3.31 | 4.86 |
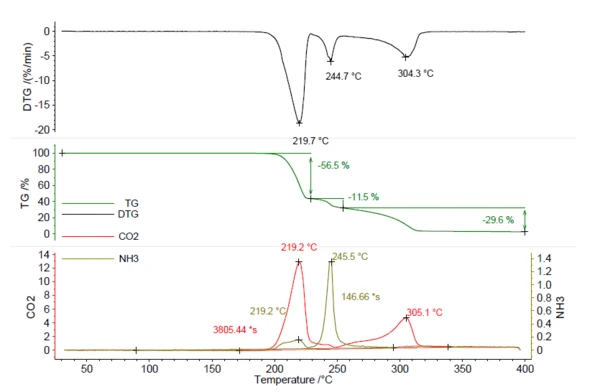
5.25mg 아조디카르본아미드 샘플을 질소 분위기에서 5K/min으로 400°C까지 가열했습니다. 결과 열화상은 그림 5에서 볼 수 있습니다. 총 56.5%, 11.5%, 29.6%의 세 가지 질량 손실 단계가 219°C, 245°C, 304°C에서 DTG 곡선의 피크와 함께 관찰되었습니다.CO2와NH3의 흔적은 그림 1의 NH4HCO3와 동일한 방식으로 생성되었으며 빨간색과 올리브색으로 표시되어 있습니다. 이는CO2와NH3 모두 다양한 질량 손실 단계에서 방출되었으며 TGA 단계만으로는 정량화할 수 없음을 보여줍니다. 이 화합물의 정량화를 위해서는 진화 가스 분석 데이터가 필요합니다. 보정 곡선을 사용하여 방출된 암모니아를 계산한 결과 0.22mg NH3 (4%)가 나왔습니다. 또한 같은 방법으로 방출된이산화탄소의 양을 계산한 결과 2.78mg(53%)이 나왔습니다. 이 지식은 발포 중에 발포제의 전체 양이 방출되는지 확인하기 위해 제조 공정에 유용합니다. 제품에 small 미량이 남아 있는 경우, 추가 방출을 시작하려면 219°C 이상의 온도가 필요합니다.
결론
열 중량 측정과 적외선 분광법의 조합은 물, 이산화탄소 및 암모니아와 같은 영구 가스의 방출을 감지하는 데 적합한 방법입니다. 여기서 관심을 가질 수 있는 것은 식별뿐만 아니라 정량화입니다. 이를 위해서는 알려진 물질로 보정 곡선을 생성해야 합니다. 이 예에서는 중탄산암모늄이 이러한 요구 사항을 완벽하게 충족합니다. NH4HCO3의 세 가지 다른 부분을 분해하여 H2O,CO2 및 NH3에 대한 교정 곡선을 동시에 생성할 수 있습니다. 예상되는 편차는 다섯 번째 측정으로 결정되었습니다. 이 준비를 통해 폴리머 폼에 사용되는 발포제 아조디카본아미드에서 미량의 NH3와CO2를 식별하고 정량화할 수 있었습니다.