Введение
Термический анализ включает в себя методы определения физических и химических свойств или изменений свойств в зависимости от температуры. Термогравиметрия позволяет количественно оценить изменения массы, например, выделение реакционных газов и газов разложения. Когда эти газы попадают в газоизмерительную ячейку, становится возможной идентификация выделившихся газов. Так называемое соединение TGA-FT-IR представляет собой проверенную комбинацию аналитического и спектроскопического методов анализа.
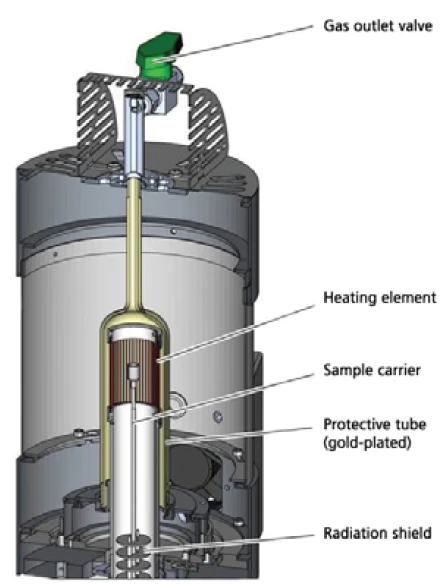
Совершенно новым дополнением к хорошо зарекомендовавшей себя установке STA 449 F1 Jupiter® (рис. 1) является высокоскоростная печь (сечение на рис. 2), которая может работать со скоростью нагрева до 1000 К/мин (в настоящее время доступны системы печей для самых разных применений, охватывающие диапазон температур от -150°C до 2400°C).
Влияние скорости нагрева и связанной с ней скорости высвобождения на результаты термогравиметрических и спектроскопических измерений будет рассмотрено в этой заметке.

Результаты
a) Полипропилен PP
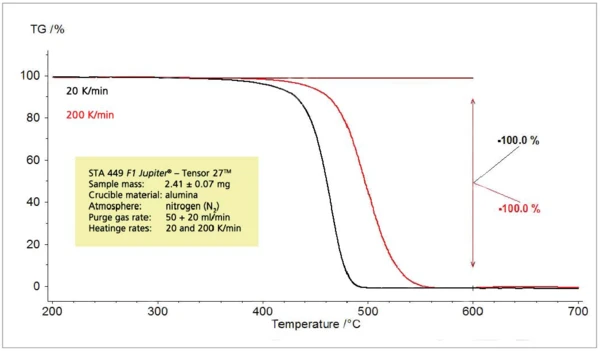
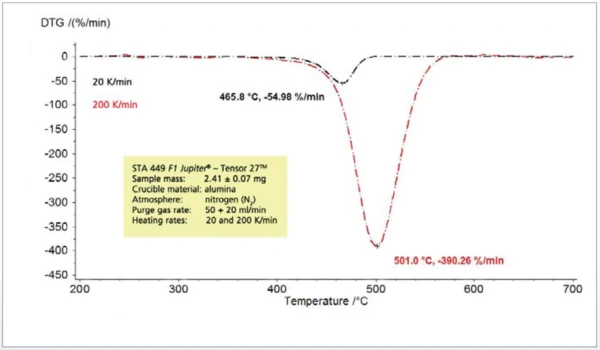
При изменении скорости нагрева в ходе термоаналитических экспериментов обнаруженные эффекты смещаются в сторону более высоких температур с увеличением скорости нагрева (рис. 3). Это хорошо известно и может быть использовано для кинетических оценок. С повышением температуры высвобождения скорость высвобождения также значительно возрастает (рис. 4). Поэтому концентрация анализируемых газов в постоянном потоке газа-носителя также возрастает, и анализируемые газы можно легко обнаружить и идентифицировать. Однако этапы потери массы не зависят от скорости нагрева.
б) CaCO3
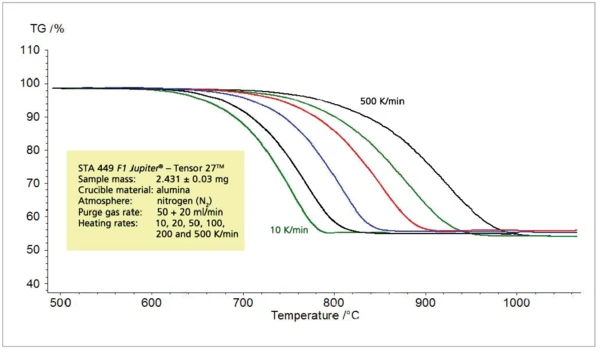
Зависимость между скоростью нагрева и температурой разложения при постоянной высоте ступеньки, рассмотренная для пиролиза пропилена, прослеживается и при термическом разложении карбоната кальция на оксид кальция и диоксид углерода (рис. 5 и 6).
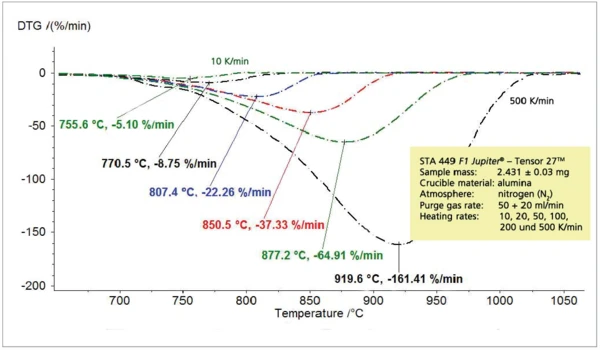
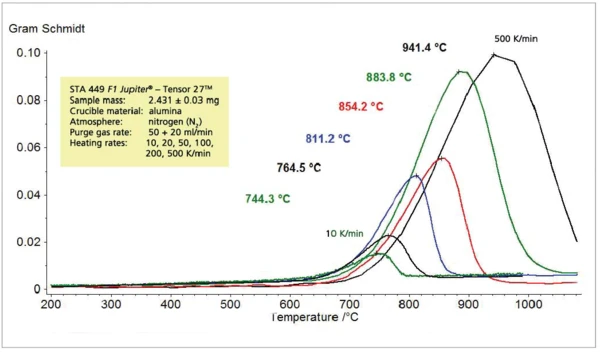
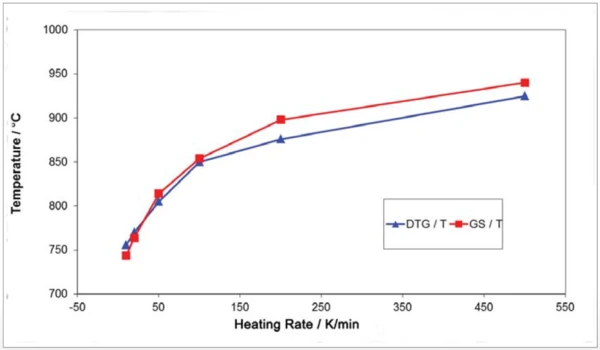
На рисунке 7 показана интенсивность поглощения соответствующих следов Грама-Шмидта, которая, как ожидается, будет увеличиваться с ростом скорости нагрева. Следует отметить, что перенос выделившихся газов образца в газовую ИК-измерительную ячейку практически не задерживается из-за высокой скорости нагрева. Это видно из сравнения максимальной скорости высвобождения (DTA) с максимальной интенсивностью ИК-излучения (GS) на рисунке 8.
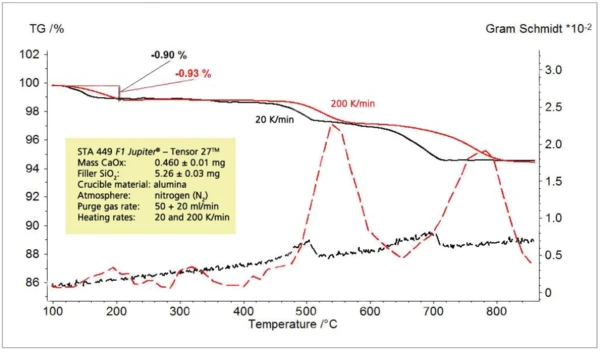
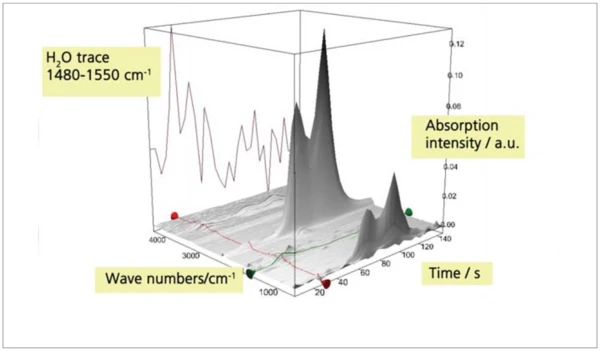
c) CaC2O4 x H2Oв смеси с SiO2
Для исследования предела обнаружения была приготовлена смесь моногидрата оксалата кальция (CaC2O4 x H2O) и кварцевого песка (SiO2). Соотношение смешивания selected составляло 1:10, так что ожидаемое выделение воды соответствовало примерно 1% от массы образца. Термическое выделение примерно 1 % воды из этой смеси не было обнаружено при скорости нагрева 20 К/мин; однако при скорости нагрева 200 К/мин оно было четко обнаружено (рис. 9-11).

Резюме
С помощью быстрых скоростей нагрева до 500 К/мин можно значительно увеличить скорость выделения газообразных продуктов из образца. Таким образом, их концентрация также увеличивается по сравнению с газом-носителем, что приводит к значительному улучшению предела обнаружения при использовании TGA-FT-IR-соединения.