Introduzione
I PCM (Phase Change Materials) sono materiali utilizzati come sistemi di accumulo di calore latente. L'entalpia della Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase da solido a liquido viene quindi utilizzata per l'accumulo di calore. Il campo di applicazione dei sistemi di accumulo di calore latente spazia dai riscaldatori tascabili ai tessuti funzionali, fino agli elementi per pareti e soffitti nell'edilizia. Le proprietà termofisiche di un campione di PCM di estratti vegetali sono state studiate con l'aiuto dell'LFA 467 HyperFlash® e del DSC 204 F1 Phoenix® .
Condizioni di prova
LFA:

- campione solido da 30°C a 150°C nel portacampioni standard (riscaldamento)
- da 220°C a 30°C campione liquido nel portacampioni in PEEK (raffreddamento), vedi figura 1

DSC:

- -riscaldamento e raffreddamento da 10°C a 225°C
Risultati della misurazione
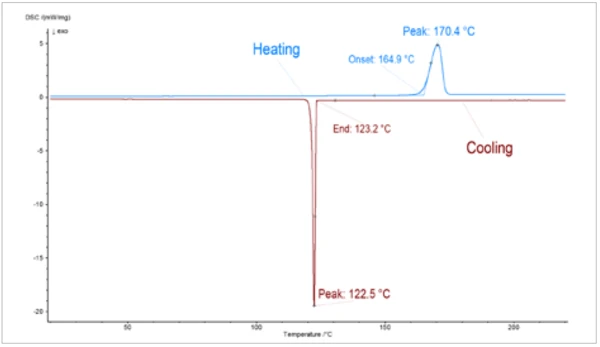
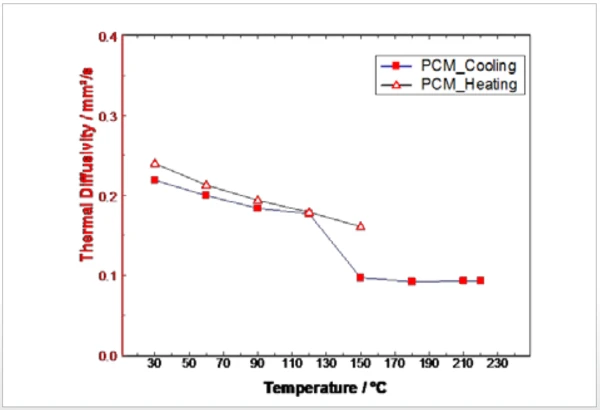
La Figura 2 mostra il riscaldamento e il raffreddamento del campione di PCM mediante DSC. La Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione del campione inizia a circa 165°C (onset), mentre la CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione durante il raffreddamento ricomincia solo a circa 123°C. Questo effetto è visibile anche per le misure LFA. I quadrati rossi della figura 3 rappresentano la Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica del campione PCM durante il raffreddamento (da liquido a solido). L'aumento della Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica può essere correlato alla Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase. Poiché i punti di misurazione sono stati registrati durante il raffreddamento, la Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase appare tra 120°C e 150°C. I triangoli rossi della figura 3 rappresentano la Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica durante il riscaldamento del campione di PCM. Entrambe le misure sono in buon accordo tra loro. Solo a 150°C si nota una differenza significativa, attribuibile ai diversi stati dei campioni (liquido e solido) derivanti dalle diverse temperature di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione e CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione.
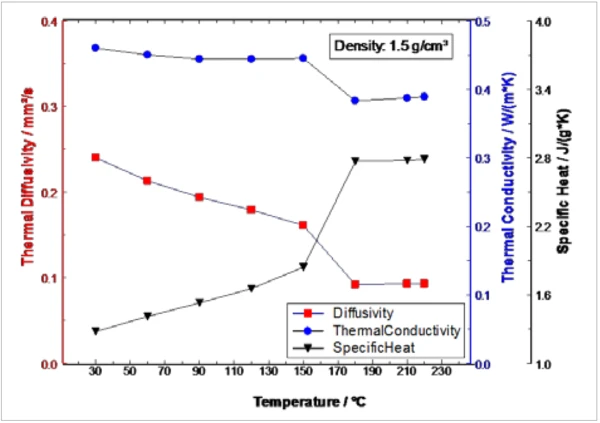
La Figura 4 mostra le proprietà termofisiche durante il riscaldamento del campione di PCM tra 30°C e 220°C come combinazione delle due misure. La Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase da solido a liquido può essere chiaramente identificata nella Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica, così come nella Capacità termica specifica (cp)La capacità termica è una grandezza fisica specifica del materiale, determinata dalla quantità di calore fornita al campione, divisa per l'aumento di temperatura risultante. La capacità termica specifica è correlata all'unità di massa del campione. capacità termica specifica e nella conducibilità termica attraverso un passaggio tra 150°C e 180°C.
Sintesi
Lo speciale portacampioni per liquidi e paste (portacampioni in PEEK) consente di studiare la Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica dei campioni di PCM anche nella Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione, mediante LFA. Le misure comparative con e senza il portacampioni per liquidi nell'intervallo dei solidi sono in buon accordo, purché vi sia un buon contatto tra il campione e il portacampioni (analisi a 3 strati). Le misure DSC consentono di trarre conclusioni sul comportamento di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione e CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione dei campioni e forniscono dati sulla Capacità termica specifica (cp)La capacità termica è una grandezza fisica specifica del materiale, determinata dalla quantità di calore fornita al campione, divisa per l'aumento di temperatura risultante. La capacità termica specifica è correlata all'unità di massa del campione. capacità termica specifica. Dalle misurazioni di entrambi i metodi si possono poi trarre affermazioni affidabili sulla conducibilità termica del campione PCM nell'intervallo solido e liquido.