L'importance des matériaux thermoélectriques et leurs possibilités d'application
L'optimisation de l'efficacité énergétique est l'un des principaux défis du21e siècle. Dans de nombreuses applications industrielles, d'énormes quantités d'énergie thermique inutilisée sont générées. Cette chaleur résiduelle est produite par les fours de Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion, les usines d'incinération, les centrales électriques et même les véhicules à moteur - et pourrait être utilisée pour produire de l'énergie électrique. En plus d'avoir un impact positif sur l'environnement, cela contribuerait également de manière significative à l'augmentation de l'efficacité et de la rentabilité globales des installations industrielles. C'est là que la thermoélectricité entre en jeu.
les "générateurs thermoélectriques", comme on les appelle, sont développés et peuvent être utilisés dans tous les domaines où il existe des différences de température utilisables. De telles applications nécessitent le développement de matériaux thermoélectriques à haut rendement.
Détermination des propriétés thermophysiques et thermoélectriques
Pour le développement et l'optimisation des matériaux thermoélectriques, la connaissance des propriétés thermophysiques et thermoélectriques est essentielle. Pour évaluer l'efficacité, on utilise le chiffre de mérite (valeur ZT). Ce chiffre thermoélectrique décrit dans quelle mesure un matériau particulier est adapté ou non à une utilisation dans un générateur thermoélectrique. La valeur ZT fournit donc des informations sur l'efficacité du matériau.
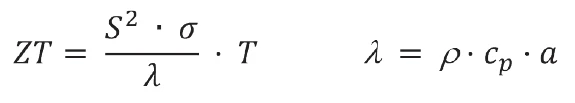
Avec le Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique. SBA 458 Nemesis®, le Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck, S, et la Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique.conductivité électrique, σ, peuvent être déterminés simultanément. Le LFA permet de mesurer directement la Capacité thermique spécifique (cp)La capacité thermique est une grandeur physique spécifique au matériau, déterminée par la quantité de chaleur fournie à l'échantillon, divisée par l'augmentation de température qui en résulte. La capacité thermique spécifique est liée à une unité de masse de l'échantillon.capacité thermique spécifique, Capacité thermique spécifique (cp)La capacité thermique est une grandeur physique spécifique au matériau, déterminée par la quantité de chaleur fournie à l'échantillon, divisée par l'augmentation de température qui en résulte. La capacité thermique spécifique est liée à une unité de masse de l'échantillon.cp, et la Diffusivité thermiqueLa diffusivité thermique (a avec l'unité mm2/s) est une propriété propre au matériau qui permet de caractériser la conduction thermique instable. Cette valeur décrit la rapidité avec laquelle un matériau réagit à un changement de température.diffusivité thermique, a. Avec la densité, ρ, la Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique, λ, peut être calculée.
Avec le Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique. SBA 458 Nemesis® et les appareils à flash laser LFA 427, LFA 457 et LFA 467, NETZSCH offre une solution complète pour la détermination de la valeur ZT.
La skutterudite, un matériau approprié pour les applications thermoélectriques
Actuellement, les coûts énormes de développement et l'efficacité actuellement faible des matériaux thermoélectriques empêchent souvent leur application. Pour y remédier, l'efficacité des matériaux thermoélectriques doit être considérablement augmentée grâce à de nouveaux développements et à des modifications.
L'objectif est de développer des matériaux présentant une faible Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique, λ, avec simultanément une conductivité élevée, σ, et un Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck élevé, S. La difficulté réside dans le fait que ces trois propriétés ne peuvent être influencées indépendamment l'une de l'autre que dans certaines conditions.
La skutterudite, en particulier, peut présenter d'excellentes propriétés électriques. La skutterudite est un matériau composé de cobalt et d'arsenic, souvent contaminé par des terres rares. Elle appartient à la classe des sulfures. Elle doit son nom à la ville de Skutterud en Norvège, où ce minéral naturel, le CoAs3, a été découvert pour la première fois en 1928. Ce n'est qu'au milieu des années 50 que ses excellentes propriétés électriques ont été reconnues. La skutterudite se caractérise par une mobilité très élevée des porteurs de charge et un Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck de la taille de medium. Sa Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique, en revanche, est beaucoup trop élevée pour permettre son utilisation efficace dans des applications thermoélectriques à cette époque. Dans les années 70, on a découvert la structure cristalline typique de la skutterudite, qui peut être modifiée de manière optimale. Deux vides dans la cellule élémentaire peuvent être comblés par l'insertion d'atomes étrangers. De cette manière, la Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique de la skutterudite peut être réduite. Depuis lors, les skutterudites sont des candidats potentiels pour des convertisseurs thermoélectriques plus efficaces avec lesquels, par exemple, la chaleur résiduelle des systèmes d'échappement des automobiles peut être directement convertie en électricité. Les exemples de mesures suivants montrent comment la valeur ZT de la skutterudite peut être déterminée à l'aide d'un seul échantillon.
Mesures de l'AFB
Pour le calcul de la valeur ZT sans dimension de la skutterudite, la Diffusivité thermiqueLa diffusivité thermique (a avec l'unité mm2/s) est une propriété propre au matériau qui permet de caractériser la conduction thermique instable. Cette valeur décrit la rapidité avec laquelle un matériau réagit à un changement de température.diffusivité thermique (figure 1, courbe rouge) et la Capacité thermique spécifique (cp)La capacité thermique est une grandeur physique spécifique au matériau, déterminée par la quantité de chaleur fournie à l'échantillon, divisée par l'augmentation de température qui en résulte. La capacité thermique spécifique est liée à une unité de masse de l'échantillon.capacité thermique spécifique (figure 1, courbe noire) ont été déterminées avec le LFA 467 HyperFlash® sur un échantillon d'un diamètre de 12,7 mm. Les mesures ont été effectuées entre la température ambiante et 400°C.
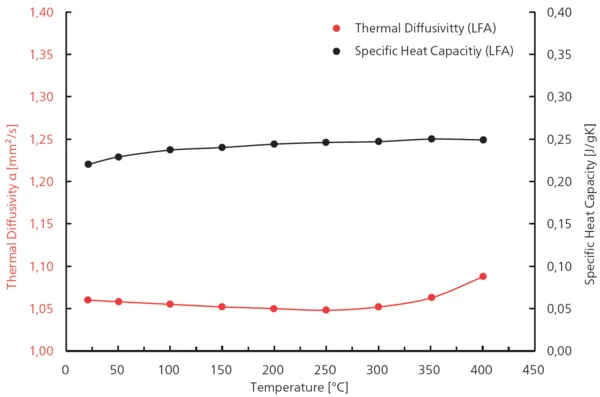
Le calcul de la Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique est basé sur les résultats obtenus à l'aide de l'équation suivante : λ = a-Capacité thermique spécifique (cp)La capacité thermique est une grandeur physique spécifique au matériau, déterminée par la quantité de chaleur fournie à l'échantillon, divisée par l'augmentation de température qui en résulte. La capacité thermique spécifique est liée à une unité de masse de l'échantillon.cp-ρ(voir figure 2).
Mesure de l'ASB
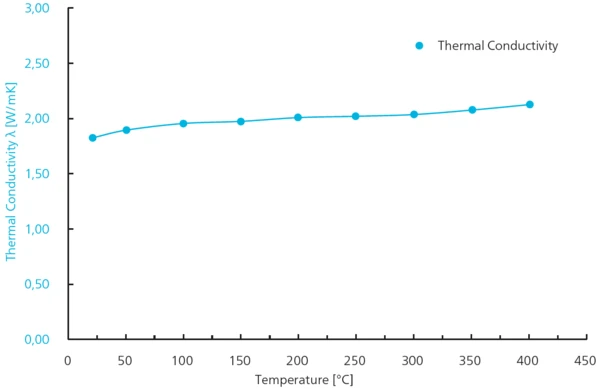
Avec le Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique. SBA 458 Nemesis®, le Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck et la Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique.conductivité électrique de l'échantillon déjà utilisé pour la mesure LFA ont été déterminés entre RT et 350°C. Le Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck a augmenté de 100 μV/K à près de 160 μV/K tandis que la Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique.conductivité électrique a diminué d'environ 1300 S/cm à 1000 S/cm. Les résultats des mesures présentent une excellente reproductibilité (± 2 %) pour les deux paramètres (voir figure 3).
Valeur ZT
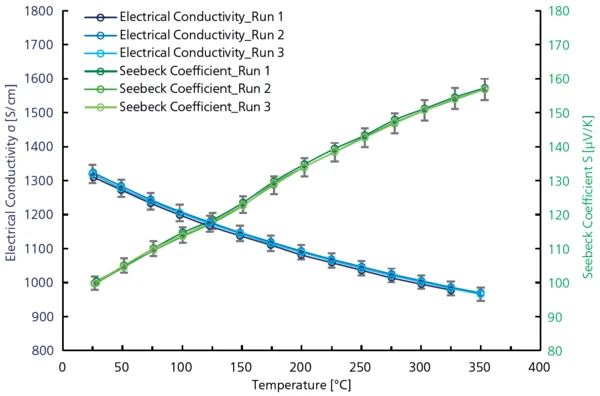
La valeur ZT est calculée à partir des résultats obtenus avec le LFA et le Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique. SBA sur le même échantillon (voir figure 4) en utilisant l'équation suivante :
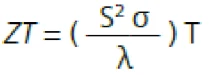

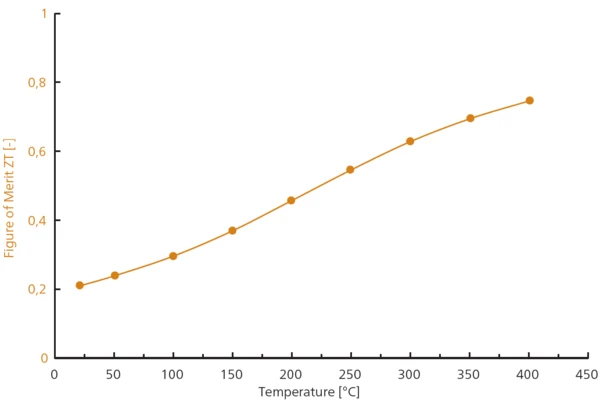
Le graphique de la figure 5 représente l'augmentation de la valeur ZT entre la température ambiante et 400°C avec un maximum à 0,75.
Résumé
Il a été démontré que les propriétés thermophysiques - y compris la Diffusivité thermiqueLa diffusivité thermique (a avec l'unité mm2/s) est une propriété propre au matériau qui permet de caractériser la conduction thermique instable. Cette valeur décrit la rapidité avec laquelle un matériau réagit à un changement de température.diffusivité thermique et la Conductivité thermiqueLa conductivité thermique (λ avec l'unité W/(m-K)) décrit le transport d'énergie - sous forme de chaleur - à travers un corps de masse sous l'effet d'un gradient de température (voir fig. 1). Selon la deuxième loi de la thermodynamique, la chaleur s'écoule toujours dans la direction de la température la plus basse.conductivité thermique, la Capacité thermique spécifique (cp)La capacité thermique est une grandeur physique spécifique au matériau, déterminée par la quantité de chaleur fournie à l'échantillon, divisée par l'augmentation de température qui en résulte. La capacité thermique spécifique est liée à une unité de masse de l'échantillon.capacité thermique spécifique, le Coefficient SeebeckLe coefficient Seebeck est le rapport entre la tension thermoélectrique induite et la différence de température entre deux points d'un conducteur électrique.coefficient Seebeck et la Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique.conductivité électrique - peuvent être déterminées à l'aide d'un seul échantillon. L'utilisateur gagne ainsi un temps précieux puisqu'il n'est pas nécessaire de préparer l'échantillon pour en ajuster la géométrie.