POLYMERS
Epoxy Resin — Melting & Curing
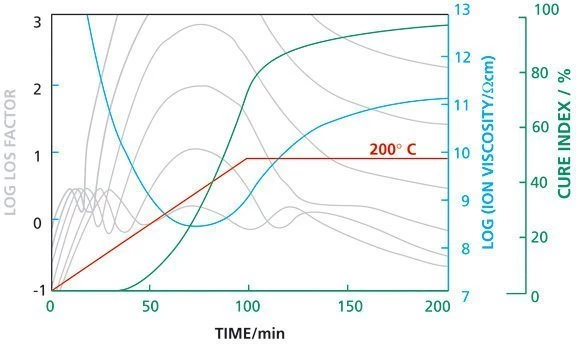
As temperature is ramped, the multi-frequency loss factor ε" shows a series of dipole RelaxationWhen a constant strain is applied to a rubber compound, the force necessary to maintain that strain is not constant but decreases with time; this behavior is known as stress relaxation. The process responsible for stress relaxation can be physical or chemical, and under normal conditions, both will occur at the same time. relaxation peaks as the epoxy resin passes through its Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition temperature.
The loss factor then rises rapidly as the epoxy melts, reflecting the dramatically increasing Ionic mobility in the resin. The Ion ViscosityIon viscosity is the reciprocal value of the ion conductivity, which is calculated from the dielectric loss factor.ion viscosity curve is derived from the Ionic mobility component of the loss factor and is a frequency independent parameter related to the viscosity of the polymer before gelation and to rigidity after gelation. The Ion ViscosityIon viscosity is the reciprocal value of the ion conductivity, which is calculated from the dielectric loss factor.ion viscosity initially decreases reflecting the effect of increasing temperature on the dynamic viscosity, of the resin. The initiation of reaction, however, competes with the temperature effect by restricting mobility and results in a clearly defined viscosity rises, reflecting the increasing viscosity and cure state of the material. To illustrate the Degree of CureThe degree of curing describes the conversion achieved during crosslinking reactions (curing). degree of cure, the dielectric cure index may be utilized. (measurement with DEA 288)