POLYMERS
Polyamide (PA) — Decomposition
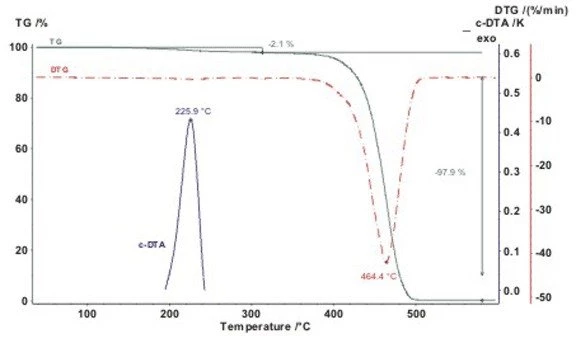
With the help of the c-DTA®® calculated DTA signal, endothermal and ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal effects can be identified.
In the following graph, the 2-step Decomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition of a polyamide (PA) sample in a nitrogen atmosphere is presented. The melting peak, determined byc-DTA®® to be at 226°C, identifies the sample as PA6. (measurement with TG 209 F1 Libra®®)
It should be kept in mind when processing this material that the first mass loss step occurs already during melting; this is apparently the release of processing additives.