Introduction
The Identify DSC curve recognition and database system is a novel and powerful software tool for identification of unknown samples and for quality control. One general issue of DSC is the dependence of the DSC curve on the sample mass and the heating rate applied. Higher values of the sample mass and also of the heating rate both tend to shift caloric effects like glass transitions or melting peaks to higher temperatures. The purpose of this study was to determine how Identify search results are affected by such temperature shifts.
Typical Example
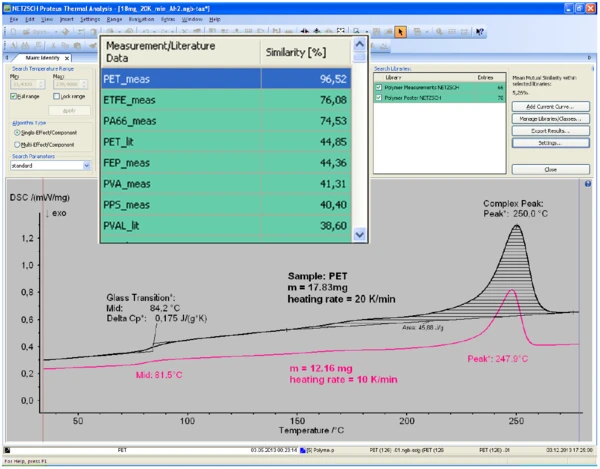
Figure 1 illustrates Identify results for a 17.83 mg PET sample measured at a heating rate of 20 K/min (2nd heating after cooling at 10 K/min). The DSC curve was correctly identified as that of PET, even though the database curve was measured at a different heating rate (10 K/min) on a different PET sample mass (12.16 mg). Obviously, the shifts in the Température de Transition VitreuseThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition and melting peak temperatures due to the different measurement conditions had only minor impact on the search result: the similarity between both curves is 96.5%, an almost perfect match!
Systematic Study
The impact of the measurement conditions of ‘sample mass’ and ‘heating rate’ on the DSC curve and, hence, the Identify result was systematically studied for HDPE. Five different HDPE samples with masses of 1, 5, 10, 15 and 20 mg were heated from room temperature to 200°C at a rate of 10 K/min over two heating cycles.
Figure 2 shows how the melting peaks of the 2nd heating curves shifted to higher temperatures and became broader with increasing sample mass – as expected. If we consider the curve obtained for the 10-mg sample as a reference, high similarity values between this curve and the curves for 1, 5, 15 and 20 mg can be observed (see table in figure 2). For Identify, the curves obtained for the 5-, 10-, and 15-mg samples are almost identical since their similarity is higher than 99%. The curves for the 1-mg and 20-mg samples with similarity values higher than 92% would most probably be recognized correctly as well.
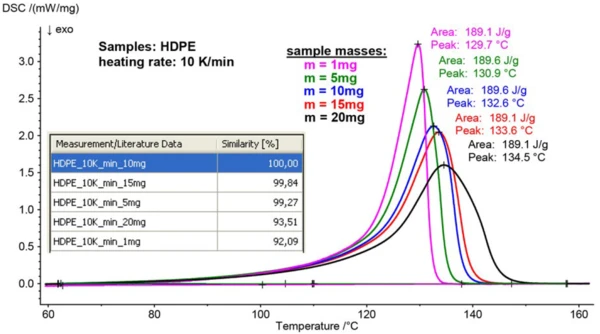
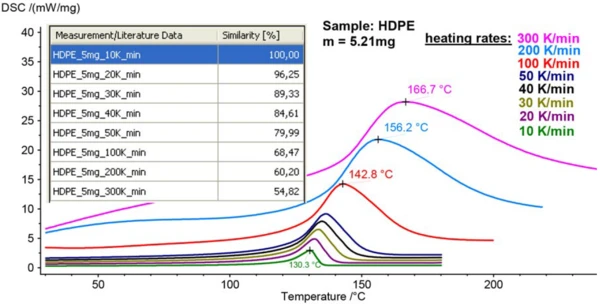
Figure 3 illustrates the impact of different heating rates on the melting peak of the same HDPE sample with a mass of 5.21 mg. With heating rates increasing from 10 K/min to 300 K/min, the peak temperature shifted from 130.3°C to 166.7°C, and the peaks broadened again significantly.
The table in figure 3 shows the similarity values from Identify between the curve obtained at 10 K/min (reference) and all other data sets, respectively. The similarity between the curves measured at 10 K/min and 20 K/min was as high as 96.3%. Similarity values decreased by about 10% for every doubling of the heating rate.
Concluding Remarks
- It was demonstrated that Identify can reliably match DSC curves for the same materials, yielding high similarity values even under widely varying sample measurement conditions. When using “standard” search parameters, a difference of a factor of 2 in the sample mass or heating rate still results in very high similarity values, and therefore, probably correct identification of the sample.
- For quality control purposes, in which greater distinction between samples is desired, “demanding” instead of “standard” search parameters of Identify can be selected to discern slight differences in the DSC curves, which will lead to large variations in similarity values.
- DSC curves measured under different measurement conditions could all be added to the user libraries of Identify and will consequently be recognized in the future. Identify also allows DSC curves measured with different sample masses or heating rates to be grouped together into a class (e.g. the material class ‘HDPE’). This feature also enhances the potential for correct sample identification independent of measurement conditions.