Introduzione
I materiali plastici in generale sono eccellenti isolanti. Grazie all'elevata resistenza meccanica e al peso ridotto, sono particolarmente adatte al mercato dell'elettricità e dell'elettronica (E&E), nonché all'industria dei trasporti e degli elettrodomestici. Un materiale plastico comunemente utilizzato per queste applicazioni appartiene alla famiglia delle poliammidi: Il PA6, che presenta una buona qualità superficiale, una buona lavorabilità e prezzi leggermente inferiori rispetto ad altri PA, è particolarmente adatto. In molte di queste applicazioni, il materiale plastico viene rinforzato con fibre di vetro corte per migliorare ulteriormente le prestazioni meccaniche.
Tuttavia, questi materiali possono prendere fuoco quando sono sufficientemente vicini a una fonte di accensione come una scintilla elettrica. Una misura comune per garantire la sicurezza antincendio è l'aggiunta di ritardanti di fiamma (FR). Il tipo e la quantità di ritardante di fiamma utilizzato dipende dall'applicazione e dai requisiti associati stabiliti dai vari standard di infiammabilità.
In generale, si desidera una bassa quantità di ritardante di fiamma, per avere il minimo effetto sulle proprietà e sul comportamento di lavorazione della plastica. Come qualsiasi altro additivo, i ritardanti di fiamma aumentano la viscosità dei polimeri fusi, un aspetto particolarmente critico nell'industria elettronica, dove la miniaturizzazione e quindi le pareti molto sottili sono standard. Per il PA6 esiste una varietà di ritardanti di fiamma.
Gli incendi innescati anche da una sola scintilla elettrica sviluppano fumo fin dall'inizio, motivo per cui la maggior parte delle vittime di incendi viene uccisa dal fumo tossico. Inoltre, il fumo può diventare abbastanza denso da rendere difficile l'orientamento visivo o addirittura ostacolare la fuga di una persona intrappolata. Le sostanze corrosive presenti nel fumo possono anche danneggiare le attrezzature altrimenti non interessate dall'incendio. La tossicità e la corrosività spesso osservate provengono da polimeri alogenati o ritardanti di fiamma. Per questo motivo, per evitare questi problemi si utilizzano ritardanti di fiamma speciali non alogenati e ritardanti di fiamma a base di grafite.

Condizioni di misura
Per evidenziare l'effetto di diversi ritardanti di fiamma non alogenati sul comportamento al fuoco del PA6, campioni dei diversi composti sono stati stampati a iniezione in piastre di 100 x 100 x 4 mm3 e testati nel TCC 918 (figura 1). Lo strumento consente di determinare il rilascio di calore, la perdita di massa, la densità e la composizione dei fumi. I campioni sono stati posizionati su un portacampioni orizzontale che è stato inserito nella cella di carico. La cella di carico controlla la massa del campione durante la misurazione. Un riscaldatore elettrico radiante conico irradia uniformemente il campione dall'alto. Tra la superficie del campione e il riscaldatore conico si trova un accenditore a scintilla. Questo accende i gas infiammabili che si sviluppano dal campione quando viene riscaldato. I gas di combustione prodotti passano attraverso il cono di riscaldamento e vengono raccolti da un sistema di condotti di scarico con un ventilatore centrifugo e una cappa. Nella sezione di misurazione del condotto di scarico è possibile misurare il flusso di massa e la temperatura del gas di fumo, nonché le concentrazioni di O2,CO2 e CO e la trasmissione della luce laser attraverso il gas di fumo.
Prima di iniziare i test, il sistema di analisi dei gas (Siemens Oxymat/Ultramat) è stato calibratificato con gas di calibratazione e il fattore C è stato controllato utilizzando il bruciatore di metano con un rilascio di calore definito. L'analizzatore di gas utilizzato era dotato di opzione O2 eCO2. Dopo aver riscaldato il riscaldatore a cono, l'otturatore è stato chiuso e il portacampioni preparato è stato posizionato sulla piastra di massa. Quindi, il sistema ha rimosso automaticamente l'otturatore per l'inizio della misurazione. I gas evaporati sono stati accesi dal sistema di accensione automatica. Le condizioni di misura sono riassunte nella Tabella 1.
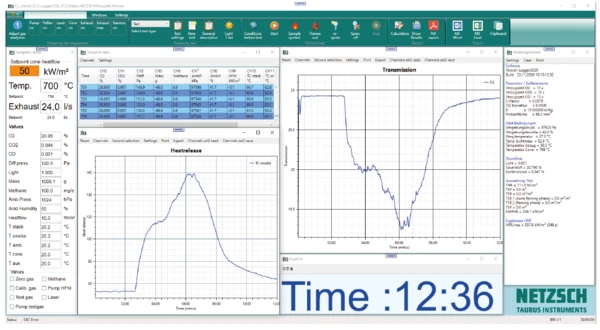
La Figura 2 mostra i risultati della misura su PA 6 pulito e la visualizzazione nel software TCC. La colonna di sinistra mostra i dati di ingresso della misura; al centro, una tabella con i valori misurati da 751 a 756 s e due grafici di esempio dei dati misurati; la colonna di destra presenta una panoramica dei valori di analisi di selectper questa particolare misura.
Tabella 1: Condizioni di misura
| Supporto del campione | Orizzontale | |
| Flusso di calore | 50 kW/m² | |
| Portata nominale del condotto | 24.0 l/s | |
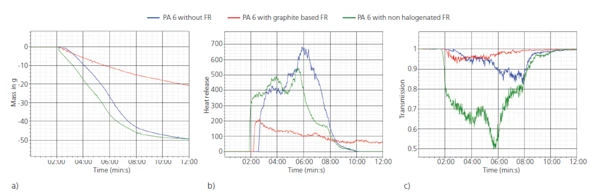
La Figura 3 consente di osservare più da vicino i risultati. La Figura 3a mostra la perdita di massa, b) il tasso di rilascio di calore e c) la trasmissione in funzione del tempo per i tre diversi campioni. Si può notare che il campione di PA6 con 20 wt% di ritardante di fiamma a base di grafite (curva rossa) mostra la perdita di massa, il rilascio di calore e il rilascio di fumi (riduzione minima della trasmissione) più bassi di tutti i campioni. In confronto, il campione con il 20% di ritardante di fiamma non alogenato (curva verde) si comporta in modo molto simile al materiale PA6 puro (curva blu). Per quanto riguarda il rilascio di calore, mostra valori leggermente inferiori e inoltre il rilascio di calore termina prima. Per quanto riguarda la trasmissione, invece, l'emissione di fumo è molto più elevata rispetto al PA6 puro.
Sintesi
Queste indagini dimostrano che nel caso di questa particolare PA6 e dei carichi di FR studiati, il ritardante di fiamma a base di grafite si comporta molto meglio e riduce significativamente gli effetti dannosi che un incendio può avere sull'ambiente circostante. Nel caso del FR non alogenato, sarebbe necessario studiare ulteriori cariche per identificare una composizione con prestazioni migliori.