Tips & Tricks
How to Avoid Incorrect Results Due to Improper Sample Preparation
A thermogravimetric balance measures the mass changes in a sample during a temperature/time program (DIN 51005). As a result, it is possible to determine the temperatures of chemical and physical processes that induce a mass change.
The processes include vaporization, sublimation, desolvation, and thermal and oxidative decomposition, among others.
It is well known that the thermogravimetric curve is influenced by the following factors:
- Heating rate
- Sample geometry
- Sample mass
If, for example, the heating rate and the sample mass are increased, the detected TGA effects are also shifted to higher temperatures. However, it is also possible to play with the factors heating rate and sample mass, in order to obtain as much information as possible from the measurement curve: better separation of overlapped effects by variation of the heating rate and/or sample mass, magnification of small-scale effects by increasing them, etc.
Improper sample preparation can be the cause of more than just a shift of the mass losses monitored by the TGA: It can lead to significantly different results. With thermogravimetry, solid specimens can be measured as a powder or a piece of a tablet; liquids can be measured as well. However, one should be aware that reproducible TGA curves can only be obtained by consistently using the same sample preparation (sample form) and measurement conditions. In particular, the sample surface influences certain processes, which can be seen in the evaporation of solvents or in oxidative decomposition (combustion). As a consequence, these effects are associated with different temperatures depending on weather the investigated sample is a powder or composed of a single piece. In the following, thermogravimetric measurements are used to carry out kinetic analysis of the reaction. This example demonstrates how crucial sample preparation can be to draw the correct deductions.
TGA-FT-IR Measurements on Ibuprofen Tablet
The measurements were carried out on an ibuprofen tablet, Ibu 400 akut, marketed by 1A Pharma®. This tablet contains ibuprofen as an API (Active Pharmaceutical Ingredient); this is one of the most frequently used non-steroidal anti-inflammatory drugs (NSAIDs). Furthermore, it also includes excipients that act as fillers, lubricants, disintegrants, etc.
Measurements were performed at different heating rates between 5 and 20 K/min with the TG 209 F1 Nevio thermobalance in a dynamic nitrogen atmosphere. Aluminum oxide crucibles were used. The sample masses ranged from 9.93 mg to 10.09 mg. The gases evolved during heating were directly transferred into the gas cell of the FT-IR spectrometer by Bruker Optics.
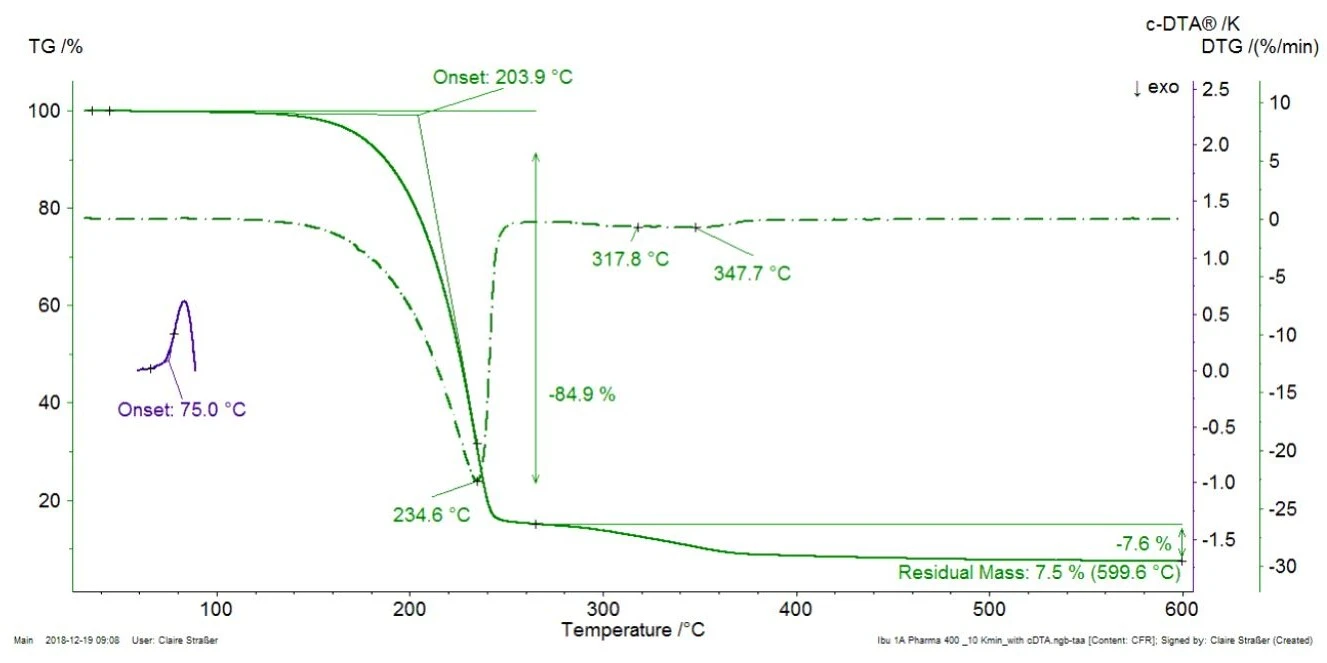
Figure 1 displays the TGA measurement on the commercial ibuprofen tablet at a heating rate of 10 K/min along with the DTG curve (first derivative of the TGA curve). Additionally, the calculated DTA signal (c-DTA®®, the difference between furnace and sample temperature) is displayed between 70°C and 100°C (blue curve).
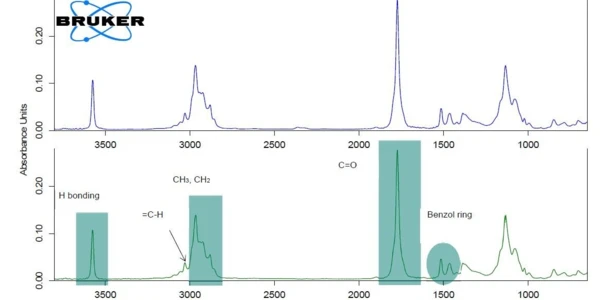
For better readability of the plot, the c-DTA®® signal was only displayed in the temperature range of the ibuprofen melting. The peak detected at 75°C (extrapolated onset temperature) is not associated with a mass loss; it does not result from decomposition or evaporation, which would induce mass changes in the TGA curve, but from the melting of ibuprofen. The first mass loss with an extrapolated onset temperature of 204°C amounts to 85%. It indicates the decomposition or the evaporation of a component present in the tablet, most probably, the evaporation of the active ingredient, ibuprofen [1]. For verification purposes, pure ibuprofen was also measured by means of TGA-FT-IR (figure 2). The spectra of the gases released at 232°C are very similar for the two materials.
This proves that the mass loss detected at 235°C (DTG peak, figure 1) in Ibu 400 akut actually results from the evaporation of the active ingredient (ibuprofen) and is not due to the decomposition of an excipient. In figure 1, Ibu 400 akut exhibits two further mass-loss steps between 250°C and 400°C which are partially overlapped. They are probably due to the thermal decomposition of excipients present in the tablet as microcrystalline cellulose or magnesium stearate [2].
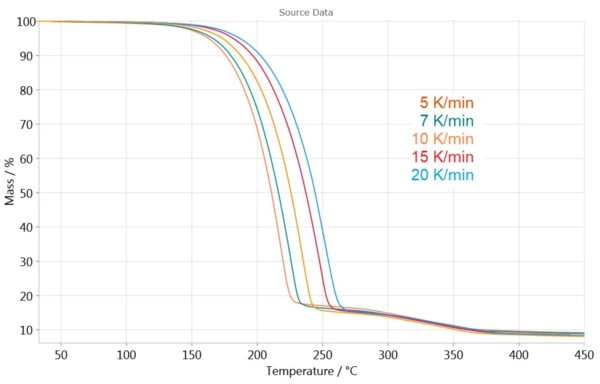
Figure 3 shows the TGA measurements at different heating rates. The effects are shifted to higher temperatures as heating rates increase. This dependence of the TGA curve on the heating rate allows for determination of the reaction kinetics.
Determination of the Reaction Kinetics by Means of Kinetics Neo
The TGA curves obtained are the basis for the kinetic evaluation of the reactions occurring in the measured temperature range. For this, the NETZSCH Kinetics Neo software was used. It allows for modelling the kinetics of single- to multi-step reactions.
This software can assign each individual step to different reaction types with kinetic parameters of their own, such as activation energy, order of reaction, and pre-exponential factor. Based on the results, Kinetics Neo is able to simulate the reaction(s) for user-specified temperature programs.
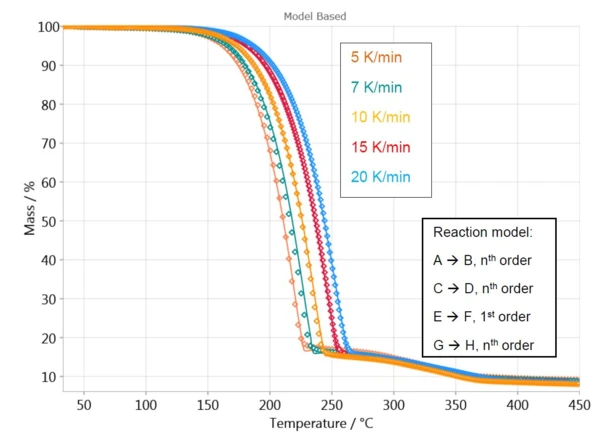
To implement, the thermogravimetric curves are first imported into the Kinetics Neo software. Then, a reaction model is selected for each step (for example: nth order reaction). Based on the reaction model chosen, the software calculates thermogravimetric curves. The pertinence of the model is assessed via the correlation coefficient between the measured and calculated curves.
For the first mass loss, the software calculates a curve with a one-step reaction model, A → B. The sample behavior in the temperature range from 250°C to 450°C is described with three independent steps (C→D, E→F and G→H) because this is the best fit for the measured data in this temperature range.
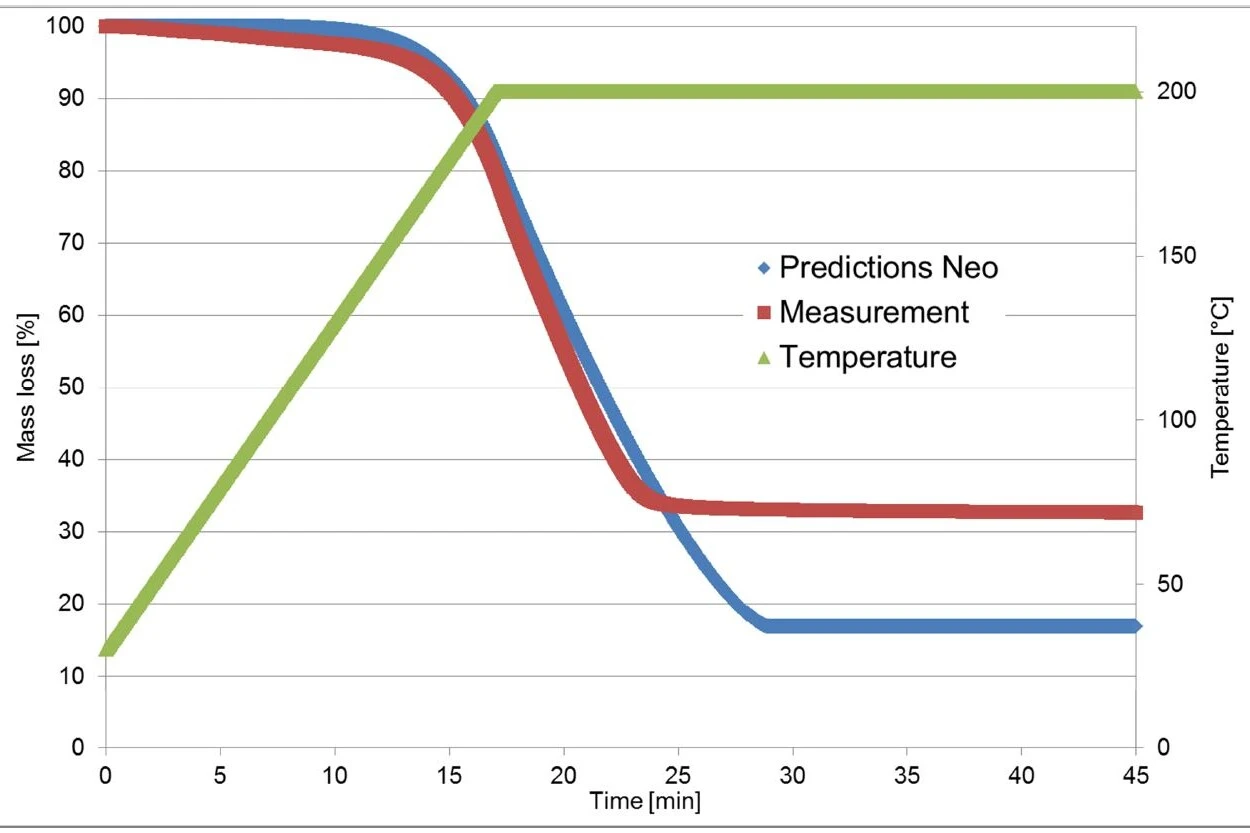
Figure 4 displays a comparison of the measured and calculated curves for such a model. With a correlation coefficient of higher than 0.999, the kinetic model describes the reaction process very well.
For each reaction step, Kinetics Neo calculates the kinetic parameters: activation energy, reaction order, contribution of the step to the global process, etc. Table 1 presents them for all four steps.
Tab. 1. Kinetic parameters for the four steps
| Reaction | A → B | C → D | E → F | G → H |
| Reaction type | nth order | nth order | 1st order | nth order |
| Activation energy [kJ/mol] | 77.823 | 181.866 | 148.941 | 460.643 |
| Log(PreExp) [Log(1/s)] | 6.814 | 14.911 | 10.511 | 38.543 |
| Reaction Order | 0.286 | 1.332 | 1 | 13.410 |
| Contribution | 0.912 | 0.022 | 0.034 | 0.033 |
What Is the Reason for the Difference Observed?
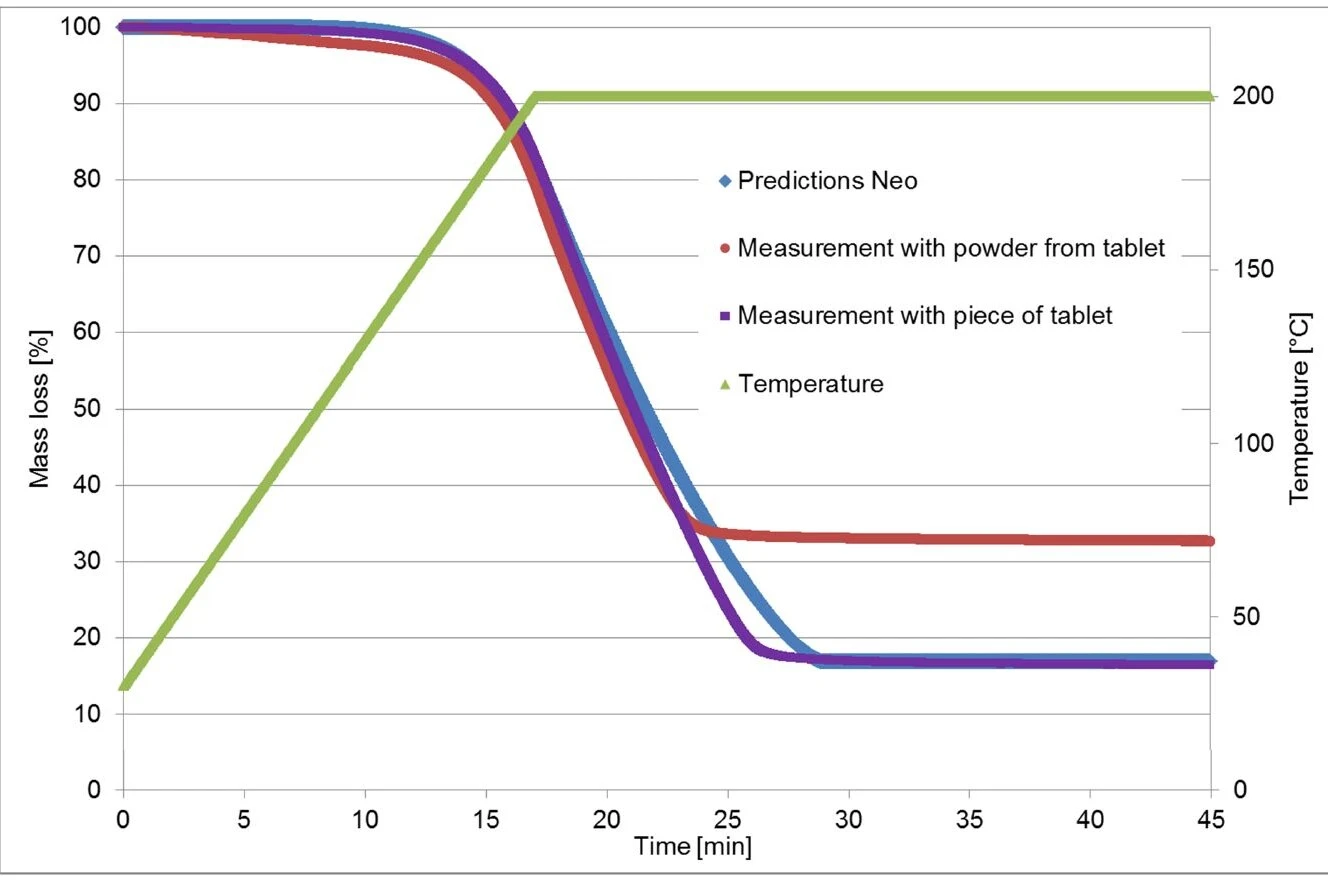
The thermogravimetric measurements used for calculation in Kinetics Neo were carried out on a piece of an Ibu 400 akut tablet. In contrast with that, the validation measurement was carried out on a powder produced by crushing the tablet.
As mentioned before, the first mass-loss step is due to the evaporation of ibuprofen, which is dependent on the sample surface [1]. One can expect the larger sample surface associated with the crushed material to have a heavy influence on the TGA curve.
In a second experiment, the previous measurement (heating to 200°C and isothermal) was carried out again, but this time using a piece of the tablet. The new thermogravimetric curve now corresponds very well to the one calculated by Kinetics Neo! (See figure 6.)
Conclusion
TGA measurements were carried out on an ibuprofen tablet (commercial name: Ibu 400): one on a solid piece and one on a powder. FT-IR measurements were able to demonstrate that the first mass-loss step results from the evaporation of the active ingredient. In addition, this process is highly dependent on the sample surface, so the results of measurements carried out on a piece of the tablet differ from those carried out on the powder. This also has a great impact on the kinetic analysis. Such a kinetic analysis is particularly useful when investigating the thermal stability of a pharmaceutical.
References
[1] A thermal analysis study of ibuprofen, S. Lerd-kanchanaporn and D. Dollimore, Journal of Thermal Analysis, Vol. 49 (1997), Issue 2, pp 879-886
[2] NETZSCH Application Note 120: Compatibility Studies on Diclofenac Sodium – Fast and Easy with Thermal Analysis; Figures 5 and 9