Metals & Alloys
Aluminum Alloy - Melting & Solidification
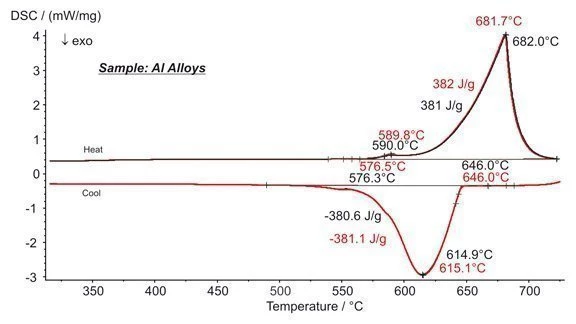
Two different samples of aluminium alloy (Al-Mg-Si) were measured with the DSC 404 F3 Pegasus® during heating and cooling.
Melting Temperatures and EnthalpiesThe enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state. The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt).Melting and also solidification of the alloy is clearly visible in the measurement results. The differences between the two different samples, however, are small. The characteristic temperatures (onset, peak) agree all within 0.3 K. The differences in the peak areas are less than 1% for both heating and cooling. The good agreement between the two measurement results demonstrates the excellent capability and reproducibility of the DSC 404 F3 Pegasus®.