Introduction
Les pièces en matériaux polymères sont largement utilisées dans tous les domaines où la réduction du poids et la production rentable jouent un rôle décisif. Bien que les pièces moulées par injection en matériaux thermoplastiques soient utilisées dans l'industrie automobile depuis des décennies, la demande de solutions légères pour les automobiles modernes ne cesse d'augmenter. En particulier pour le développement des véhicules électriques et la réduction des émissions deCO2, de plus en plus de composants automobiles fabriqués à partir de matériaux légers sont utilisés.
L'utilisation accrue des matières plastiques nécessite un moyen de garantir une qualité et une stabilité constantes des pièces. L'analyse des matériaux joue ici un rôle majeur. Les propriétés mécaniques des pièces sont fortement influencées par de nombreuses étapes du processus. Par exemple, le simple fait de peindre une matière plastique peut modifier ses propriétés physiques au point que, dans le pire des cas, elle se brisera lorsqu'elle sera exposée à une charge raisonnable. Il est donc important de garantir la qualité constante des matériaux tout au long du processus de fabrication, du début à la fin. Les méthodes d'analyse thermique, telles que la calorimétrie à balayage différentiel (DSC), sont des outils idéaux pour de telles questions. Dans le cas étudié ici, un composant de boîtier en polyamide 6 renforcé de fibres de verre présentait une fragilisation au niveau du crochet de l'agrafe lors de la connexion avec les articulations de l'agrafe. Lors de l'installation de la pièce, le clip s'est rompu. Pour de telles défaillances, il est crucial d'examiner tous les facteurs d'influence potentiels tout au long de la chaîne de fabrication.
Résultats des tests
L'analyse DSC de la pièce endommagée et d'une pièce de contrôle iO a permis d'identifier rapidement la cause de la défaillance. Les courbes DSC sont présentées dans la figure 1. Pour l'analyse de la composition du matériau, lesdeuxièmes courbes de chauffage sont toujours évaluées car les effets de l'historique thermique ne sont plus présents. Parallèlement à la transition vitreuse de l'échantillon à 50,9°C, la pièce de contrôle (courbe verte) présente une endothermie de Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion à 221°C avec une enthalpie de Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion de 53,7 J/g (typique du PA 6 pur). La partie en niO (endommagée), cependant, a montré un comportement sensiblement différent, avec une température de pointe à 215°C et une enthalpie de 45,2 J/g.
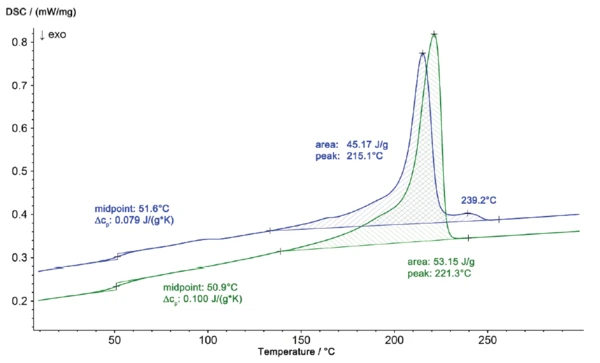
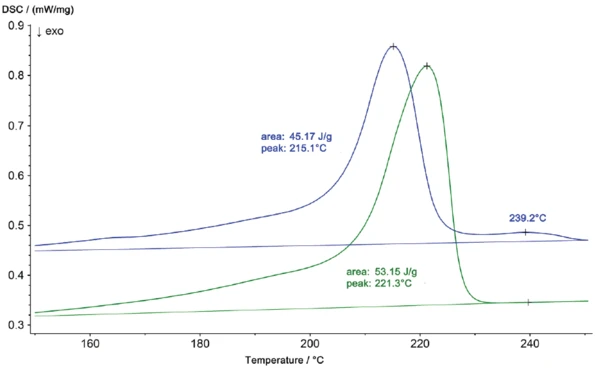
Le profil de Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion de la pièce en niO, représenté sur une échelle agrandie dans la figure 2, montre également un deuxième pic à 239°C. Les résultats des mesures DSC révèlent que le matériau de la pièce endommagée n'est plus du polyamide 6 pur mais un mélange de polyamide 6 et de polyamide 66. Ces deux composants peuvent former un eutectique, ce qui explique le passage de la Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). température de fusion de 221°C (PA 6 pur) à 215°C (PA 6 + PA 66). Les différences entre les deux échantillons se manifestent également par des profils de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation différents lors du refroidissement (figure 3)
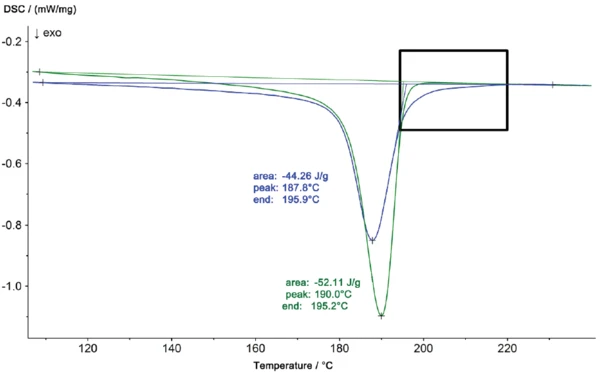
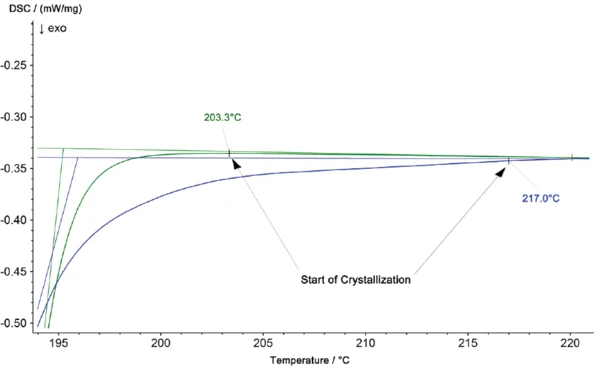
Dans l'analyse DSC, la CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation est observée comme un effet ExothermiqueUne transition d'échantillon ou une réaction est exothermique si elle produit de la chaleur.exothermique. L'agrandissement de la figure 4 montre en outre une température plus élevée pour la CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation du matériau provenant de la partie iO à 217°C, par rapport à 203°C pour l'échantillon de PA6 pur. La surface du pic est également plus petite pour la partie iO.
Conclusion
Cet exemple démontre clairement que la composition du matériau a une influence mesurable sur les propriétés d'une pièce finie et que les défaillances peuvent être évitées en utilisant l'analyse thermique pour contrôler la qualité de la matière première. Le contrôle de la qualité peut être réalisé avec relativement peu d'efforts grâce à l'analyse thermique au moyen de la DSC.