Introduzione
Il lattosio è uno zucchero disaccaride composto da galattosio e glucosio che si trova nel latte dei mammiferi. Il lattosio costituisce circa il 2%-8% del latte (in peso), anche se la quantità varia a seconda delle specie e degli individui. Il nome deriva da lac (gen. lactis), la parola latina che indica il latte, più la desinenza -ose utilizzata per denominare gli zuccheri [3].
Il lattosio è spesso utilizzato nella tecnologia alimentare o come eccipiente nei prodotti farmaceutici. La conoscenza delle proprietà termiche del lattosio è essenziale perché la sua transizione vetrosa è in relazione diretta con le proprietà fisiche - come l'appiccicosità e la fluidità - delle polveri contenenti zucchero del latte, e questo a sua volta influenza la lavorazione. [4]
Di seguito, l'influenza della velocità di riscaldamento sulle proprietà termiche dell'α-lattosio monoidrato FlowLac® 90 fornito da MEGGLE è stata analizzata mediante DSC. Come prodotto essiccato a spruzzo, presenta tipicamente un contenuto amorfo compreso tra il 10% e il 15%. [5]
Condizioni di prova
Polyma Le misure sono state eseguite con il DSC 214 NETZSCH in atmosfera dinamica di azoto. I campioni di massa compresa tra 4,21 mg e 4,74 mg sono stati pesati in crogioli di alluminio Concavus® sigillati con un coperchio forato e riscaldati a 280°C a diverse velocità di riscaldamento (20, 50, 100 e 200 K/min).
Risultati del test
Le figure 2 e 3 illustrano le curve di misurazione DSC per le diverse velocità di riscaldamento.
Una variazione della capacità termica con un punto medio tra 62°C (misurazione a 20 K/min) e 85°C (misurazione a 200 K/min) indica la transizione vetrosa del campione. Il picco EndotermicoUna transizione campionaria o una reazione è endotermica se per la conversione è necessario il calore.endotermico rilevato tra 148°C e 185°C (temperatura di picco) deriva dal rilascio dell'acqua. Ciò è in accordo con i risultati pubblicati in [2], secondo cui il lattosio monoidrato rilascia l'acqua idrata quando viene riscaldato a temperature superiori a 150°C.
Il secondo picco, situato tra 222°C e 248°C, è dovuto alla Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione dei cristalli di α-lattosio anidrato. Sebbene l'andamento delle curve sia molto simile, l'influenza della velocità di riscaldamento si nota su tutti gli effetti (transizione vetrosa, disidratazione e Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione). In primo luogo, si spostano verso temperature più elevate con l'aumento della velocità di riscaldamento. In secondo luogo, l'aumento della velocità di riscaldamento porta a un'amplificazione degli effetti DSC. Ciò è dovuto all'influenza della velocità di riscaldamento sulla cinetica dei processi.
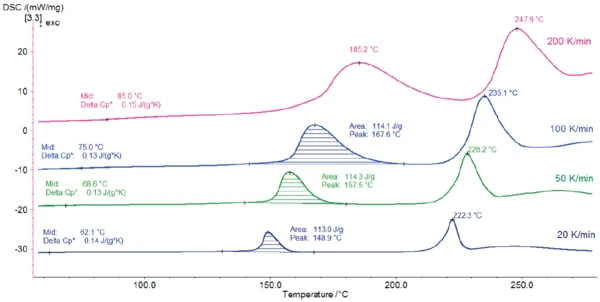
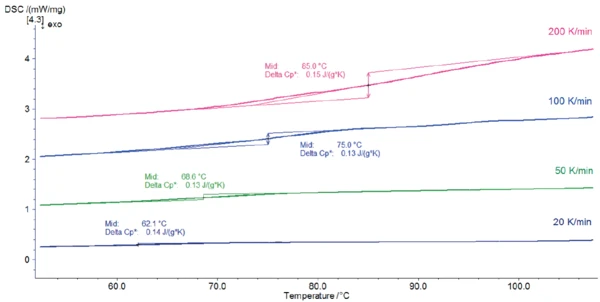
L'aumento della velocità di riscaldamento è utile per migliorare il rilevamento degli effetti small. In questo esempio, ad esempio, la transizione vetrosa del lattosio è più facile da rilevare nelle misurazioni effettuate a velocità di riscaldamento più elevate. D'altra parte, diminuire la velocità di riscaldamento aiuta a separare gli effetti di sovrapposizione. Nella misurazione a 200 K/min, il picco del rilascio di acqua si sovrappone parzialmente al picco di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione a 248°C, rendendo difficile la valutazione dell'entalpia di picco. Al contrario, l'energia di disidratazione può essere determinata con precisione per velocità di riscaldamento inferiori.
Conclusione
Gli effetti termici dell'α-lattosio monoidrato possono essere facilmente determinati mediante calorimetria differenziale a scansione (DSC). La temperatura di transizione vetrosa e i picchi di disidratazione e Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione dipendono dalla velocità di riscaldamento.
Per una migliore valutazione, l'aumento della velocità di riscaldamento può essere uno strumento utile quando gli effetti small nella curva DSC devono essere amplificati, mentre la diminuzione della velocità di riscaldamento può essere utile quando gli effetti sovrapposti devono essere separati.