Introdução
A Calorimetria de Taxa Acelerada (Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC®) é um método para estudar os Pior cenário possívelEm relação a um reator químico, o pior cenário possível é a situação em que a produção de temperatura e/ou pressão causada pela reação fica fora de controle.piores cenários e reações térmicas de fuga. Em contraste com outras técnicas calóricas, como a calorimetria de reação, a calorimetria de combustão ou a calorimetria de varredura diferencial (DSC), o equipamento do tipo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® permite um ambiente de amostra AdiabáticoAdiabático descreve um sistema ou modo de medição sem nenhuma troca de calor com o ambiente. Esse modo pode ser realizado usando um dispositivo de calorímetro de acordo com o método de calorimetria de taxa acelerada (ARC®). O principal objetivo desse dispositivo é estudar cenários e reações de fuga térmica. Uma breve descrição do modo adiabático é "sem entrada de calor - sem saída de calor".adiabático. A adiabaticidade é essencial para observar o progresso mais violento possível da reação. As Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reações de decomposição, que são de interesse especial nesse contexto, produzem calor e pressão, pois as reações geralmente são altamente exotérmicas e formam gases de Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. decomposição. O ambiente AdiabáticoAdiabático descreve um sistema ou modo de medição sem nenhuma troca de calor com o ambiente. Esse modo pode ser realizado usando um dispositivo de calorímetro de acordo com o método de calorimetria de taxa acelerada (ARC®). O principal objetivo desse dispositivo é estudar cenários e reações de fuga térmica. Uma breve descrição do modo adiabático é "sem entrada de calor - sem saída de calor".adiabático da amostra é realizado dentro do calorímetro do tipo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® por meio de um conjunto de aquecedores ao redor do compartimento da amostra e um regime inteligente de controle de temperatura. Um dos objetivos é detectar a temperatura na qual a autodecomposição de uma amostra ou de uma mistura de amostras começa. Outro objetivo é evitar qualquer troca de calor entre a amostra e seu entorno após o início da Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reação de decomposição exotérmica. Assim que a Taxa de autoaquecimentoUm tipo especial de dispositivo calorímetro é usado para detectar a taxa de autoaquecimento de uma substância. O método relacionado é chamado de calorimetria de taxa acelerada (ARC®). taxa de autoaquecimento exceder um determinado limite (que geralmente está na faixa de 0,02 K/min), todos os aquecedores ao redor da amostra acompanharão a temperatura da amostra. Sem troca de calor, não haverá perda de calor para o ambiente e, se não houver dissipação de calor, todo o calor da reação permanecerá dentro da amostra, aumentando assim a temperatura da amostra. Quanto mais alta for a temperatura da amostra, mais rápida será a taxa de reação. Esse experimento não apenas fornece a temperatura inicial da Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reação de decomposição em condições quase isotérmicas, mas também permite a determinação do aumento máximo de temperatura e do aumento máximo de pressão em condições adiabáticas.
O fator PHI (φ) ou "inércia térmica"
A partir dos dois sinais medidos, temperatura e pressão, a taxa máxima pode ser calculada, e geralmente são feitas previsões para a temperatura na qual a reação investigada leva no mínimo vinte e quatro horas para atingir sua taxa máxima de desenvolvimento de temperatura, a taxa de tempo até o máximo (TMR24h).
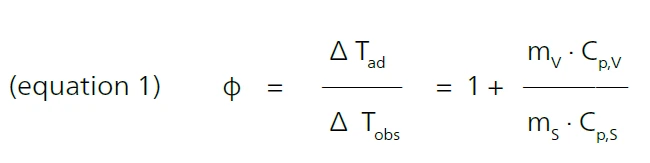
Um parâmetro essencial para o cenário de teste é o que é conhecido como Fator PHIO fator PHI (Φ) é equivalente à inércia térmica. Ambos descrevem a relação entre a massa e a capacidade de calor específica de uma amostra ou mistura de amostras em comparação com a do recipiente ou recipiente de amostras. fator PHI (φ). Ele fornece a razão entre a massa e o calor específico da amostra e o recipiente da amostra, em que ΔTad é o aumento de temperatura sob condições adiabáticas, ΔTobs é o aumento de temperatura observado sob determinadas condições, m é a massa, Capacidade térmica específica (cp)A capacidade térmica é uma quantidade física específica do material, determinada pela quantidade de calor fornecida à amostra, dividida pelo aumento de temperatura resultante. A capacidade térmica específica está relacionada a uma unidade de massa do corpo de prova.cp é a Capacidade térmica específica (cp)A capacidade térmica é uma quantidade física específica do material, determinada pela quantidade de calor fornecida à amostra, dividida pelo aumento de temperatura resultante. A capacidade térmica específica está relacionada a uma unidade de massa do corpo de prova.capacidade de calor específico, s é a amostra e v é o recipiente [1].
O fator φ, também conhecido como Inércia térmicaA inércia térmica é equivalente ao fator PHI. Ambos descrevem a relação entre a massa e a capacidade de calor específica de uma amostra ou mistura de amostras em comparação com a do recipiente ou contêiner de amostras.inércia térmica, é melhor quanto mais próximo de 1, o que significa, no caso ideal, que os resultados do teste são definidos pela amostra e não pela influência do recipiente. Por outro lado, a equação mencionada acima indica que a relação entre a massa da amostra e a massa do recipiente é, de alguma forma, produzida pela reatividade da própria amostra, juntamente com o volume máximo do recipiente da amostra e os materiais disponíveis para os recipientes. Para mostrar como esses parâmetros influenciam o fator φ, a tabela 1 resume os fatores φ calculados para duas amostras (peróxidos orgânicos e peróxido de hidrogênio), dois materiais de recipiente (aço inoxidável e titanium) e para uma variedade realista de massas de amostra.
Tabela 1: Fatores Ф calculados para várias condições de medição
| Massa de peróxido de hidrogênio / g | 0.25 | 0.50 | 1.0 | 2.0 | 5.0 | 8.0 |
| Ф para 10,0 g titanium vessel | 7.41 | 4.20 | 2.60 | 1.80 | 1.32 | 1.20 |
| Massa de peróxido orgânico / g | 0.25 | 0.50 | 1.0 | 1.5 | 5.0 | 8.0 |
| Ф para 7,0 g de aço inoxidável | 9.86 | 5.43 | 3.21 | 1.5 | - | - |
| Massa de peróxido de hidrogênio / g | 0.25 | 0.50 | 1.0 | 2.0 | 5.0 | 8.0 |
| Ф para 7,0 g de aço inoxidável | 5.92 | 3.46 | 2.23 | 1.82 | - | - |
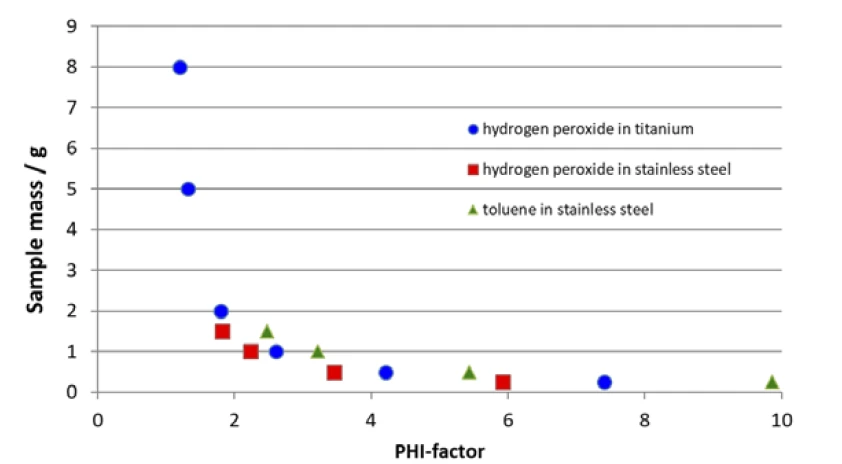
A correlação entre a massa da amostra e o fator φ calculado mencionada acima é mostrada adicionalmente na Figura 1. Como a capacidade de calor específica da amostra a ser investigada, juntamente com a capacidade de calor específica do material do recipiente, geralmente é fornecida, o único parâmetro disponível para alterar o fator φ é a massa da amostra.
O aumento da massa da amostra pode aproximar o fator φ de 1, mas pode haver limitações no volume do recipiente, bem como limitações associadas ao próprio equipamento. É imprescindível ter em mente a faixa de pressão, a faixa de temperatura e a taxa máxima de rastreamento do calorímetro do tipo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® usado para não exceder nenhuma delas; caso contrário, os dados podem deixar de ser significativos. Pode-se ver na figura 1 que, devido ao seu volume total de 2,6 ml, o recipiente de aço inoxidável (figura 3) está limitado a uma massa de amostra inferior a 2,0 g. Como os recipientes geralmente não são preenchidos mais do que a metade, o fator φ esperado está entre 2 e 4, dependendo da capacidade de calor específica da própria amostra. Somente com 1,5 mg de peróxido de hidrogênio, que tem uma capacidade de calor específica relativamente alta, é possível estabelecer um fator φ melhor do que 2. Mesmo com o uso de um recipiente de titanium com 8,6 ml de volume, é difícil obter massas de amostra superiores a 3,0 g e fatores φ na faixa de 1,5.
Todas as amostras que apresentam potencial de perigo térmico também são caracterizadas por um risco maior em relação ao manuseio em um ambiente de laboratório. Do ponto de vista da segurança, é claro que é muito melhor manusear amostras de risco em small quantidades. Levando em consideração as limitações discutidas acima, surge um dilema. Quanto menor o fator φ, mais significativos devem ser os resultados. Isso, no entanto, exigiria larger quantidades de amostra. No entanto, a diminuição da massa da amostra para resolver os problemas de segurança aumentará o fator φ. Para superar esse dilema, o VariPhi patenteado foi usado dentro de um módulo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® do Calorímetro de múltiplos módulos (MMC)Um dispositivo de calorímetro de modo múltiplo que consiste em uma unidade de base e módulos intercambiáveis. Um módulo é preparado para calorimetria de taxa acelerada (ARC®), o ARC®-Module. Um segundo é usado para testes de varredura (Scanning Module) e um terceiro e quarto estão relacionados a baterias e polímeros, testes farmacêuticos para células tipo moeda (Coin Cell Module).MMC 274 Nexus®.
O Calorímetro de Múltiplos Módulos (MMC 274 Nexus®)
O Calorímetro de múltiplos módulos (MMC)Um dispositivo de calorímetro de modo múltiplo que consiste em uma unidade de base e módulos intercambiáveis. Um módulo é preparado para calorimetria de taxa acelerada (ARC®), o ARC®-Module. Um segundo é usado para testes de varredura (Scanning Module) e um terceiro e quarto estão relacionados a baterias e polímeros, testes farmacêuticos para células tipo moeda (Coin Cell Module).MMC 274 Nexus® Multiple Module Calorimeter (figura 4) oferece três módulos de medição diferentes [2]. O Coin-Cell Module é especializado na investigação de baterias e o Scanning Module [3, 4] pode ser usado para avaliar dados calóricos de uma única execução de aquecimento. O Módulo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® (figura 5) pode ser usado para estudos de risco térmico e foi empregado para os resultados apresentados neste trabalho.
Substância de teste: Solução de peróxido de hidrogênio
O peróxido de hidrogênio (H2O2) se decompõe termicamente em água e oxigênio. Essa Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reação de decomposição pode ser iniciada termicamente e é altamente exotérmica. É por isso que o peróxido de hidrogênio geralmente é manuseado como uma solução aquosa de até 35%. Em termos de estudos de segurança térmica, é uma substância ideal, pois forma água e oxigênio durante a Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. decomposição, o que torna a limpeza e a reutilização dos recipientes bastante convenientes.
O módulo ARC® com VariPhi
A Figura 5 mostra a configuração do módulo Calorimetria de taxa acelerada (ARC)O método que descreve os procedimentos de teste isotérmico e adiabático usados para detectar reações de decomposição termicamente exotérmicas.ARC® do Calorímetro de múltiplos módulos (MMC)Um dispositivo de calorímetro de modo múltiplo que consiste em uma unidade de base e módulos intercambiáveis. Um módulo é preparado para calorimetria de taxa acelerada (ARC®), o ARC®-Module. Um segundo é usado para testes de varredura (Scanning Module) e um terceiro e quarto estão relacionados a baterias e polímeros, testes farmacêuticos para células tipo moeda (Coin Cell Module).MMC. O recipiente de amostra é colocado dentro do compartimento do calorímetro e a temperatura da amostra é detectada por meio de um termopar fixado diretamente na parede externa do recipiente de amostra. O próprio recipiente é conectado a um medidor de pressão por meio de uma passagem. Bem no centro dessa configuração, o aquecedor interno, chamado VariPhi, é colocado dentro da amostra.
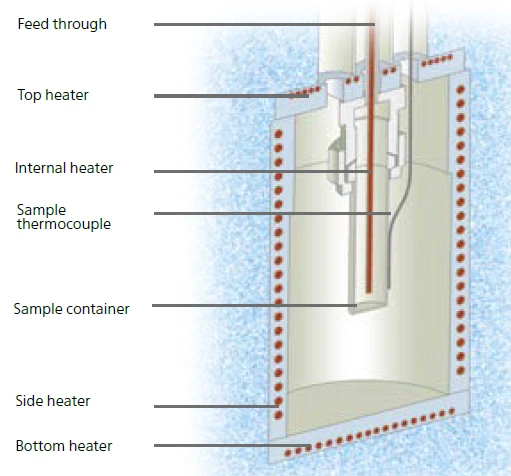
Esse aquecedor patenteado da VariPhi é a solução para o dilema descrito acima. Por um lado, ele pode ser usado para uma execução de triagem a fim de detectar rapidamente se uma amostra desconhecida apresenta ou não potencial perigoso. Nesse caso, seria fornecida energia constante ao aquecedor VariPhi. Juntamente com a taxa de aquecimento resultante, um sinal de fluxo de calor pode ser calculado para distinguir entre os efeitos endotérmicos e exotérmicos da amostra. Por outro lado, o aquecedor VariPhi também pode ser usado para compensar parcial ou totalmente a influência do recipiente da amostra (fator φ; eq. 1). Nesse caso, o aquecedor VariPhi aplica a quantidade de calor à amostra que normalmente seria perdida pelo aquecimento do recipiente da amostra. Como a amostra é a parte mais quente durante uma Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reação de decomposição com autoaquecimento, o calor seria perdido para aquecer o recipiente antes de ser detectado pelo termopar, que é fixado fora do recipiente (figura 5). De acordo com a equação 1, o fator φ pode ser parcial ou totalmente compensado para atingir as condições ideais com relação ao fator φ. Dessa forma, é possível ajustar o fator φ para um valor que reflita as condições reais de um reator ou pode ser ajustado para φ = 1 a fim de estudar os Pior cenário possívelEm relação a um reator químico, o pior cenário possível é a situação em que a produção de temperatura e/ou pressão causada pela reação fica fora de controle.piores cenários. A entrada de energia necessária para a compensação é dada pela massa e pela capacidade de calor específica do vaso.
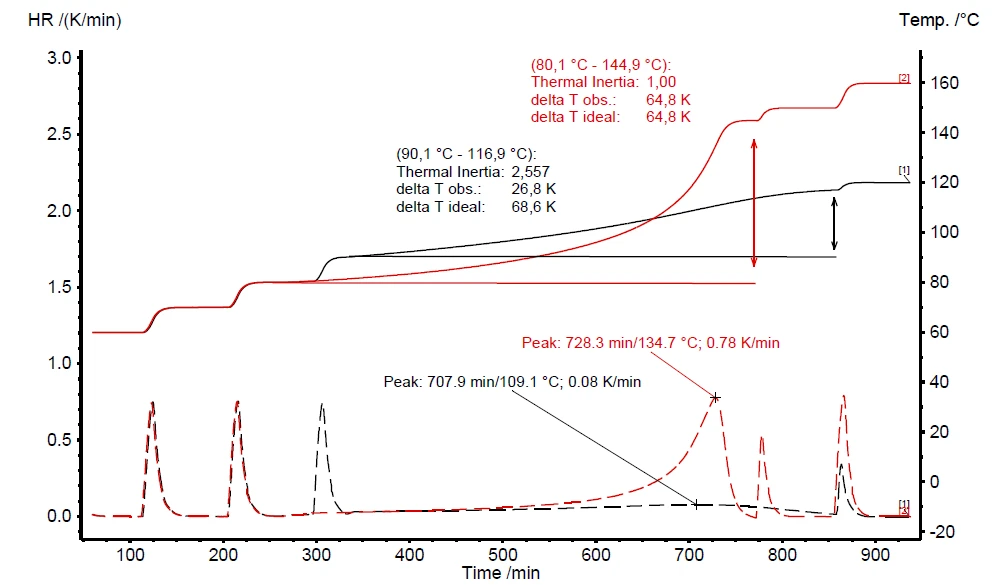
Se um teste de triagem de risco térmico tiver detectado autoaquecimento e aumento de pressão (figura 6), é imperativo realizar um teste adicional de Fuga térmicaO descontrole térmico é a situação em que um reator químico fica fora de controle em relação à produção de temperatura e/ou pressão causada pela própria reação química. A simulação de um descontrole térmico geralmente é realizada usando um dispositivo de calorímetro de acordo com a calorimetria de taxa acelerada (ARC®).fuga térmica. Os resultados desse teste Heat-Wait-Search (HWS)Heat-Wait-Search é um modo de medição usado em dispositivos de calorímetro de acordo com a calorimetria de taxa acelerada (ARC®).heat-wait-search (Heat-Wait-Search (HWS)Heat-Wait-Search é um modo de medição usado em dispositivos de calorímetro de acordo com a calorimetria de taxa acelerada (ARC®).HWS) são mostrados na figura 7. Ela compara as diferenças entre os resultados de medição compensados (curva vermelha) e os não compensados (curva preta). As condições de medição estão resumidas na tabela 2.
Em contraste com o teste de varredura, o teste correspondente do peróxido de hidrogênio em Heat-Wait-Search (HWS)Heat-Wait-Search é um modo de medição usado em dispositivos de calorímetro de acordo com a calorimetria de taxa acelerada (ARC®).heat-wait-search detecta o início do autoaquecimento já a 90°C (figura 7, curva preta). A taxa máxima de autoaquecimento foi detectada como sendo de 0,08 K/min, juntamente com um aumento de temperatura de 26,8 K (ΔTobs). O aumento de temperatura observado é avaliado pela subtração da temperatura inicial (Tstart, início do evento ExotérmicoUma transição de amostra ou uma reação é exotérmica se houver geração de calor.exotérmico) da temperatura final do evento ExotérmicoUma transição de amostra ou uma reação é exotérmica se houver geração de calor.exotérmico (Tfinal) [1].

Os resultados de medição discutidos acima, representados pela curva preta na figura 7, são realizados sem o uso do aquecedor interno, chamado VariPhi; o fator φ associado é 3,14. Ao empregar o VariPhi para a mesma configuração de amostra e usar sua potência para compensar a massa e a capacidade de calor específica do recipiente (φ = 1), o aumento de temperatura medido foi determinado como sendo de 64,8 K (curva vermelha, figura 7). Isso confirma bem a expectativa de um aumento significativo tanto em ΔTobs quanto na velocidade da reação. Quanto menor o fator φ, menos calor é perdido no aquecimento do recipiente da amostra; além disso, todo o calor da reação pode permanecer dentro do recipiente da amostra para acelerar as reações de autoaquecimento. A linha tracejada na figura 7 confirma uma Taxa de autoaquecimentoUm tipo especial de dispositivo calorímetro é usado para detectar a taxa de autoaquecimento de uma substância. O método relacionado é chamado de calorimetria de taxa acelerada (ARC®). taxa de autoaquecimento quase dez vezes maior para a medição usando VariPhi (curva vermelha, figura 7) do que para a medição não compensada. Esses resultados demonstram o enorme impacto do fator φ com relação ao potencial de risco esperado das reações químicas.
Se o site VariPhi não estiver disponível, as medições geralmente não podem ser realizadas em condições de baixo φ devido a limitações causadas pelas propriedades do material do recipiente da amostra, quantidade máxima de amostra, pressão esperada etc. Nesse caso, a ASTM E1981 - 81(2012) sugere a seguinte aproximação para as condições ideais de medição.

O valor "delta T ideal" é calculado de acordo com a equação 3 durante a avaliação de dados no software NETZSCH Proteus® . O resultado não compensado (curva preta na figura 7) indica um " ΔTobs" de 26,8 K e um fator φ de 2,56. A suposição de um resultado de medição em condições ideais (φ = 1) espera que " ΔTideal" seja 68,6 K. Essa suposição feita por meio da equação 3 está próxima do resultado de medição de 64,8 K obtido com o uso do aquecedor VariPhi (curva vermelha na figura 7).
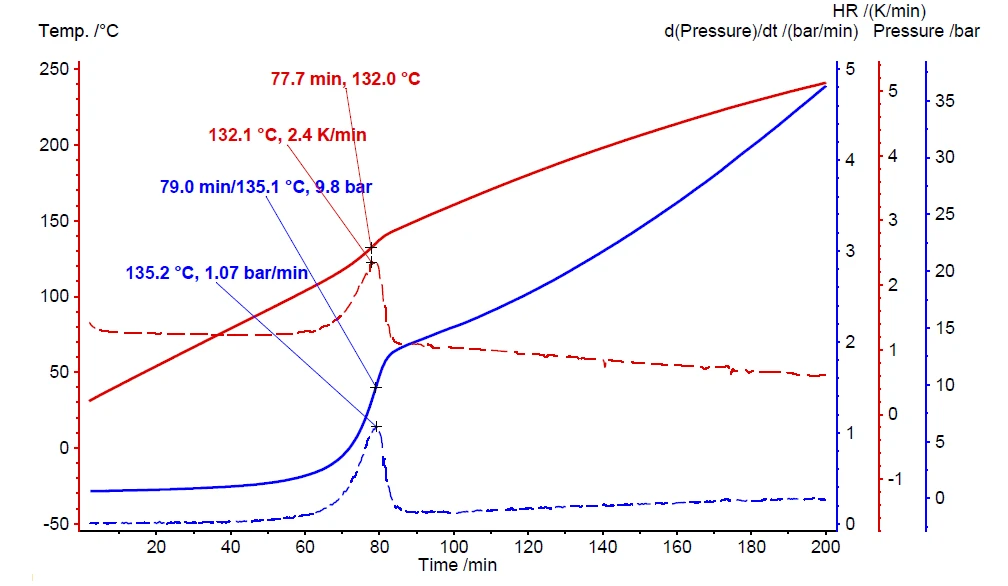
Tabela 2: Condições de medição para a varredura (figura 6) e para os testes de calor e espera (figura 7)
| Módulo Calorímetro de múltiplos módulos (MMC)Um dispositivo de calorímetro de modo múltiplo que consiste em uma unidade de base e módulos intercambiáveis. Um módulo é preparado para calorimetria de taxa acelerada (ARC), o ARC-Module. Um segundo é usado para testes de varredura (Scanning Module) e um terceiro e quarto estão relacionados a baterias e polímeros, testes farmacêuticos para células tipo moeda (Coin Cell Module).MMC | Varredura | ||
ARC® com compensação | |||
| Material do recipiente | Aço inoxidável | Aço inoxidável | Aço inoxidável |
| Tipo de vaso | Fechado | Fechado | Fechado |
| Massa do recipiente | 7176.00 mg | 7119.74 mg | 7119,66 mg |
| Aquecimento | Potência constante (250 mW) | ||
| Atmosfera | Ar | Ar | Ar |
| Taxa de gás de purga | Estático | Estática | Estática |
| Faixa de temperatura | RT ... 250°C | RT ... 250°C | RT ... 250°C |
| Massa da amostra | 512.35 mg | 749.79 mg | 749.46 mg |
| Fator Ф | 4.15 | 3.14 | 3.14 |
| Fator Ф (comp.) | 3.14 | 1.00 | |
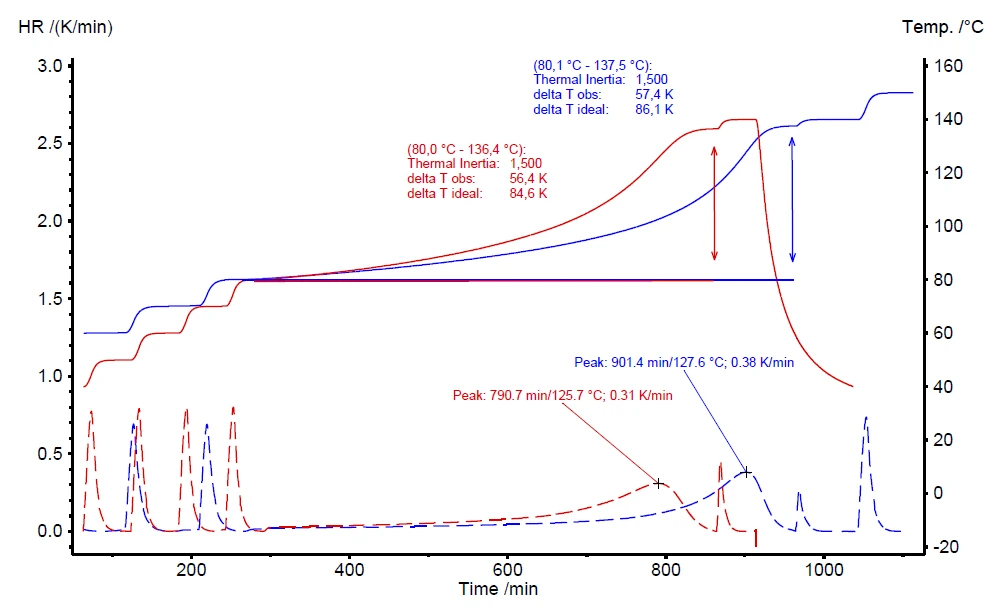
Uma outra vantagem do aquecedor VariPhi é compensar o fator φ para melhorar a comparabilidade de diferentes condições de medição. A Figura 8 compara duas medições em diferentes quantidades de peróxido de hidrogênio. A curva vermelha representa uma medição com 0,500 g de H2O2 (φ = 4,21) e a medição azul foi realizada com 1,00 g (φ = 2,60). Devido às diferentes massas de amostra, os fatores φ são significativamente diferentes: 4,21 e 2,60, respectivamente. O aquecedor VariPhi foi empregado para compensar ambas as medições para φ = 1,5. Os resultados avaliados são muito semelhantes para as duas medições, inclusive a temperatura inicial (Tstart), a Taxa de autoaquecimentoUm tipo especial de dispositivo calorímetro é usado para detectar a taxa de autoaquecimento de uma substância. O método relacionado é chamado de calorimetria de taxa acelerada (ARC®). taxa de autoaquecimento (HR) e o aumento de temperatura observado (ΔTobs).
Conclusão
A Reação de decomposiçãoUma reação de decomposição é uma reação induzida termicamente de um composto químico que forma produtos sólidos e/ou gasosos. reação de decomposição do peróxido de hidrogênio (H2O2) foi investigada como um cenário de teste para demonstrar o uso de um aquecedor adicional dentro do equipamento do tipo ARC®. O aquecedor patenteado VariPhi pode ser empregado para compensar a configuração do teste com um fator φ do mundo real ou com o valor ideal de φ = 1. Essa configuração de compensação da perda de calor permite medições de baixo φ até mesmo em quantidades de amostras de small. Do ponto de vista da segurança, a capacidade de variar o fator φ acaba sendo uma grande vantagem para laboratórios que testam o potencial perigoso de produtos químicos e misturas de reação.


