General Properties
Short Name: PMMA
Name: Polymethylmethacrylate
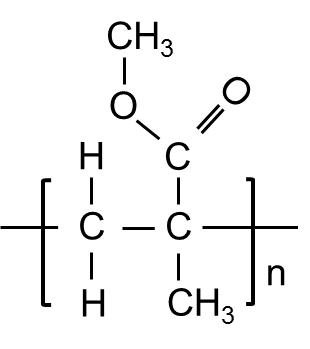
PMMA is produced from the monomer methyl methacrylate mostly by radical polymerization and is entirely amorphous. The structure of the thermoplastic (e.g., mean chain length or degree of cross-linking) is influenced by the pressure, temperature course and duration of the polymerization process. This, in turn, influences the physical and chemical properties of the material.
Structural Formula
Properties
NETZSCH Measurement
| Sample Mass | 12.33 mg |
| Heating Rates | 10 K/min |
| Crucible | Al, pierced lid |
| Atmosphere | N2 (50 ml/min) |
Evaluation
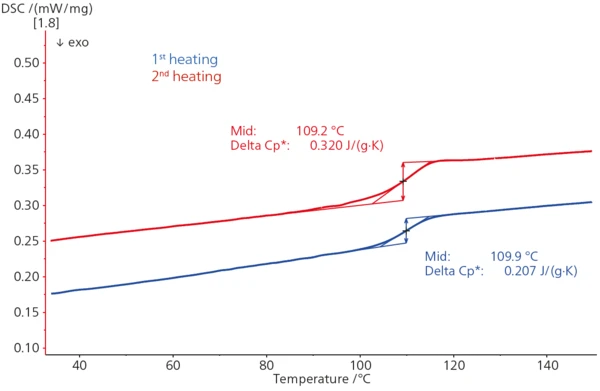
In the above DSC curve − typical for amorphous materials − a Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition can be seen as a step in the endothermal direction with step heights (Δcp) of 0. 21 J/(g*K) (1st heating, blue) and 0.32 J/(g*K) (2nd heating, red) caused by the change in specific heat during transition from a glassy, brittle into a flexible, rubber-like state. The Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition midpoint temperature occurs at 110°C in the 1st heating (blue) and at 109°C in the 2nd heating.