Introduction
Des milliards de kilos de moquette sont produits chaque année dans le monde, dont un pourcentage ( large ) finit dans les décharges, ce qui est problématique car le nylon n'est pas biodégradable. La capacité limitée des décharges et l'impact environnemental de l'élimination des déchets de moquette de cette manière ont fait de la récupération du nylon à partir des déchets de moquette une entreprise de plus en plus importante.
Comme les compositions des moquettes varient et peuvent contenir du Nylon-6 et/ou du Nylon-6,6 ainsi qu'une variété d'autres matériaux, tels que d'autres fibres polymères (par ex. PP, PE, polyester), des adhésifs en latex, des colorants et des charges inorganiques (par ex. CaCO3 et BaSO4),1 la caractérisation de la composition des déchets de moquette et de leur profil de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition thermique est essentielle pour le processus de recyclage. L'analyse thermogravimétrique (TGA) couplée à l'analyse des gaz évolués (EGA) par FT-IR, MS ou GC-MS permet l'analyse simultanée du profil de perte de masse thermique d'un matériau et l'identification des espèces gazeuses évoluant au cours de la Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition.
Dans cette étude, des matériaux récupérés dans des déchets de moquette ont été analysés par TGA-FT-IR, TGA-MS et TGA-GC-MS afin de comparer les capacités des trois méthodes combinées d'analyse thermique et d'analyse des gaz dégagés pour identifier la composition de la moquette recyclée.
1C. Mihut, D. K. Captain, F. Gadala-Maria, et M.D. Amiridis. "Review : Recycling of Nylon from Carpet Waste", Polymer Eng. Sci., Vol. 41(9), pp. 1457-1470, 2001
Expérimental
Les mesures TGA-FT-IR et TGA-MS ont été réalisées à l'aide d'un analyseur thermogravimétrique (TGA) NETZSCH TG 209 F1 Libra® couplé à un spectromètre FT-IR BRUKER Optics TENSOR™ et à un spectromètre de masse quadripolaire NETZSCH QMS 403 Aëolos® (Figure 1). Pour les mesures TGA-GC-MS, le NETZSCH TG 209 F1 Libra® a été couplé à un chromatographe en phase gazeuse Agilent Technologies 7890A équipé d'un spectromètre de masse quadripolaire Agilent 5975C (Figure 2).


Les échantillons de moquette recyclée ont été chauffés dans la thermobalance de 25 à 600°C à 10 K/min sous azote (40 ml/min ; TGA-FT-IR et TGA-MS) ou sous hélium (65 ml/min ; TGA-GC-MS). Les gaz évaporés ont été transférés de la thermobalance à l'instrument d'analyse EGA via une ligne de transfert chauffée à 220°C pour le couplage FT-IR et MS ou à 300°C pour le couplage GC-MS. Pour l'analyse GC-MS, les gaz ont été échantillonnés toutes les quatre minutes et injectés sur une colonne Agilent HP-5MS maintenue à 150°C et éluée avec un débit d'hélium de 2 ml/min. Pour les mesures FT-IR et MS, les gaz ont été introduits en continu dans une cellule à gaz IR maintenue à 200°C ou directement dans l'analyseur MS.
Résultats et discussion
TGA-FT-IR
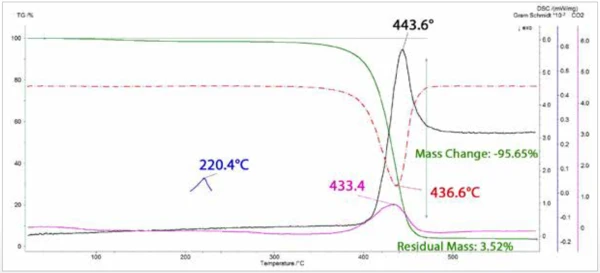
Les courbes de perte de masse (TGA) et de taux de perte de masse (DTG) sont tracées à la figure 3, ainsi que les courbes d'absorption IR totale intégrée (Gram Schmidt) et l'intensité intégrée de la bande d'étirement asymétrique duCO2. Une seule étape de perte de masse a été observée, avec un taux maximal à 436,6°C. Les pics des courbes DTG etCO2 coïncident presque, suivis de près par le pic de la courbe de Gram Schmidt. L'endotherme de Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion à 220°C déterminé avec l'analyse NETZSCH brevetée c-DTA est également illustré.
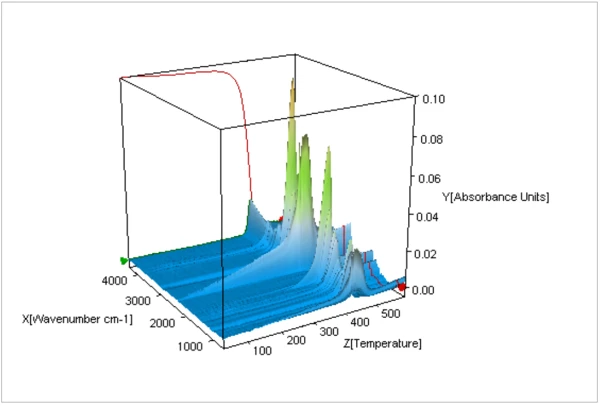
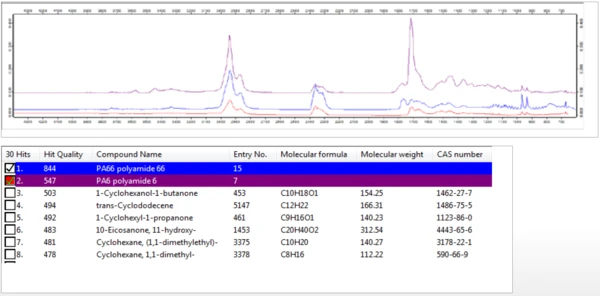
Un tracé tridimensionnel des spectres FT-IR des gaz dégagés pendant la durée de la Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition thermique est présenté à la figure 4. Les spectres extraits individuellement ont été comparés à une base de données de spectres IR afin d'identifier les espèces dégagées à différentes températures au cours de la Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition thermique. Comme le montre la figure 5, les spectres FT-IR des gaz dégagés à 460°C correspondaient à ceux du Nylon-6,6 (PA66) et du Nylon-6 (PA6).
TGA-MS
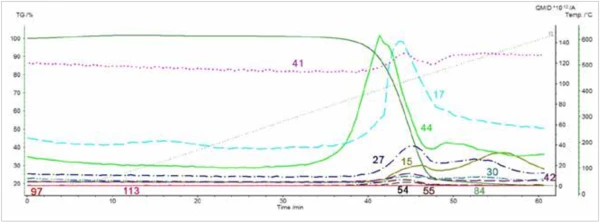
L'évolution duCO2 a été identifiée par l'analyse MS, mais les espèces organiques n'ont pas été identifiées avec certitude en recherchant les spectres de masse extraits de l'acquisition dans la bibliothèque de spectres de masse du NIST. Néanmoins, les pics dans les courants d'ions (Figure 6) pour les numéros de masse 15, 41 et 55 sont compatibles avec le Nylon-6, et les pics dans les courants d'ions pour les numéros de masse 17 et 54 sont compatibles avec le Nylon-6.6. Les courants pour les masses d'ions 27, 30 et 44 sont également montrés. Ils présentent également des pics pendant la Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition, mais les ions produits sont communs aux deux polymères. Aucun pic n'a été observé dans le courant ionique pour les masses 113 (caprolactone) ou 84 (cyclopentanone) ; cependant, ces ions ne sont pas attendus lors de l'analyse par spectrométrie de masse à impact électronique (EIMS).2
Analyse TGA-GC-MS
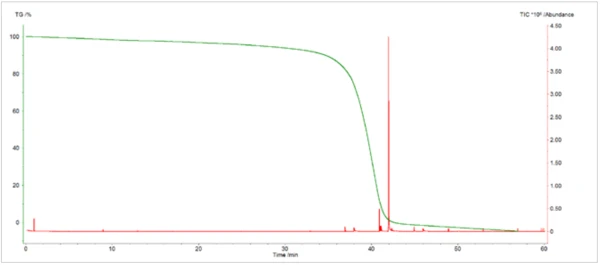
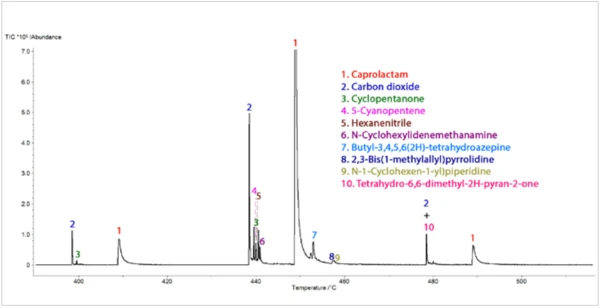
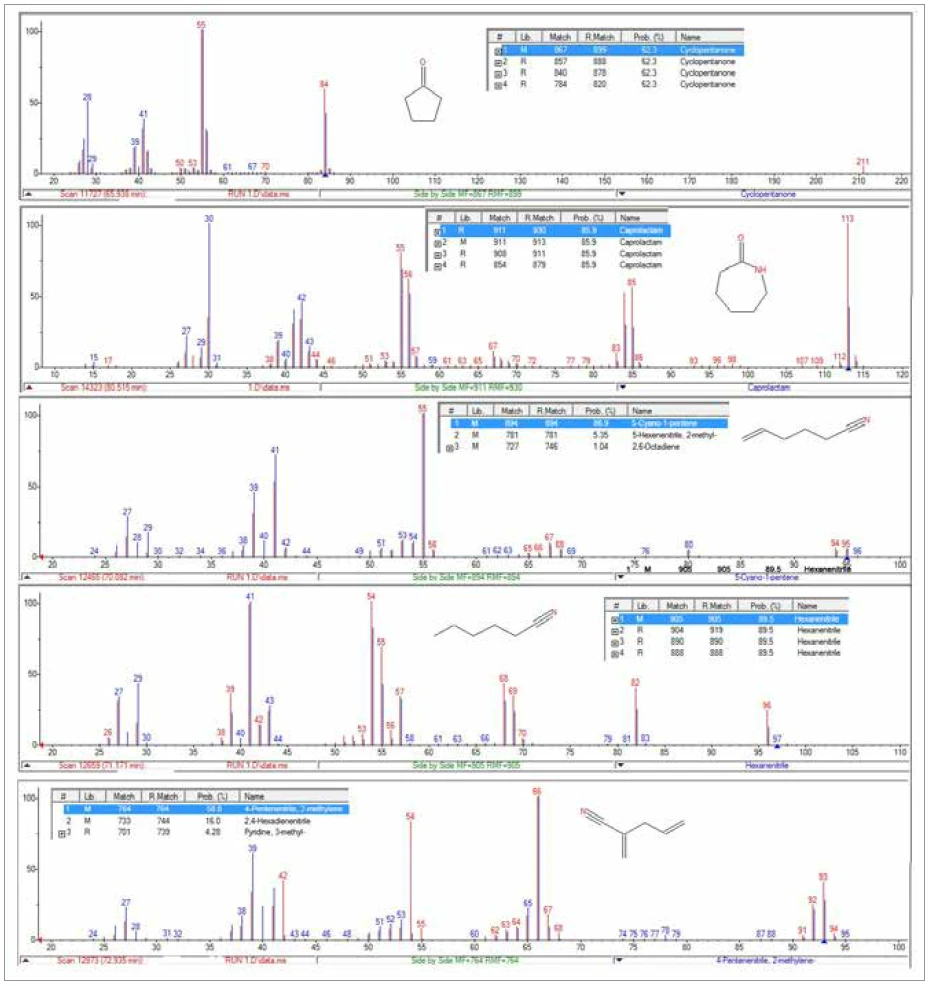
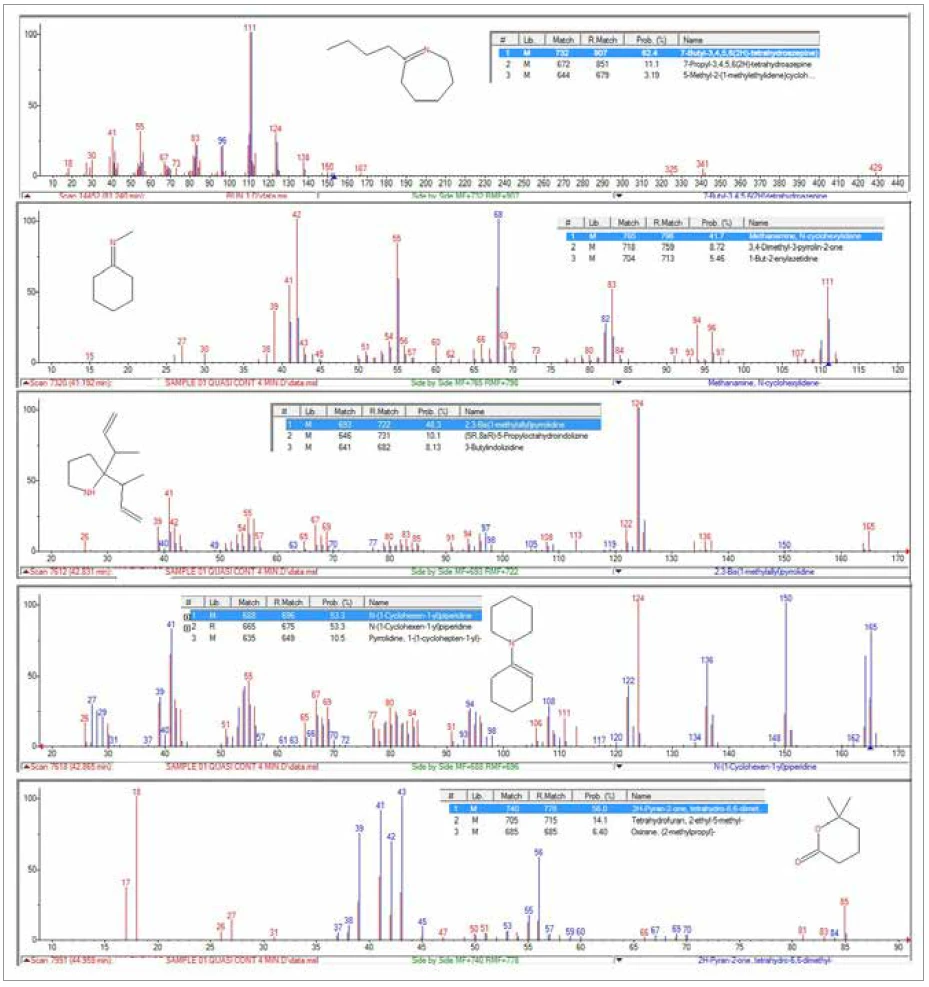
L'analyse TGA-GC-MS a été réalisée en mode quasi-continu en échantillonnant les gaz dégagés pendant la PyrolyseLa pyrolyse est la décomposition thermique de composés organiques dans une atmosphère inerte.pyrolyse de l'échantillon toutes les quatre minutes. La figure 7 présente une superposition du chromatogramme d'ions totaux (TIC) de la mesure GC-MS avec la courbe de perte de masse thermique. La figure 8 montre une vue élargie du TIC avec les identifications de pics qui ont été déterminées à partir de recherches dans la bibliothèque des spectres de masse extraits. Le caprolactame, un produit de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition primaire du Nylon-6, était le principal composant des gaz dégagés. Il a commencé à apparaître dans l'échantillonnage de gaz vers 400°C et a continué à apparaître par impulsions jusqu'à environ 500°C. L'apparition duCO2 dans les échantillons de gaz prélevés entre 400°C et 480°C était également cohérente avec les résultats des analyses TGA-FT-IR et TGA-MS. La séparation chromatographique des composants gazeux au cours de l'analyse GC-MS a permis d'identifier une variété d'autres espèces organiques qui n'avaient pas été identifiées par l'analyse FTIR ou MS (Figure 9). La cyclopentanone est un produit de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition thermique très caractéristique du Nylon-6,6.3
Conclusion
Chaque méthode d'analyse de gaz évoluée présente des forces et des faiblesses spécifiques qui la destinent normalement à des applications particulières. La GC-MS est généralement la plus informative des trois méthodes en raison de la séparation chromatographique des composants gazeux, qui permet leur identification individuelle. Dans cette étude, la GC-MS a permis l'identification la plus claire du caprolactame, confirmant que le matériau était principalement constitué de Nylon-6. Elle a également permis d'identifier des produits cyclopentanone et nitrile qui sont plus caractéristiques du Nylon-6,6. Diverses autres espèces organiques cycliques, qui pourraient être des produits du Nylon-6,6, ont été identifiées pour la première fois dans le cadre de cette étude. Les résultats de la spectrométrie de masse à impact électronique (EIMS) et du FT-IR ont confirmé la présence des deux polymères de nylon dans le matériau de la moquette recyclée. Les masses des ions moléculaires caractéristiques du Nylon-6 et du Nylon-6,6 ont été identifiées par EIMS. Bien que le FT-IR ait identifié les deux polymères comme composants potentiels du matériau, en raison des similitudes entre les spectres, cette méthode d'analyse des gaz évolués s'est avérée la moins définitive en ce qui concerne le(s) polymère(s) de nylon spécifique(s) réellement présent(s).
Comme le démontre cette étude, l'analyse thermogravimétrique couplée à l'analyse des gaz évolués (TGA-EGA) est un outil analytique instructif et rapide qui permet non seulement de déterminer simultanément le profil de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition thermique et la composition chimique des matériaux, mais aussi d'élucider les processus chimiques responsables de la perte de masse thermique en identifiant les espèces de gaz évolués correspondantes.