30.11.2020 by Dr. Natalie Rudolph, Dr. Stefan Schmölzer
How to Determine the Process Window for SLS Powders Using DSC
Powder Bed Fusion (PBF), often called Selective Laser SinteringSintering is a production process for forming a mechanically strong body out of a ceramic or metallic powder. Sintering (SLS), the component is built up in layers in a powder bed using a laser beam that passes over the cross-section of the layer to locally melt the powder. In order to characterize a polymer powder for its suitability for SLS and to determine the possible process window, Differential Scanning Calorimetry (DSC) is used. Learn how to set up and interpret the measurements!
During Powder Bed Fusion (PBF), often called Selective Laser SinteringSintering is a production process for forming a mechanically strong body out of a ceramic or metallic powder. Sintering (SLS), the component is built up in layers in a powder bed using a laser beam that passes over the cross-section of the layer to locally melt the powder. However, to avoid inhomogeneous solidification and warpage, the melt is kept at temperatures above the crystallization temperature to prevent it from solidifying before the whole part is finished. The surrounding powder stays solid and keeps the shape of the molten geometry.
Read here our introduction to the SLS process!
The most commonly applied SLS powder to date is polyamide 12 (PA12). However, the industry is continuously looking for new polymer powders to open up opportunities for new applications and market segments.
How to set up the measurement
In order to characterize a polymer powder for its suitability for SLS and to determine the possible process window, Differential Scanning Calorimetry (DSC) measurements are needed.
A dynamic measurement is performed to determine the melting and crystallization behavior using a NETZSCH DSC 214 Polyma. In this example, a PA12 powder sample of 5 mg was weighted into an aluminum pan with concave bottom (Concavus® Al) and closed lid. The sample was cooled from room temperature to start the measurement at 0°C. It was then heated to 200°C at a heating rate of 10 K/min and cooled down at the same rate of 10 K/min back to 0°C. This cycle was repeated several times. All measurement conditions are summarized in the following table:
Table 1: Measurement conditions
| Pan | Concavus® Al, closed lid |
| Sample weight | 5.024 mg |
| Atmosphere | N2 |
| Temperature range | 0°C to 200°C at a heating and cooling rate of 10 K/min |
Determining the process window with the first cycle
In figure 1, the results of the 1st heating (blue) and cooling cycle (green) are depicted. The onset of melting occurs at 181°C and the onset of crystallization at 153.4°C (here labelled as “End” due to the analysis from low to high temperatures).
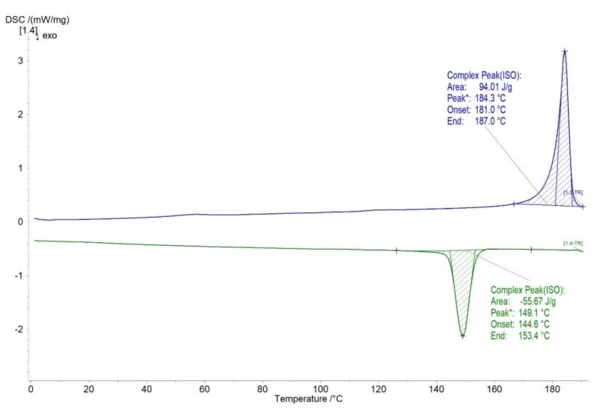
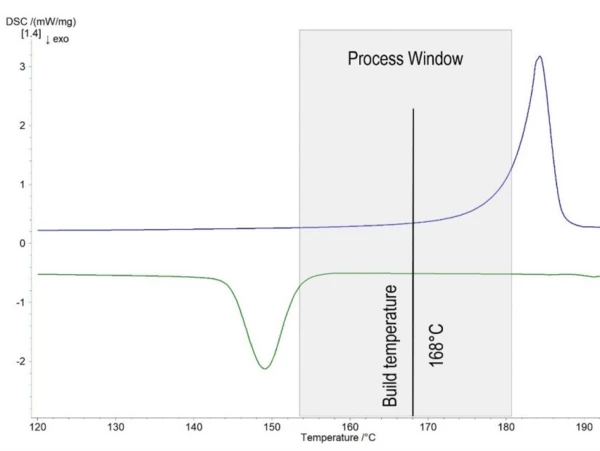
Remembering that the process temperature has to be set between the onset of melting and crystallization, the measured PA12 powder offers a process window of 27.6°C (figure 2). The typical build temperature for this material is 168°C, which is in the middle of the process window. In cases in which the build temperature is too close to the onset of crystallization, the parts have larger temperature gradients and show warpage. In cases in which the build temperature is too close to the onset of melting, the hot melt acts as hot spots. SinteringSintering is a production process for forming a mechanically strong body out of a ceramic or metallic powder. Sintering of surrounding solid powder occurs on the surfaces, resulting in lateral growth of the parts.
Comparing the first and second heating cycles to study the material in more detail
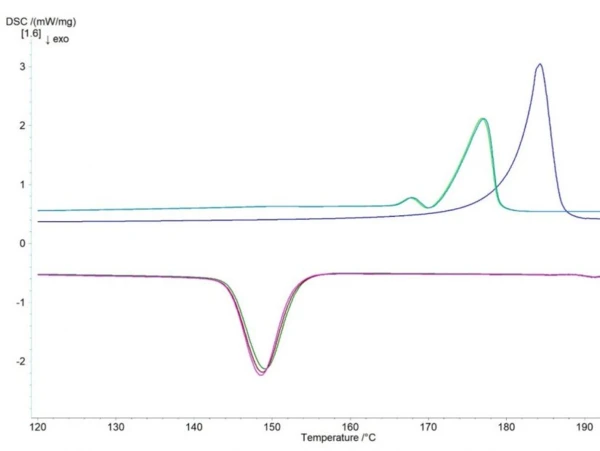
While for the determination of the process window the first heating of the powder is most important, it is advised to compare the second heating as well. In the case of this measurement, the heating and cooling cycles were repeated multiple times and figure 3 shows the results of three runs.
First, it can be seen that the melting peak of the powder (1st heating) is shifted to higher temperatures due to the slightly reduced contact surface of the powder with the pan as well as the higher surface energy of the powder. Second, it can be seen that the 2nd and all subsequent heating cycles show a double peak with lower onset temperature. This peak indicates a different crystalline structure than that of the powder, which is rather unique for this particular PA12 powder and not observed for other PA12s. It shows that during cooling, in addition to the common α- and γ-spherulites, an intermediate crystal structure is formed that can be observed as a small peak at the beginning of melting. However, while this is of scientific interest, it is not relevant for the SLS printing process.
Crystallization is time-dependent – why is that relevant in the SLS process?
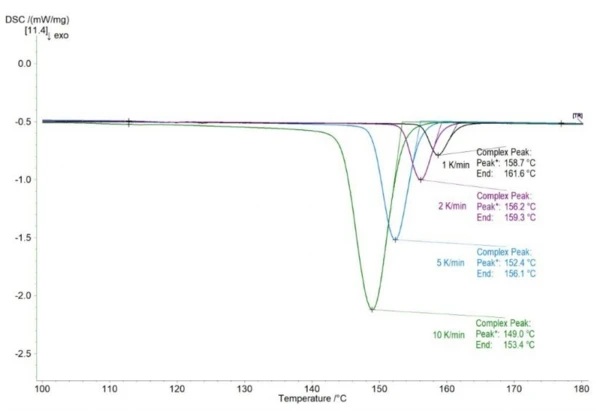
Finally yet importantly, crystallization is a time-dependent process and therefore, low cooling rates shift the crystallization peak to higher temperatures; this needs to be taken into account when determining the lower end of the process window. Figure 4 shows the crystallization peaks for DSC measurements at 10, 5, 2 and 1 K/min. It can be seen that the onset (here “End”) as well as the peak temperature shift to higher temperatures as the cooling rate decreases. Instead of an onset temperature of 153.4°C at 10 K/min, the onset occurs already at 161.6°C at 1 K/min.
Isothermal crystallization studies allow for successful work pieces
While the actual temperature on the surface of the powder bed can be measured with IR thermometers, the temperature in lower layers is unknown on a commercial SLS printer. Over the whole duration of the build, which can be up to 12 hours without subsequent cooling, isothermal crystallization can occur after some time, especially if the build temperature fluctuates too much due to coating with cold powder, non-uniform distribution of parts inside the build envelope, or unbalanced heaters, just to name a few. Therefore, isothermal crystallization studies are needed to evaluate this behavior for the selected polymer powders and thus qualify them for SLS. Read the article about isothermal crystallization behavior here!
Read more about the characterization of SLS powder in our upcoming articles!

FREE E-Book
Thermal Analysis and Rheology in Polymer Additive Manufacturing
Discover the secrets behind AM's game-changing capabilities! Our newly released ebook delves deep into the heart of AM, unveiling the power of reliable material characterization techniques, specifically thermal analysis and rheology.