Introduzione
Il politetraflouroetilene (PTFE) è noto per la sua applicazione quotidiana come rivestimento antiaderente per padelle e altre pentole. Il PTFE è molto poco reattivo e offre un'elevata resistenza chimica. Grazie a queste proprietà, non viene utilizzato solo in applicazioni mediche, ma anche nell'industria, ad esempio in contenitori e tubazioni per prodotti chimici corrosivi e reattivi. Anche componenti come cuscinetti, boccole e ingranaggi, in cui è necessaria un'azione di scorrimento, sono realizzati in PTFE.
La caratterizzazione termica di un materiale in PTFE è stata realizzata utilizzando diverse tecniche di analisi termica e di test delle proprietà termofisiche. Le misure sono state effettuate tra -170°C e 700°C (a seconda del metodo). L'espansione termica e le variazioni di densità sono state determinate mediante dilatometria a spinta (DIL, basata, ad esempio, su ASTM E831 e DIN 51045). L'analisi meccanica dinamica (DMA) è stata utilizzata per analizzare le proprietà viscoelastiche (Elasticità e modulo di elasticitàL'elasticità della gomma o elasticità dell'entropia descrive la resistenza di qualsiasi sistema di gomma o elastomero contro una deformazione o uno sforzo applicato dall'esterno. modulo di accumulo e di perdita). La Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica è stata misurata con la tecnica laser fl ash (LFA, basata, ad esempio, su ASTM E1461, DIN EN821). La combinazione dei dati di Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica con il calore specifico e la densità consente di calcolare la Conduttività termicaLa conducibilità termica (λ con unità di misura W/(m-K)) descrive il trasporto di energia - sotto forma di calore - attraverso un corpo di massa come risultato di un gradiente di temperatura (vedi fig. 1). Secondo la seconda legge della termodinamica, il calore fluisce sempre nella direzione della temperatura più bassa.conduttività termica del polimero. Il comportamento di Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione è stato studiato utilizzando l'analisi termica simultanea (STA, basata, ad esempio, su ASTM E1131, ASTM D3850, DIN 51006, ISO 11357, DIN 51004, DIN 51007, ecc.) I gas evoluti sono stati analizzati con uno spettrometro di massa (QMS) e con la spettroscopia infrarossa a trasformata di Fourier (FT-IR).
Il PTFE presenta diverse transizioni nell'intero intervallo di temperatura. Al di sotto dei 19°C si ottiene una fase triclina ben ordinata, mentre tra i 19°C e i 30°C il PTFE forma una fase esagonale parzialmente ordinata. Al di sopra dei 30°C e fino al Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa).punto di fusione (328°C), il materiale presenta una fase pseudo-esagonale molto disordinata. Altre transizioni si trovano a -115°C e 131°C, attribuibili alla fase amorfa [1]. Alcune fonti di letteratura (ad esempio, [3], [4]) descrivono la trasformazione di fase a 131°C come una transizione vetrosa.
Politetraflouroetilene = PTFE
- Meglio conosciuto come Teflon®*
- Scoperto da Roy Plunkett nel 1938
- Formula molecolare: CnF2n+2
- Massa molecolare: 100,02 g/mol
- Densità: 2,2 g/cm³
- Punto di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione: 327°C
*Teflon® è un marchio registrato di E.I. DuPont de Nemours and Company.
Il PTFE analizzato in questo lavoro è stato fornito da ElringKlinger Kunststofftechnik GmbH, Heidenheim.
Risultati del test
A) Proprietà viscoelastiche
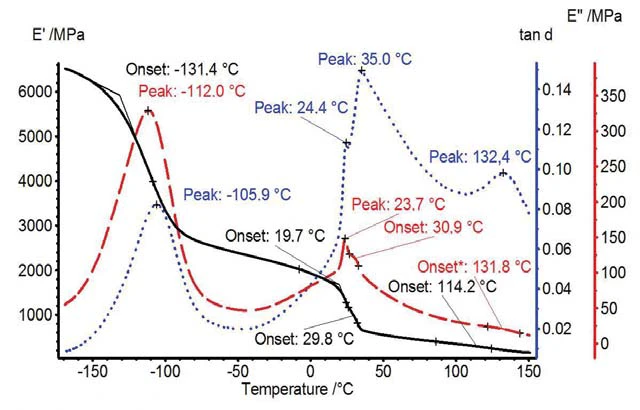
La Figura 1 presenta le proprietà meccaniche E´, E´´ e tanδ determinate. L'aumento del Elasticità e modulo di elasticitàL'elasticità della gomma o elasticità dell'entropia descrive la resistenza di qualsiasi sistema di gomma o elastomero contro una deformazione o uno sforzo applicato dall'esterno. modulo di accumulo a -131°C può essere attribuito alla transizione vetrosa della fase amorfa. Tra 20°C e 40°C si osservano due transizioni solido-solido. Un altro gradino nella curva E´ è stato osservato a 115°C a causa di una transizione solido-liquido della fase amorfa [1], talvolta caratterizzata anche come transizione vetrosa [3], [4].
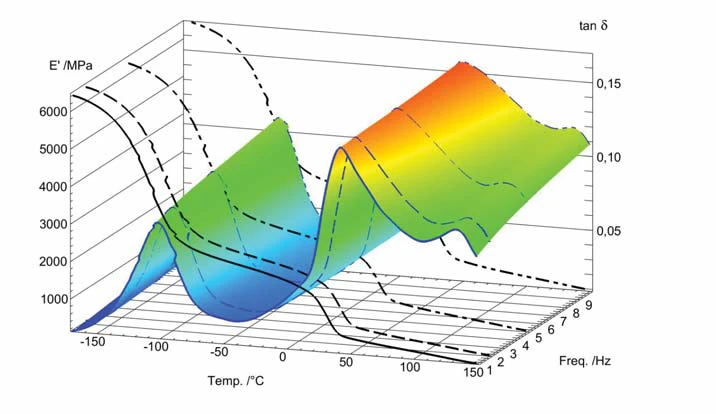
La figura 2 mostra un grafico 3-D di una misura in multifrequenza (1, 2, 5 e 10 Hz). Si può notare che tanδ aumenta con la frequenza a una data temperatura.
B) Espansione termica, variazione di densità
Il PTFE si espande con un tasso di espansione costante tra -170°C e 20°C (figura 3). A temperatura ambiente è stato rilevato un salto nell'espansione termica dovuto alla transizione solido-solido. Al di sopra della Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase, l'espansione termica aumenta continuamente con un tasso di espansione leggermente crescente.
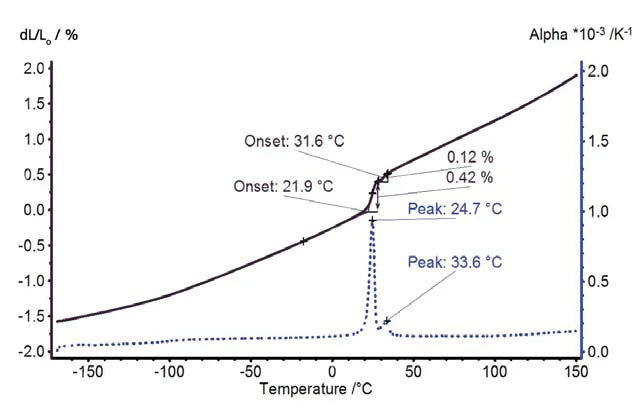
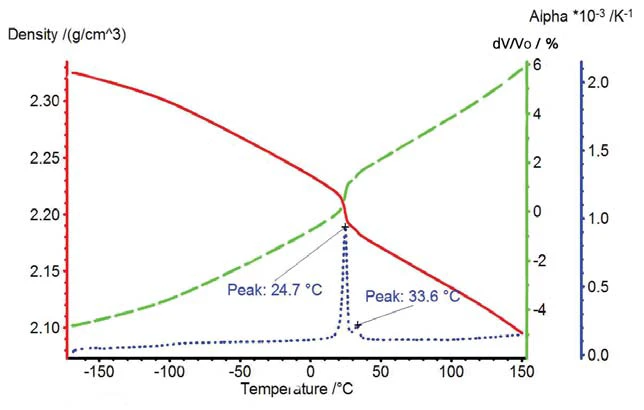
L'Espansione volumetricaIl volume di un gas, di un solido o di un liquido cambia se cambiano la temperatura, la pressione o le forze che agiscono su quel gas/solido/liquido. Nel caso dell'analisi termica, si tratta di variazioni dipendenti dalla temperatura.espansione volumetrica e la variazione di densità del PTFE sono rappresentate nella figura 4. La transizione solido-solido corrisponde a una variazione di volume superiore all'1%.
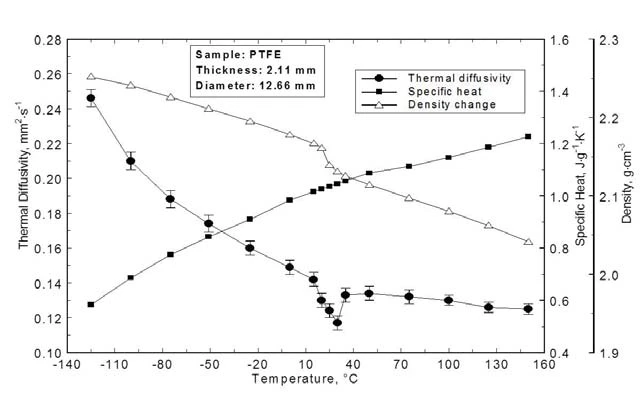
Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.Diffusività termica, variazione di densità e calore specifico
La Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica, il calore specifico e la variazione di densità del PTFE sono mostrati nella figura 5. La diffusività diminuisce continuamente con la temperatura, come previsto dalla fisica dello stato solido per la conduzione dei foni. La diffusività diminuisce continuamente con la temperatura, come previsto dalla fisica dello stato solido per la conduzione fonetica. La transizione solido-solido a RT può essere chiaramente identificata, mentre le altre transizioni a -131°C e a 115°C non sono visibili.
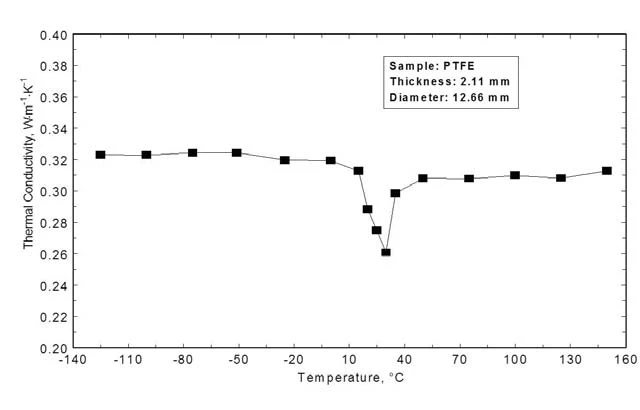
Conducibilità termica
La Figura 6 mostra la conducibilità termica calcolata attraverso la Diffusività termicaLa diffusività termica (a con unità di misura mm2/s) è una proprietà specifica del materiale per caratterizzare la conduzione termica instabile. Questo valore descrive la velocità con cui un materiale reagisce a una variazione di temperatura.diffusività termica, il calore specifico e la densità. Nell'intervallo di bassa temperatura, la conducibilità termica è quasi costante (0,32 Wm-1K-1). Durante la Transizioni di faseIl termine transizione di fase (o cambiamento di fase) è più comunemente usato per descrivere le transizioni tra gli stati solido, liquido e gassoso. transizione di fase tra 10°C e 40°C, la conducibilità termica diminuisce di oltre il 10% e anche a temperature più elevate - dopo che il segnale si è nuovamente innalzato - la conducibilità termica è significativamente inferiore rispetto alla regione precedente al cambiamento di fase.
D) Decomposizione termica, analisi dei gas
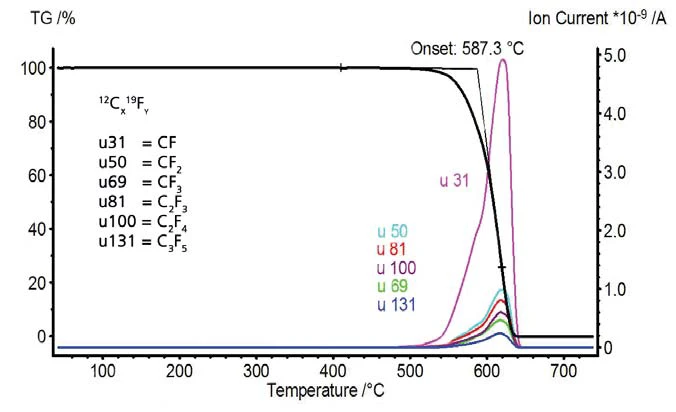
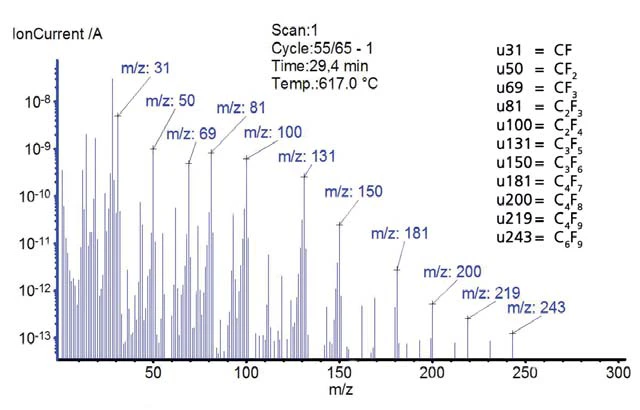
Le variazioni di massa e i segnali dello spettrometro di massa in funzione della temperatura sono rappresentati nelle figure 7 e 8. Il PTFE non mostra alcuna perdita di massa fino a quando non inizia la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione pirolitica a 587°C. Lo spettrometro di massa ha rilevato variazioni dell'intensità della corrente ionica per i numeri di massa 31, 50, 69, 81, 100, 131, 150, 181, 200, 219 e 243. Questi numeri di massa indicano frammenti tipici del PTFE. Questi numeri di massa indicano frammenti tipici del PTFE. Il politetrafl uoroetilene si decompone completamente; nell'atmosfera di gas inerte non rimane alcuna massa residua.
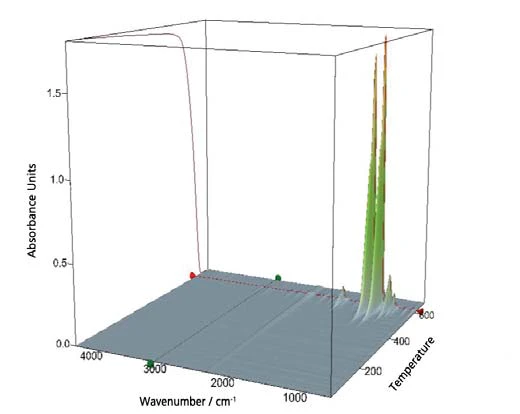
Contemporaneamente alla TGA-MS, è stata eseguita una misurazione FT-IR. La figura 9 mostra una raccolta di tutti gli spettri IR rilevati sotto forma di cubo tridimensionale. Inoltre, è incluso anche il segnale TGA sulla faccia laterale del cubo.
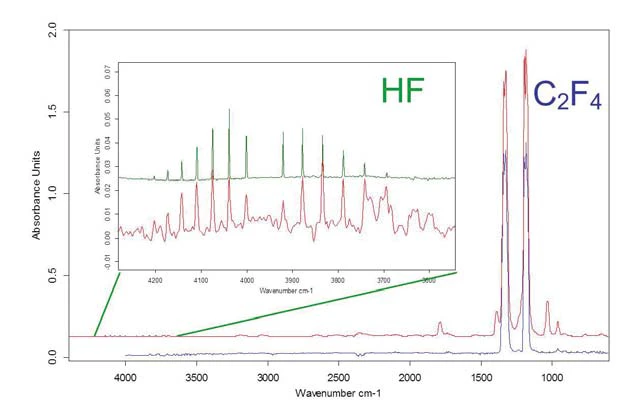
Da questo diagramma tridimensionale sono stati estratti singoli spettri a una temperatura vicina ai massimi dei picchi visibili (figura 10) e confrontati con i dati di library. Sono stati identificati HF e tetrafluoroetilene.
Conclusione
Sono state testate diverse proprietà termofisiche e termomeccaniche per comprendere meglio il PTFE. La transizione solido-solido è stata identificata da tutte le tecniche di analisi termica impiegate. Solo l'analisi meccanica dinamica è stata in grado di individuare le transizioni relative alla fase amorfa.