
17.05.2021 by Dr. Natalie Rudolph, Dr. Stefan Schmölzer
How Fillers Affect the Crystallization Behavior of SLS Powders
Due to the still limited number of available materials for the Selective Laser Sintering process, there is a constant demand for materials with different properties. The addition of any filler to SLS powder typically has an effect on the processing behavior. Today, we investigate the crystallization behavior of PA12 powder filled with copper spheres and flakes.
Il peut s'agir d'une plus grande ductilité (par exemple, PA11), d'une meilleure stabilité dimensionnelle (par exemple, ajout de billes de verre), d'une Conductivité électrique (SBA)La conductivité électrique est une propriété physique indiquant la capacité d'un matériau à permettre le transport d'une charge électrique.conductivité électrique ou thermique plus élevée (par exemple, charges telles que l'aluminium ou le cuivre) ou d'une rigidité et d'une résistance accrues (par exemple, ajout de fibres de verre ou de carbone).
Les charges agissent comme des sites de nucléation
L'ajout d'une charge à la poudre SLS a généralement un effet sur le comportement de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation car les surfaces des charges agissent comme des sites de nucléation hétérogènes. Les charges conductrices peuvent également affecter la température du matériau et, par conséquent, présenter un comportement modifié supplémentaire.les mesures dynamiques de calorimétrie différentielle à balayage (DSC) peuvent fournir une première indication d'un changement de comportement, qui peut ensuite être analysé plus en détail par des mesures isothermes à la température de fabrication ou à une température proche de celle-ci.
Ajout de charges conductrices en cuivre
Pour comprendre l'effet des charges conductrices, des chercheurs de l'Institut de technologie des polymères (LKT) de l'Université d'Erlangen-Nuremburg ont expérimenté des charges de cuivre [1]. Ils ont utilisé différents mélanges de sphères et de paillettes de cuivre (figure 1) à des teneurs variables et ont analysé le comportement de traitement ainsi que les propriétés conductrices résultantes dans le but de générer des composants complexes pour la gestion thermique. Divers échantillons ont été préparés en ajoutant à la poudre de PA12 des sphères de cuivre à 5 et 10 % et des paillettes de cuivre à 5 %. La densité d'énergie de 0,043 J/mm2 a été maintenue constante pour tous les matériaux afin de détecter tout changement dans le comportement du processus dû aux charges.

Détermination de la fenêtre de traitement et du comportement de cristallisation de la poudre PA12 avec des particules de cuivre
Sur le site NETZSCH Analyzing & Testing, un DSC 214 Polyma a été utilisé pour analyser la fenêtre de processus et le comportement de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation de ces différents mélanges de poudre de PA12 avec des particules de cuivre par rapport au matériau PA12 pur.
Pour chaque mesure, un échantillon de 5 mg a été coupé et placé dans une casserole en aluminium à fond concave (Concavus®al) et un couvercle fermé. L'échantillon a été refroidi de la température ambiante à -20°C pour commencer la mesure. Il a ensuite été chauffé à 200°C à une vitesse de 10 K/min et refroidi à la même vitesse de 10 K/min jusqu'à -20°C.
Le tableau suivant résume les conditions de mesure.
Tableau 1 : Conditions de mesure
| Cuvette | Concavus®al, couvercle fermé |
| Poids de l'échantillon | 5 mg |
| Atmosphère | N2 |
| Plage de température | -20°C à 200°C à une vitesse de chauffage et de refroidissement de 10 K/min |
Les résultats pour le PA12 pur ont été discutés en détail dans ce billet de blog précédent!
Changements dans le comportement de cristallisation
Dans la figure 2, les résultats dupremier chauffage et dupremier refroidissement sont représentés pour les 4 échantillons. Comme on peut le voir, la Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). température de fusion maximale n'est pas affectée par l'ajout des charges (la différence de surface est liée aux différentes teneurs en charges et n'a pas été normalisée dans ce graphique).
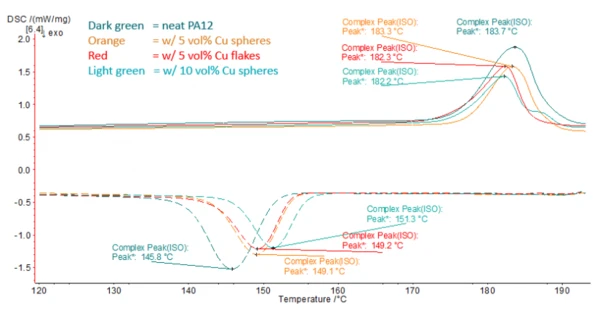
Cependant, on peut constater que la température maximale de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation ainsi que le début de la CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation sont déplacés vers des températures plus élevées avec l'augmentation de la teneur en charge.
Le pic passe de 145,8°C pour le matériau pur à 149,1°C pour 5 % de charges de cuivre et à 151,3°C pour 10 %, respectivement.
On peut en conclure que les charges agissent comme des sites de nucléation et accélèrent la CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée.cristallisation. Cela conduit à une fenêtre de traitement légèrement réduite et doit être pris en compte lors de la sélection des conditions de traitement.
Un effet similaire a été observé par WILO SE, un fournisseur de premier plan de pompes et de systèmes de pompage pour les fibres de verre et de carbone. Lisez l'étude de cas ici !
Mieux comprendre le comportement de la cristallisation
Des études supplémentaires du comportement de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée. cristallisationIsothermeLes essais à température contrôlée et constante sont dits isothermes.isotherme peuvent être utiles pour mieux comprendre ce comportement. Une description détaillée de la procédure de mesure est donnée ici pour la poudre de PA12 non chargée.
Les chercheurs du LKT ont réalisé ces études isothermes supplémentaires à 165°C sur les échantillons remplis de cuivre et ont constaté que le temps de CristallisationLa cristallisation est le processus physique de durcissement au cours de la formation et de la croissance des cristaux. Au cours de ce processus, la chaleur de cristallisation est libérée. cristallisation est réduit en raison de l'ajout de charges, ce qui confirme l'effet de nucléation. L'étude complète peut être consultée ici [Open Access] !
À propos de l'Institut de technologie des polymères (LKT)
L'Institut de technologie des polymères est un institut de recherche universitaire de l'Université Friedrich-Alexander d'Erlangen-Nuremberg. Il est l'un des leaders de la recherche sur la fabrication additive, en particulier sur le SLS. Outre ces domaines de recherche, l'institut travaille également sur des sujets interdisciplinaires tels que le mélange de matériaux d'apport, la simulation du traitement et des applications, les thermoplastiques réticulés par rayonnement, le traitement doux et bien d'autres encore.
Sources d'information
[1] Lanzl, L., Wudy, K., Greiner, S., Drummer D., Selective Laser Sintering of Copper Filled Polyamide 12 : Characterization of Powder Properties and Process Behavior, Polymer Composites, pp. 1801-1809, 2019.

Livre électronique GRATUIT
Analyse thermique et rhéologie dans la fabrication additive de polymères
Découvrez les secrets des capacités de l'AM qui changent la donne ! Notre nouvel ebook plonge au cœur de l'AM, dévoilant la puissance de techniques fiables de caractérisation des matériaux, en particulier l'analyse thermique et la rhéologie.