Introduzione
Molti solidi farmaceutici presentano PolimorfismoIl polimorfismo è la capacità di un materiale solido di formare diverse strutture cristalline (sinonimi: forme, modificazioni).polimorfismo, cioè la capacità di una sostanza di esistere in diverse forme cristalline; di conseguenza, hanno disposizioni molecolari diverse nel reticolo cristallino. A causa delle loro differenze strutturali, i polimorfi possono avere proprietà solide diverse, come densità, colore, Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa).punto di fusione, solubilità, proprietà meccaniche, ecc. Di conseguenza, la forma polimorfa può avere un'influenza significativa sulla lavorazione farmaceutica e può, ad esempio, influenzare la macinazione, la granulazione e la comprimibilità [1].
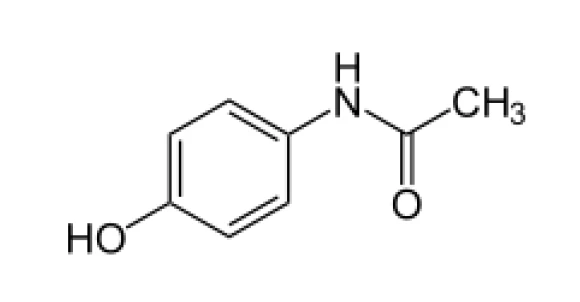
Anche il paracetamolo, utilizzato per il trattamento del dolore e della febbre, è un polimorfo, cioè esiste in diverse forme cristalline. Ognuna di queste modifiche si comporta in modo diverso, in particolare per quanto riguarda la stabilità termodinamica e la capacità di compressione. Quest'ultima proprietà influenza la comprimibilità del paracetamolo, cioè la sua capacità di essere trasformato in una compressa sotto l'effetto della pressione. È stato dimostrato che il paracetamolo ortorombico (chiamato anche forma II) può essere utilizzato per la compressione diretta. La sua migliore comprimibilità rispetto alla forma monoclinica del paracetamolo (forma I) è dovuta alla presenza di piani di scorrimento paralleli nella struttura cristallina ortorombica. [5, 6]
Tuttavia, la forma monoclinica è quella più frequentemente utilizzata in commercio [4], probabilmente per la sua migliore stabilità termodinamica. Poiché la modificazione del paracetamolo è strettamente associata alle sue proprietà, è essenziale sapere in quale forma è presente prima della lavorazione, in particolare per la produzione di compresse che richiedono la compattazione. Di seguito, la modificazione del paracetamolo viene determinata mediante calorimetria differenziale a scansione.
Condizioni di misura
1.69 mg di paracetamolo sono stati misurati in un crogiolo di alluminio sigillato con un coperchio forato. Il campione è stato riscaldato due volte a 200°C a una velocità di riscaldamento di 10 K/min. Tra i due riscaldamenti, è stato raffreddato a una velocità controllata di 10 K/min.
Risultati del test
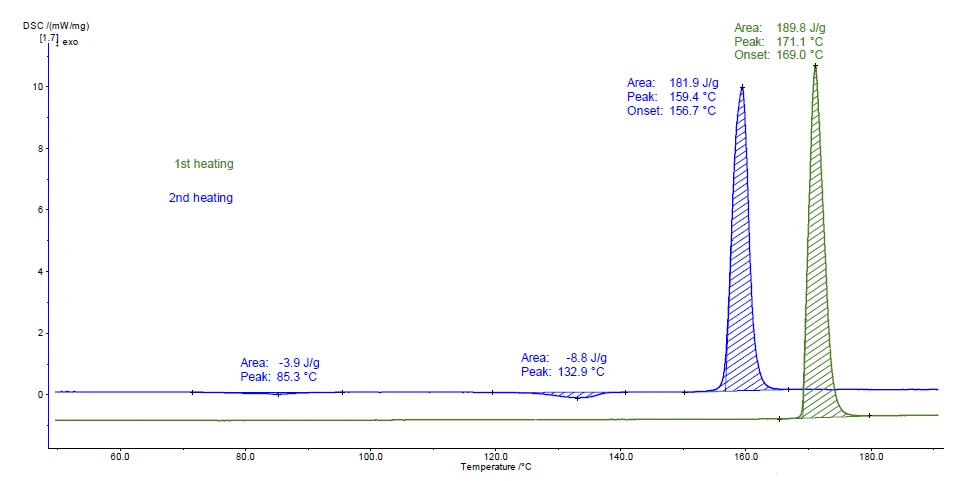
La Figura 2 mostra le curve di misurazione per i due riscaldamenti. Il picco rilevato a 169°C (temperatura di insorgenza) durante il primo riscaldamento (curva verde) corrisponde molto bene al valore del Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa).punto di fusione della modifica cristallina I (vedi tabella 1).
Tab 1: Temperatura di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione delle diverse modificazioni del paracetamolo [2, 3]
I picchi esotermici a 85°C e 133°C (temperatura di picco) che si verificano nel secondo riscaldamento indicano la CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione. La modifica formata fonde successivamente a 156,7°C (temperatura di insorgenza); ciò è tipico della forma cristallina II.
Conclusione
La caratterizzazione dello stato solido è di importanza centrale per l'industria farmaceutica, poiché i farmaci sono prodotti prevalentemente come solidi. Selectione della forma solida ottimale è un aspetto critico nello sviluppo dei farmaci, poiché essi possono esistere in più forme di struttura cristallina. Questo PolimorfismoIl polimorfismo è la capacità di un materiale solido di formare diverse strutture cristalline (sinonimi: forme, modificazioni).polimorfismo mostra diverse proprietà fisiche che influenzano non solo le proprietà biofarmaceutiche ma anche la comprimibilità.
Un singolo riscaldamento DSC alla Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). temperatura di fusione consente di identificare la modifica del paracetamolo in esame, permettendo di stimare il suo comportamento durante la compressione.