Введение
Кофеин - основной активный ингредиент кофе. Однако в некоторых случаях - например, при повышенном кровяном давлении, чувствительном желудке, беременности или взаимодействии с лекарственными препаратами - предпочтительнее пить кофе без кофеина. В 1903 году была разработана первая процедура декофеинизации кофейных зерен. Для извлечения кофеина из сырых зерен использовались вода и бензол (метод Розелиуса).arcПозже вместо бензола стали использовать другие растворители, такие как дихлорметан или этилацетат. В настоящее время распространены такие новые процессы, как экстракция чистой водой (швейцарский водный процесс), триглицеридный процесс или экстракция сверхкритическим диоксидом углерода. [1]
Однако кофе без кофеина все равно содержит некоторое количество остаточного кофеина. В чистых сырых кофейных зернах содержится от 0,8 до 4 % кофеина, в зависимости от сорта кофейных зерен [2]. В ЕС допускается остаточное содержание 0,1% кофеина в необжаренных зернах при продаже кофе без кофеина [3]. Количество кофеина, переходящего из зерна в чашку сваренного кофе, зависит от количества используемой кофейной гущи, вида обжарки, размера зерен кофейной гущи после помола, времени и давления экстракции, а также температуры воды. В результате содержание кофеина в чашке (150 мл) кофе без кофеина составляет около 3 мг, в то время как в обычной чашке кофе содержится от 50 до 100 мг кофеина. Содержание кофеина в растворимом кофе также меньше, чем в обычном. В зависимости от сорта используемых зерен и процесса производства растворимый кофе содержит лишь около 50 % кофеина, содержащегося в фильтр-кофе. Средние значения колеблются между 30 и 90 мг на чашку объемом 150 мл. Производители растворимого кофе без кофеина утверждают, что их продукция содержит менее 5 мг кофеина на порцию.
Основное отличие растворимого кофе от заварного заключается в способе приготовления. Как и обычный кофе, растворимый кофе получают из зерен, которые перемалывают и варят, а затем сублимируют или высушивают в виде концентрированного порошка. Это означает, что после смешивания порошка с водой он приобретает обычный кофейный вкус и текстуру.
В данном исследовании различные сорта растворимого кофе с кофеином и без него были исследованы с помощью TGA-GC-MS (термогравиметрическая газохроматографическая масс-спектрометрия, STA 449 F3 Jupiter® , соединенная с Agilent GC 8890 и Agilent MDS 5975) для определения содержания кофеина.
Для подготовки образцов их слегка измельчали и сжимали в тигле, затем переносили в STA. Измерения ТГА проводились с поправкой на базовую линию. Образцы нагревались в инертной атмосфере до 850°C для выделения летучих соединений, таких как кофеин. Выделившиеся соединения собирались на криоловушке ГХ при -50°C, затем отделялись и идентифицировались после проведения ТГА.
Таблица 1: Параметры измерения ТГА
| Образец | 1 (сублимированный) | 2 (высушенный распылением) | 3 (сублимированный) | 3a без кофеина (сублимированный) | Чистый кофеин | |||
| Масса образца | 7.26 мг | 7.13 мг | 7.46 мг | 7.38 мг | 10.39 мг | |||
| Тигель | Открытый тигель из Al2O3 (85 мкл) | |||||||
| Держатель образца | TGA, тип S + накладная пластина | |||||||
| Печь | SiC | |||||||
| Температурная программа | RT - 850°C | |||||||
| Скорость нагрева | 10 К/мин | |||||||
| Газовая атмосфера | Гелий | |||||||
| Расход газа (общий) | 70 мл/мин | |||||||
Таблица 2: Параметры измерения ГХ-МС
Режим криоловушки | ||||||||
| Колонка | Agilent HP-5ms | |||||||
| Длина колонки | 30 m | |||||||
| Диаметр колонки | 0.25 мм | |||||||
| Температура криоловушки | -50°C, 81 мин | |||||||
| Температура колонки | 45°C, 83 мин Изотерма от 45°C до 300°C, 10 К/мин | |||||||
| Газ | Гелий | |||||||
| Поток газа (разделение) | 20 мл/мин (10:1) | |||||||
| Клапан | каждые 1 мин | |||||||
Результаты и обсуждение
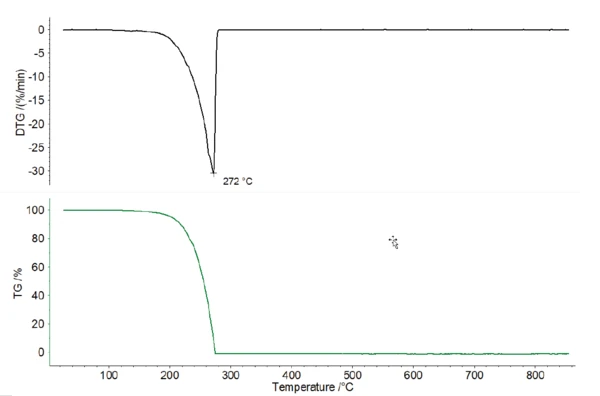
Все образцы кофе показали несколько значительных ступеней потери массы между комнатной температурой и 850°C; см. рис. 1. При скорости нагрева 10 К/мин было невозможно четко разделить этапы потери массы. Все четыре образца показали относительно схожее поведение. Кроме того, только с помощью ТГА не удалось четко определить высвобождение кофеина. Чистый кофеин демонстрирует пик ДТГ (скорость потери массы) при 272°C (см. рис. 2), который перекрывается другими эффектами в образцах кофе.
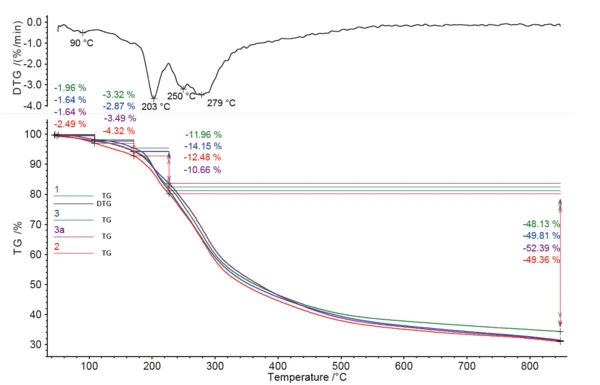
Метод ГХ-МС необходим для разделения выделяющихся соединений и идентификации кофеина в этой сложной смеси. Полученный суммарный ионный ток показывает количество газообразных соединений, обнаруженных для каждого образца; см. таблицу 3.
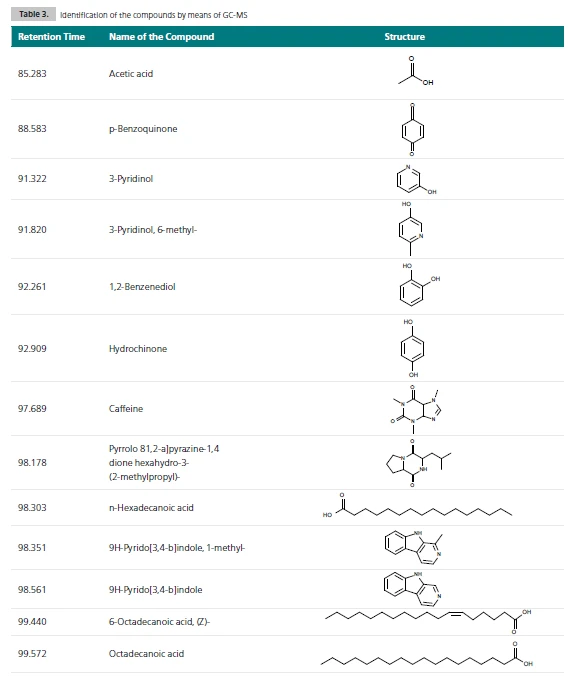
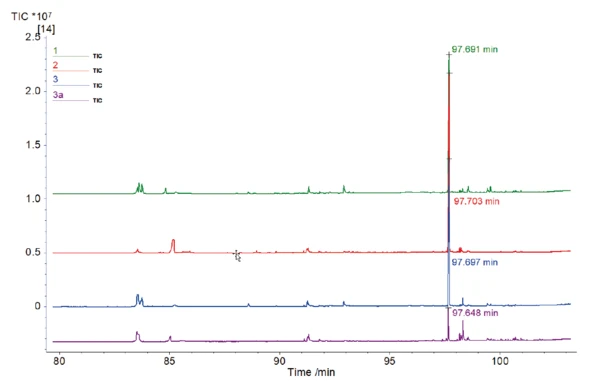
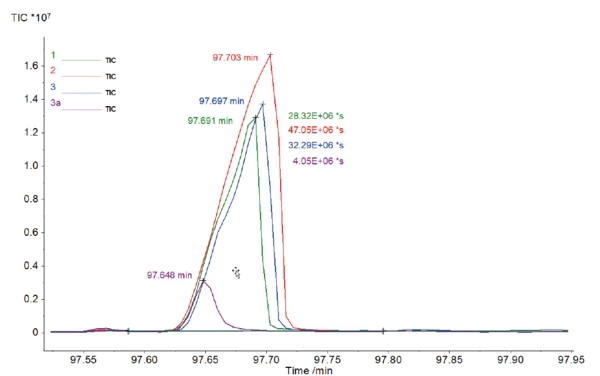
С помощью NIST library основной пик каждой хроматограммы можно отнести к кофеину; см. рисунок 3. Было показано, что высококипящие вещества, такие как кофеин, могут быть перенесены на МС без конденсации.
При более пристальном рассмотрении этого пика (рис. 4) с временем удерживания около 98 мин видны различные размеры и, соответственно, различные площади под этими пиками. Площадь под пиком может быть сравнена относительно и связана с количеством содержащегося кофеина. Среднее значение площади для всех образцов кофе с кофеином составило 35,88-106*с. Результат для образца кофе без кофеина составил 4,05-106*с. При относительном сравнении этих значений можно сделать вывод, что в данном случае образец без кофеина содержит меньше кофеина, чем обычные образцы, примерно в 9 раз.
Количество кофеина на чашку сильно зависит от технологии производства растворимого кофе и количества кофейного порошка, используемого на чашку. Рекомендации по порциям варьируются от 2 до 4 г на чашку.
Резюме
Метод ТГА-ГХ-МС предлагает огромное количество различных вариантов для анализа и сравнения различных пищевых продуктов. Непрерывный сильный нагрев систем переноса позволяет переносить вещества с высокой температурой кипения без конденсации. Помимо идентификации различных выделяющихся органических соединений, стало возможным провести относительное сравнение содержания кофеина. Хотя один из образцов был очищен от кофеина, остаточный кофеин все равно был обнаружен. Это свидетельствует о том, что ГХ-МС является очень чувствительной системой для обнаружения следов выделяющихся газов в диапазоне мкг.