
23.02.2023 by Prof. Dr. Michael Gasik (Aalto University Helsinki, Finland)
DMA on Biomaterials: Seeing the Invisible!
An article by Prof. Dr. Michael Gasik (Aalto University Helsinki, Finland)
Many biomaterial types are presently available for use in different implants, especially in orthopedic and dental cases. Metallic alloys, ceramics and composites are used, be it with or without live cells. There is a growing application field of different scaffolds used in tissue engineering applications to support and promote the formation of new tissues, and many of these are being made by 3D (bio)printing. Biological tissue regeneration is known to be one of the most demanding challenges, requiring biomaterial structures with correct biomechanical properties [1] that mimic in vivo behavior [2]. Proper biomaterials assist the body in rebuilding the damaged tissue and minimize the associated pain and healing time [3].
This article by Prof. Dr. Michael Gasik (Aalto University Helsinki, Finland) shows a new application of the dynamic-mechanical analysis technique (DMA), called BEST (Biomaterials Enhanced Simulation Testing), used for characterizing and improving biomaterials and medical devices; this method goes beyond the known classical viscoelastic analyses.

Prof. Michael Gasik from Aalto University in Finland (Department of Chemical and Metallurgical Engineering) began working in thermal analysis applications in 1985 and has been collaborating with NETZSCH-Gerätebau GmbH for almost that long.
His focus has been on materials for high-temperature applications and for hydrogen technology. Since 2000, he has been actively working with biomaterials, medical devices and regenerative medicine applications. In 2019, he was appointed as the Ambassador of the European Orthopedic Research Society.
Prof. Michael Gasik is a co-founder of Seqvera Ltd. and inventor of the BEST method – Biomaterials Enhanced Simulation Testing – which has been implemented for the first time in NETZSCH DMA equipment.
One focus point of the research activities of Prof. Michael Gasik is the determination of the mechanical properties of biomaterials. In this context, he uses DMA data generated with a NETZSCH DMA 242 Artemis as a basis for further calculations to characterize these materials. Read more about his approach:
Challenges
Numerous studies were already carried out and clinical data was collected regarding the shape, design, and surface state of biomaterials, as well as the geometry of implants and their suitability for the different tissues’ quality and location. Significant differences were also reported for implanted materials that were seemingly identical but originated from different sources [4]. Biomechanical characterization of bone and soft tissues is more problematic than for metallic, ceramic and polymer materials. Published datasets are often not based on comparable measurement protocols and conditions, leading to a lack of consistency. Generalization of these data is very difficult or almost impossible when it comes to providing simple, robust and relevant information.
For biomechanical characterization, one usually reverts to assuming a material as being a type of elastic or viscoelastic matter in order to approximate the properties into individual numbers, usually referred to as “Elastic modulusThe complex modulus (elastic component), storage modulus, or G’, is the “real” part of the samples the overall complex modulus. This elastic component indicates the solid like, or in phase, response of the sample being measurement. elastic modulus”. This, however, fits only with linear elastic materials for very small deformations, and the NPL guidelines [5] list nine methods for calculating the Elastic modulusThe complex modulus (elastic component), storage modulus, or G’, is the “real” part of the samples the overall complex modulus. This elastic component indicates the solid like, or in phase, response of the sample being measurement. elastic modulus which can lead to different values. The vast majority of biomaterials and tissues are clearly not elastic ones, so it is a significant oversimplification to try to artificially reduce data to some fixed numbers: What, for example, would be the benefit of knowing the “Elastic modulusThe complex modulus (elastic component), storage modulus, or G’, is the “real” part of the samples the overall complex modulus. This elastic component indicates the solid like, or in phase, response of the sample being measurement. elastic modulus of mucosa” spanning from 0.1 to 680 MPa by different sources?
Unfortunately, issues related to inertia effects (high frequencies) or instrument limits (low frequencies) are not always sufficiently documented in the test protocols published. Even if instrument inertia is eliminated, the sample itself will always have finite inertia, which can produce artefacts from momentum diffusion, viscoelastic waves, and secondary flows – all of which can violate the assumption of homogeneous and linear deformation [6]. More sophisticated models have a substantial number of artificial fitting parameters, and there are great experimental difficulties in carrying out such tests within existing standards, protocols, and ad hoc test methods [7].
For processes like 3D bioprinting, there are several challenges which must be overcome, such as controlling properties of the bioinks, managing flow and its effect on the cells’ viability, and ensuring optimal biophysical properties of the constructs after printing and upon implantation [8]. Higher resolution and speed with control in the 3D microenvironment are required, and an optimal combination of mechanical and transport properties must be achieved within the space and time scale; these are needed in particular for diffusion-limited vascularization. New Medical Device Regulations (2017/745) demand that proper mechanical evaluation should be carried out, resulting in adherence to the Health Technology Assessment Regulations (2021/2282).
Unfortunately, many different methods of biophysical testing yield rather different outcomes, and it is not easy to obtain realistic, true properties. There are many reasons for the differences ‒ uneven contact, phase state, inertia and elastic instability effects, fitting with improperly assumed models, limitation in StrainStrain describes a deformation of a material, which is loaded mechanically by an external force or stress. Rubber compounds show creep properties, if a static load is applied.strain definition, lack of proper loading history assessment, etc. Hence, it is very important to have a robust approach which can quantify both a biomaterial’s behavior and its performance in the process rather than just generating some specific numbers.
The BEST Concept
To address these challenges, we have developed the patented BEST method (Biomaterials Enhanced Simulation Testing). It can be applied for many hard and soft biomaterials, including hydrogels, 3D-printed constructs, and controlled drug delivery. BEST solutions are targeting problems caused especially by improper and fragmented testing, and are established on an integrated approach based on a fundamental causality principle: “There was no response by the specimen before the stimulus was applied.”
BEST is performed under controlled conditions with required coherent stimuli in the DMA environment. It assesses changes in the specimen’s properties in time, phase and stimulus domains [9]. In post-processing, BEST integrates the data, convolutes the specimen’s history, and extracts unknown values, all without requiring the user to select the model of material (data analysis is essentially model-free). Invariant parameters obtained with a proprietary quantum regression algorithm incorporate the specimen history, showing the position and direction of the development of a biomaterial [10].
The key BEST feature is invariant processing of DMA data, which usually remains unexplored by the user. This new method overcomes common limitations in the linearity of tissue properties in many models, namely a scaling property (homogeneity) and a superposition property (additivity), which are not generally held for Fourier transformation used in linear viscoelasticity.
Hence, BEST applies a correct testing protocol and uses idempotent methods to extract parameters from a single specimen/test, resulting in high output data without the use of complex math (no need in complex moduli) or the assumption of linearity, and is capable of reprocessing also other rheology data in such a way as to not lose their value.
DMA Application Example
In the example shown here, the method described above was deployed based on measurements performed with a NETZSCH DMA 242 Artemis® to characterize the properties of acrylic hydrogel for 3D bioprinting without using an assumed model. The gel was placed into a 1-mL syringe with a 29G needle and set up in the customized DMA sample holder which is usually used for bending; it was tested in stepwise CreepCreep describes a time and temperature dependent plastic deformation under a constant force. When a constant force is applied to a rubber compound, the initial deformation obtained due to the application of the force is not fixed. The deformation will increase with time.creep mode at 25°C.
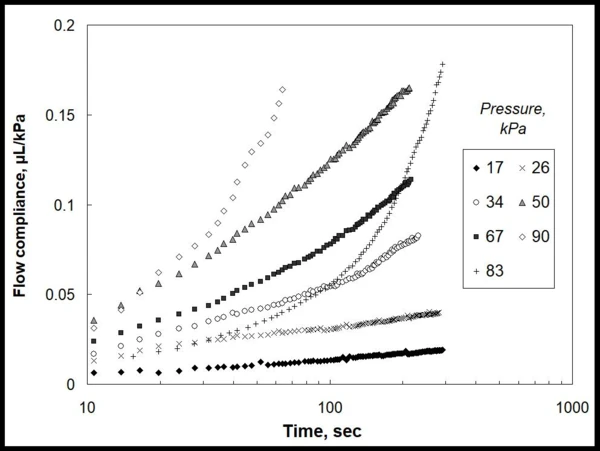
Figure 1 shows the experimental data for the gel amount extruded (µL) via a defined needle nozzle, normalized per applied local pressure (kPa). This data clearly reveals non-linearity of the flow kinetics with time and applied pressure, and there is no straightforward way to select any material model to describe these dependencies.
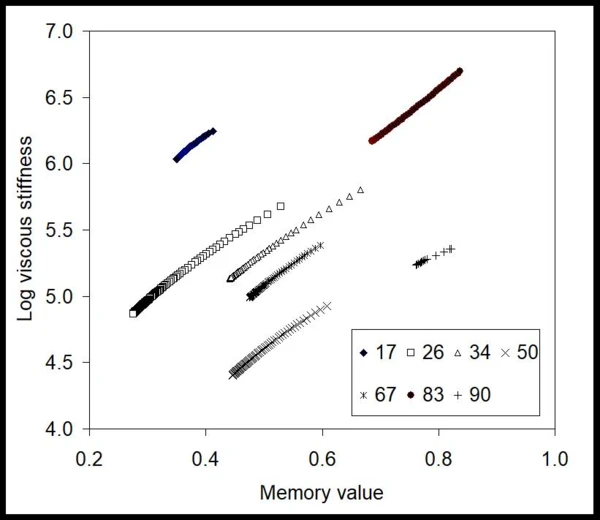
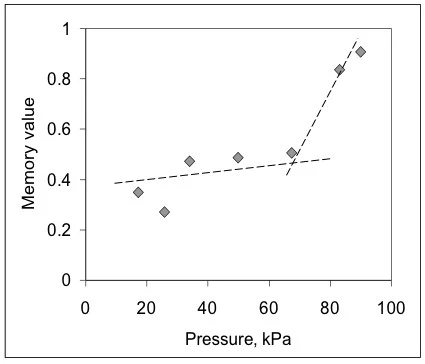
From this data, the BEST method extracted time-invariant values for the viscous stiffness of the gel under these injection conditions as well as its memory value [9,10] (fig. 2). Here, the curves are almost linear, and the slopes of the lines are nearly constant for all applied pressures (numbers in kPa). This means that the gel, despite exhibiting Non-NewtonianA non-Newtonian fluid is one that exhibits a viscosity that varies as a function of the applied shear rate or shear stress.non-Newtonian behavior, is linear in terms of model-free invariant values. It can also be seen that numeric values are changing with the applied pressure in a non-monotonic way, disclosing that there might be different limiting phenomena affecting the flow. In order to see the effect of the flow development, the plot of memory values vs. applied pressure is shown in fig. 3. This map shows that the gel in the syringe faces friction, flow resistance and possibly no-slip effects at low pressures when memory values are much lower than unity. After about 65 kPa – the onset – these values jump up, signaling that the gel achieves a more developed flow.
The presented method can determine invariant values and use them in the model-free prediction of 3D bioprinting processes, depending on the nozzle, geometry, pressure, time and other process conditions, without the need to determine the ink rheological parameters separately. The BEST method generates "first-hand" data for further predictive modeling of the 3D printing process and applies the same philosophy for characterization of the 3D-printed tissues and constructs.
SUMMARY
The developed approach demonstrates the capability to “see invisible” features of the materials and their interactions with the stimuli and environment. This way, dynamic-mechanical analysis (DMA) can provide much more information than the elastic moduli and loss tangent. Using BEST processing, one can obtain many readouts for various purposes (in some cases, even from a single specimen or test). For example, it is possible to obtain the aggregate modulus; characteristic Deborah time; creeping compliance; effective fluid diffusivity and permeability/permittivity; equivalent channel size for the fluid flow in dynamics; material memory value; swelling pressure; and more within a single experiment. And this goes beyond just biomaterials, as the BEST application is model-free and does not require any fitting parameters; further, it can also be applied to test data already created.
Literature:
[1] Hubbell J.A. Nature Biotechnol. 13 (1995) 565–576.
[2] Gasik M. Sci. Techn. Adv. Mater. 18 (2017) 550‐562.
[3] Chung C., Burdick J.A. Adv. Drug Delivery Rev. 60 (2008) 243–262.
[4] Gasik M., Lambert F., Bacevic M., Materials 14 (2021) 2845.
[5] Lord J.D., Morrell R. Measurement Good Practice Guide No. 98; NPL Teddington, UK (2006)
[6] Ewoldt R.H., Johnston M.T., Caretta L.M. In: Complex Fluids in Biological Systems; Springer, Germany (2015).
[7] Vrana N.E., Knopf-Marques H., Barthes J. (Eds.) Biomaterials for Organ and Tissue Regeneration; Woodhead Publ. UK (2020).
[8] Jammalamadaka U., Tappa K. J. Funct. Biomater. 9 (2018) 22
[9] Gasik M., Bilotsky Y. Patent US 10379106 B2 (2019).
[10] Gasik M.Patent US 10809171 B2 (2020).
Contact:
Prof. Dr. Michael Gasik, Dr. Sci.
Terkko Health Hub, Building 14
Helsinki University Central Hospital Area
Haartmaninkatu 4, FIN-00290 Helsinki
www.seqvera.com
Many thanks to Prof. Dr. Michael Gasik for this article and the insights into his research work.







