Paramètres de mesure
TG 209 F1 Libra® - GC-MS
Paramètres TGA :
- RT à 500°C
- 10 K/min
- 50 ml/min N2
- Masse de l'échantillon : 1,71 mg
- Creusets en Al2O3
Paramètres GC :
- Quasi-continu
- Four GC : 250°C
- Fractionnement : 10:1
- Colonne : HP 5 ms, 30 m
- Commutation des vannes : 30 sec
Introduction
L'AAS est l'un des analgésiques non opioïdes les plus largement utilisés. L'estérification du groupe hydroxyle phénolique de l'acide salicylique par l'acide acétique permet non seulement d'obtenir une meilleure tolérance locale, mais aussi de renforcer l'effet antipyrétique, antiphlogistique et surtout d'inhiber l'agrégation plaquettaire [1].
Le nom commun est acide 2-acétoxybenzoïque. Il forme des cristaux blancs en forme d'aiguille qui sentent légèrement l'acide acétique. L'ASA est produit par la synthèse de Kolbe-Schmitt, une substitution aromatique électrophile [2].
[1] Mutschler, Arzneimittelwirkungen
[2] Laue, Reaktionsmechanismen
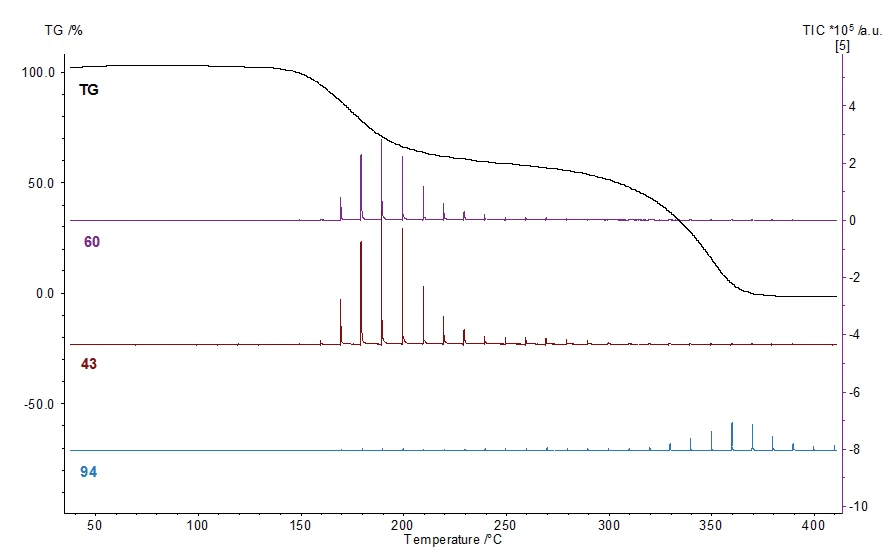
Résultats des mesures
Le couplage simultané de l'analyse thermogravimétrique (TGA) et de la GC-MS (chromatographie en phase gazeuse et spectrométrie de masse) permet d'analyser facilement le comportement thermique et les produits de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition.
La Températures et enthalpies de fusionL'enthalpie de fusion d'une substance, également connue sous le nom de chaleur latente, est une mesure de l'apport d'énergie, généralement de la chaleur, nécessaire pour convertir une substance de l'état solide à l'état liquide. Le point de fusion d'une substance est la température à laquelle elle passe de l'état solide (cristallin) à l'état liquide (fusion isotrope). fusion de l'ASA peut être détectée à l'aide de c-DTA à 142°C. L'ASA se décompose en deux étapes (minima DTG à 175°C et 350°C). Le chromatogramme des ions totaux reflète les spectres entièrement mesurés en fonction de la température. Deux maxima correspondant au DTG sont clairement visibles.
Lapremière étape TG peut être attribuée à la formation d'acide acétique (numéros de masse 43 et 60 figure 4) ; ladeuxième étape peut être attribuée au phénol (numéro de masse 94) en tant que produit de Réaction de décompositionUne réaction de décomposition est une réaction thermiquement induite d'un composé chimique formant des produits solides et/ou gazeux. décomposition.