Introduzione
Finora, la CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione isoterma delle poliolefine non è stata facile da misurare nei DSC a flusso termico a causa della rapidità delle reazioni. Se la temperatura isoterma di CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione non viene raggiunta abbastanza velocemente, il polimero cristallizza durante il raffreddamento. Inoltre, anche un breve abbassamento della temperatura al di sotto del segmento IsotermicoI test a temperatura controllata e costante sono detti isotermici.isotermico programmato indurrebbe involontariamente l'inizio della CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione.libraQuesta combinazione di velocità di raffreddamento e di rapida equiparazione alla temperatura target senza sottotono rende i DSC a compensazione di potenza più adatti dei DSC a flusso di calore per questo tipo di misure.
Grazie alla bassa massa termica del forno Arena® del DSC 214 Polyma, è il primo DSC che combina la robustezza e la maneggevolezza di un DSC a flusso termico con le possibilità di riscaldamento e raffreddamento rapido di un DSC a compensazione di potenza.
Cristallizzazione isotermica di LDPE
Il DSC 214 Polyma è stato utilizzato per effettuare test di CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione isoterma a diverse temperature su LDPE. Sono stati utilizzati parametri di regolazione adeguati per ottimizzare la transizione da un raffreddamento rapido al segmento IsotermicoI test a temperatura controllata e costante sono detti isotermici.isotermico.
Il campione di 3,04 mg è stato riscaldato a 150°C a 20 K/min. Dopo un'isoterma di 2 minuti, il polimero è stato raffreddato a otto diverse temperature comprese tra 101,5°C e 98,5°C, ogni temperatura separata dall'altra di 0,5°C. Il campione è stato quindi mantenuto alla temperatura target fino alla fine della reazione di CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione.cristallizzazione esotermica.
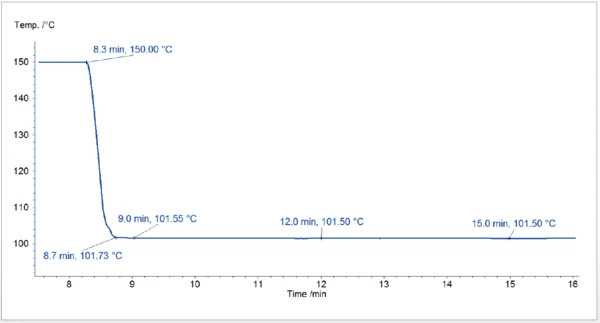
La figura 1 illustra il profilo di temperatura del raffreddamento da 150°C a 101,5°C. Esso mostra che la temperatura target è stata raggiunta rapidamente, senza subire scossoni, e che è rimasta stabile durante l'intero segmento IsotermicoI test a temperatura controllata e costante sono detti isotermici.isotermico.
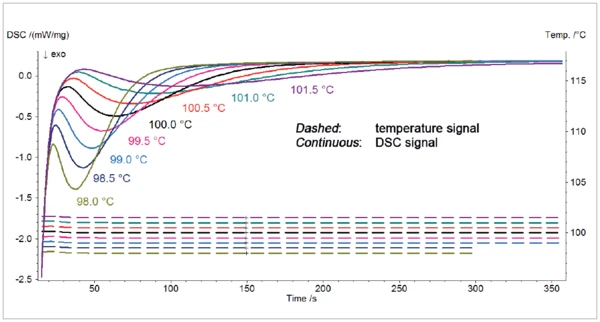
La figura 2 mostra la curva DSC risultante dei segmenti isotermici alle otto temperature dell'isoterma comprese tra 101,5°C e 98,5°C.
Il picco EsotermicoUna transizione di campioni o una reazione è esotermica se viene generato calore.esotermico rilevato durante il segmento IsotermicoI test a temperatura controllata e costante sono detti isotermici.isotermico delle misurazioni deriva dalla CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione. cristallizzazione del polietilene. Come previsto, la reazione si verifica più precocemente al diminuire della temperatura target. La pendenza del picco è maggiore al diminuire della temperatura dell'isoterma. Ciò è dovuto a una velocità di reazione più elevata.
Una differenza di soli 0,5°C tra la temperatura dell'isoterma porta a grandi differenze nelle curve di CristallizzazioneLa cristallizzazione è il processo fisico di indurimento durante la formazione e la crescita dei cristalli. Durante questo processo viene rilasciato il calore di cristallizzazione. cristallizzazione DSC risultanti, indicando una forte influenza della temperatura sulla reazione. Una temperatura inferiore, anche solo di qualche decimo di grado, avvierebbe la reazione in modo involontario. Per questo motivo la temperatura deve essere ben controllata durante il passaggio dal raffreddamento al segmento IsotermicoI test a temperatura controllata e costante sono detti isotermici.isotermico.
Dalle curve DSC alla determinazione dell'energia di attivazione della reazione di cristallizzazione
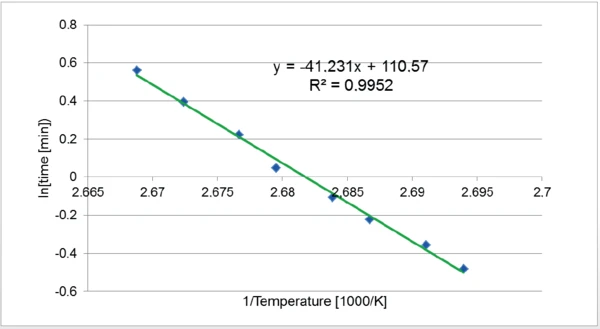
È stato effettuato uno studio cinetico secondo la norma ASTM E2070-13 (metodo di prova - Time-to-Event), dove il tempo trascorso a conversione costante e a temperatura isoterma T e l'energia di attivazione E sono correlati alla seguente equazione:
In[Δ] = E/RT + b, dove R = 8,31 J/(K∙mol)
La pendenza E/R della curva In [Δt]=f(1/T) può essere utilizzata per determinare l'energia di attivazione della reazione.
Il tempo trascorso tra l'inizio dell'isoterma e il picco massimo è stato determinato per ogni temperatura. Ciascun punto è stato tracciato nel grafico In(tempo) in funzione di 1/T. La pendenza della linea di tendenza consente di determinare l'energia di attivazione della reazione. In questo caso, è risultata pari a 434 kJ/mol.