Introduction
Introduction In pharmacy, there is hardly any active ingredient about which more has been written than acetylsalicylic acid (or ASA for short; in English-speaking countries even the brand name Aspirin™ is often used as a synonym). Its success story began at the end of the 19th century when Dr. Felix Hoffmann synthesized the substance at the BAYER laboratories for the first time without impurities. Nowadays, it is still one of the most popular pharmaceuticals used across a broad therapeutic range. It belongs to the group of non-steroidal anti-inflammatory drugs (NSAIDs) and is indicated for the treatment of pain, fever and inflammation. In addition, it is used to prevent recurrence of heart attack or stroke in high-risk patients. In 1977, ASA was added as an analgesic to the “essential drug list” of the WHO (World Health Organization). [1]
This is one of four application notes that examine in more detail the thermal behavior of acetylsalicylic acid: Decomposition in different gas atmospheres, decomposition kinetics, and the resulting gas species. [2, 3, 4]
Results and Discussion
For the investigation of the thermal decomposition of acetylsalicylic acid, thermogravimetric measurements (TGA) were carried out with the NETZSCH STA 449 F3 Jupiter®, coupled to a GC-MS system (Agilent 8890 gas chromatograph and Agilent 5975 MSD). Inert gases such as helium were used as the purge gas atmosphere. Detailed information about the measurement conditions is summarized in table 1.
Table 1: STA measurement parameters
| Parameter | Acetylsalicylic Acid |
|---|---|
| Sample mass | 4.96 mg |
| Atmosphere | Helium |
| Crucible | Al2O3, 85 µl, open |
| Temperature program | RT to 50 °C, 10 K/min |
| Flow rate | 100 ml/min |
| Sample holder | TGA, Type S |
The pyrolysis of acetylsalicylic acid exhibits two mass-loss steps (see figure 1). The first mass loss of 66.4% is related to a peak in the mass-loss rate (DTG) at 170°C. The second mass-loss step amounts to 33.4% with a peak in the DTG curve at 327°C.
In order to provide insight into the pyrolysis products, the TGA-GC-MS coupling was used to separate the complex mixture of gases and to identify the different com-ponents. The measurement parameters for GC-MS are described in table 2.
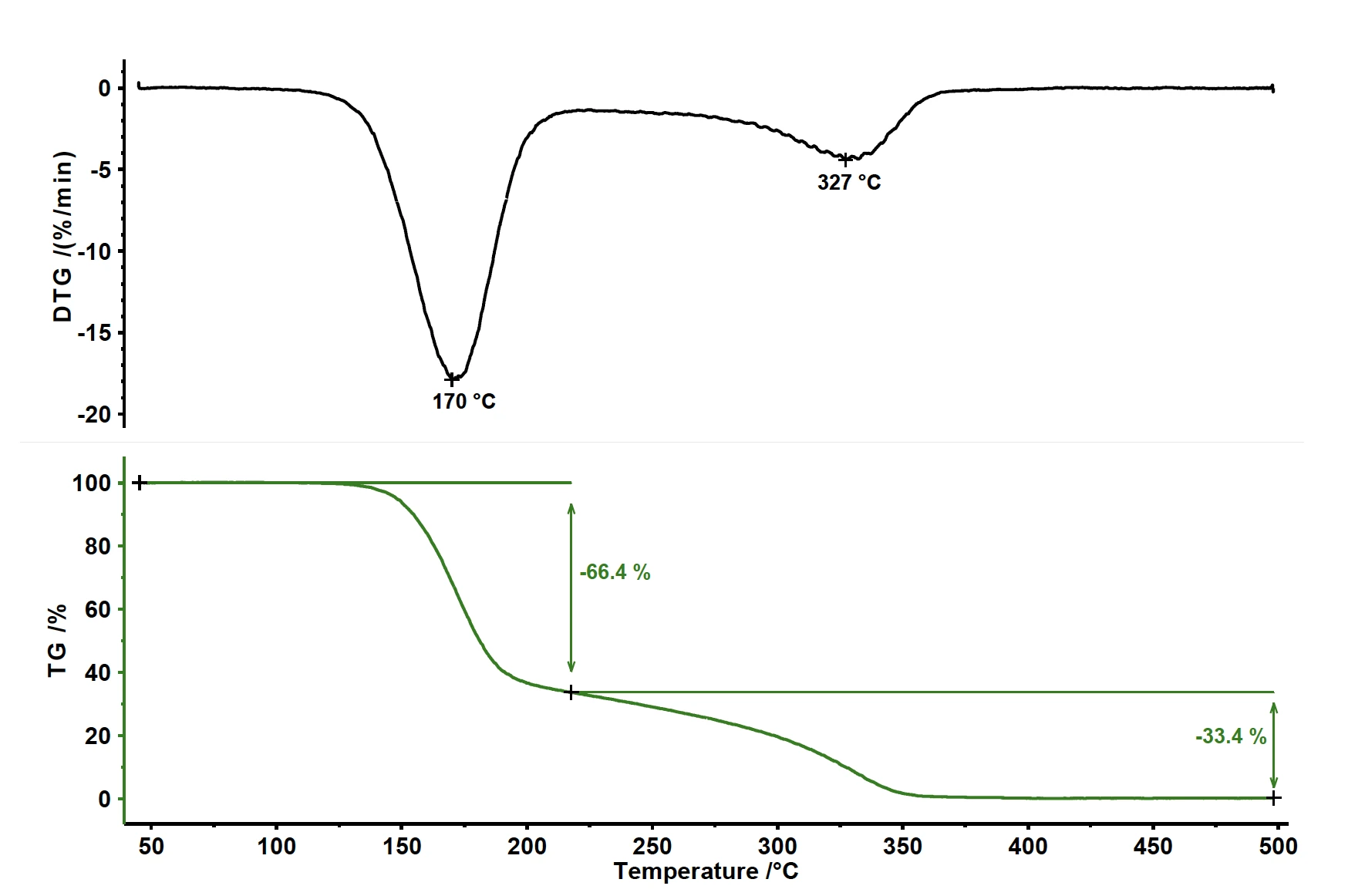
Table 2: GC-MS measurement parameters
| Parameter | Cryo Trapping Mode |
|---|---|
| Column | Agilent HP-5ms |
| Column length | 30 m |
| Column diameter | 0.25 mm |
| Cryo trap | -50°C, 45 min |
| Column diameter | 40°C, IsothermUntersuchungen bei geregelter und konstanter Temperatur werden als isotherm bezeichnet.isotherm, 48 min 40°C to 300°C, 15 K/min |
| Gas atmosphere | He |
| Column flow (split) | 2 ml/min (5:1) |
| Valve | Every 1 min |
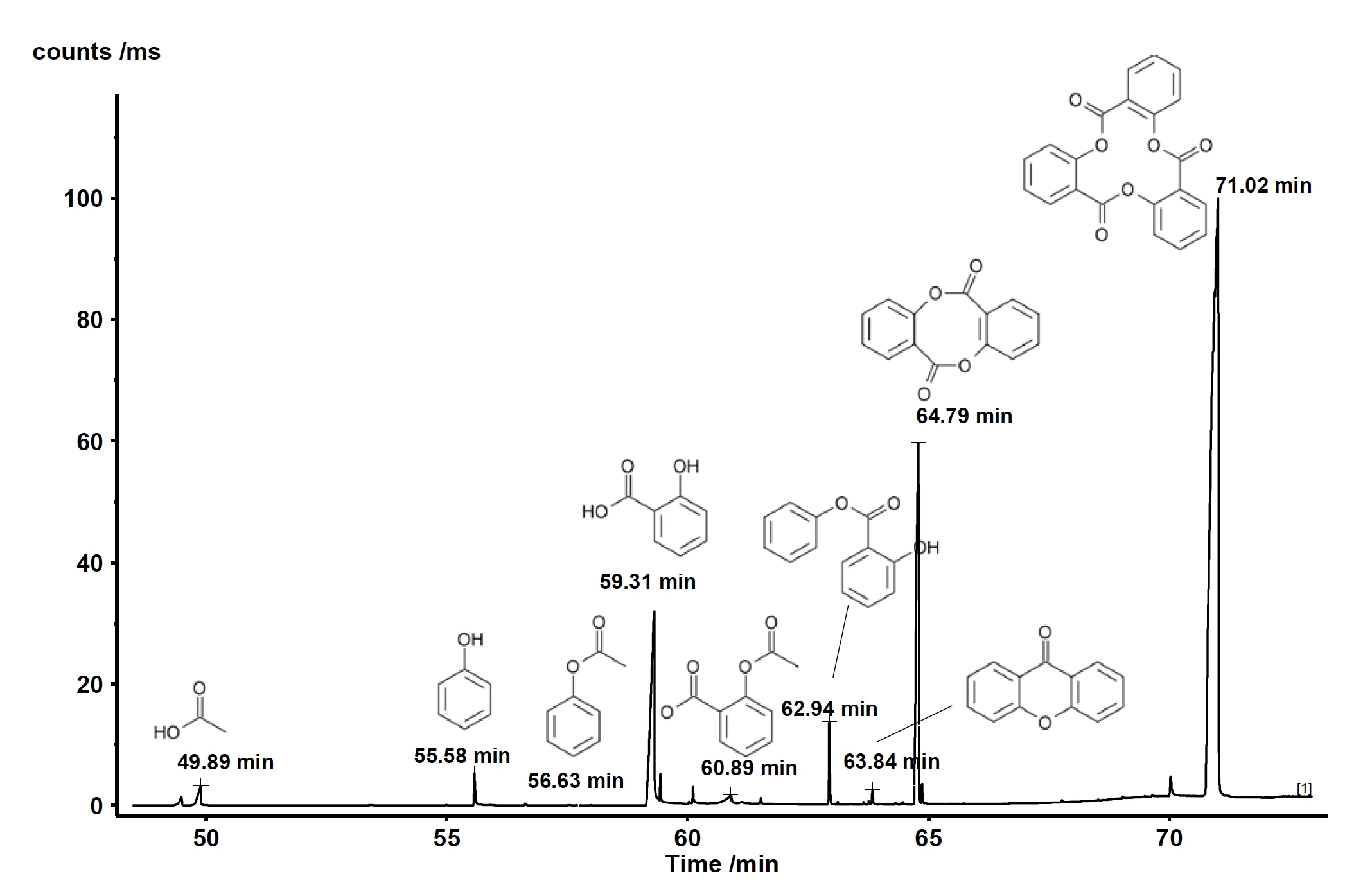
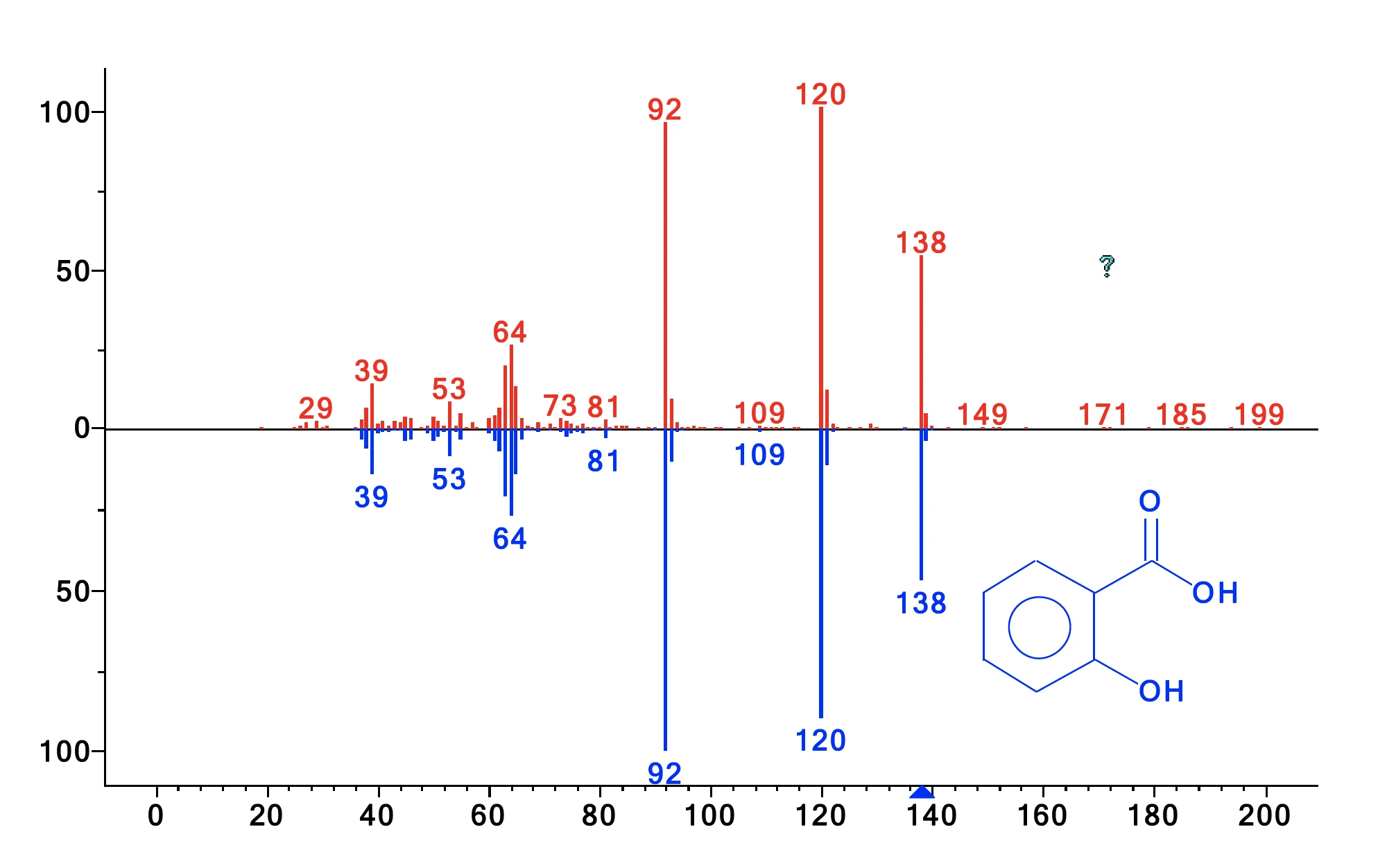
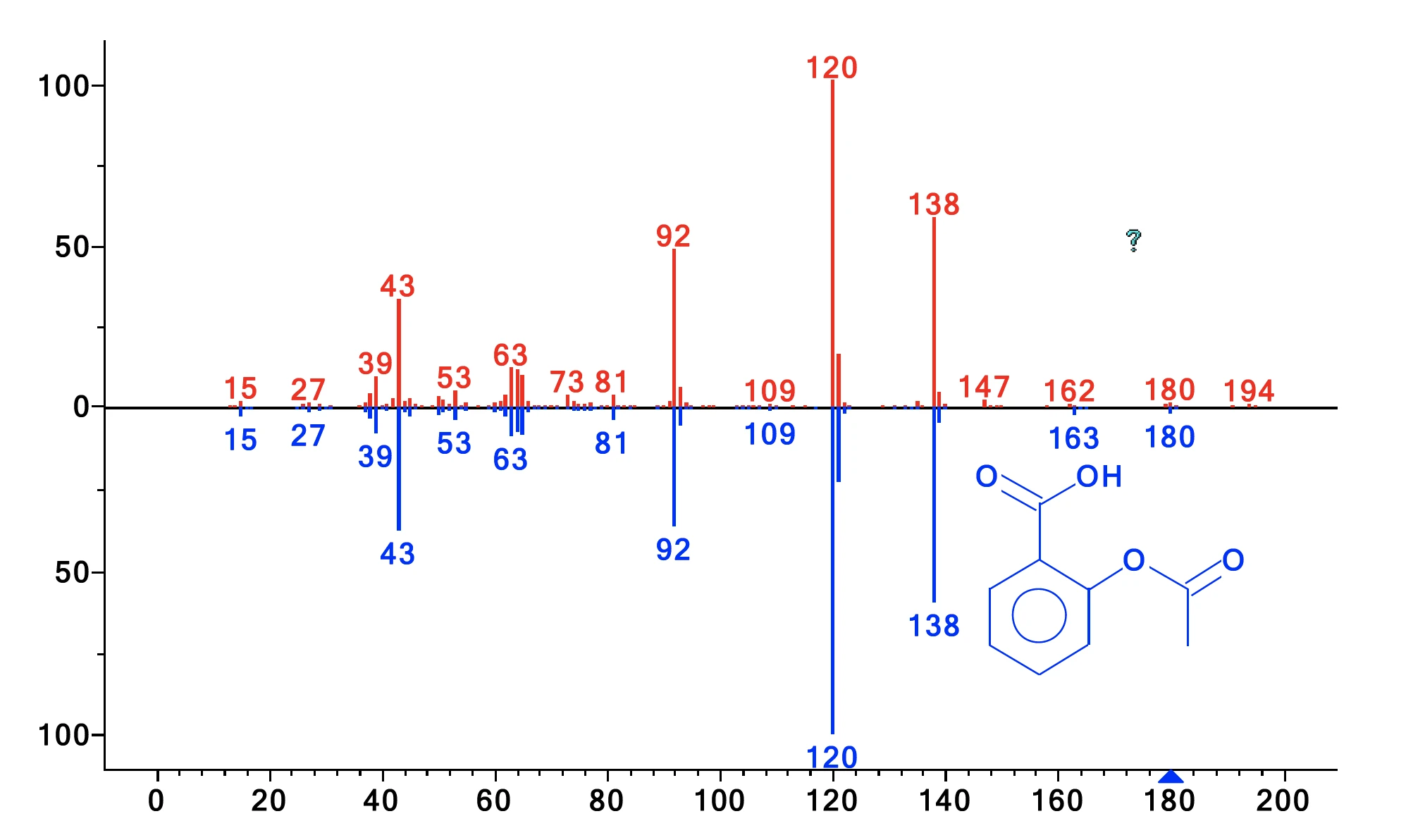
The gases released were sampled every minute on the cryo trap. After the thermogravimetric run, the cryo trap was heated from -50°C to 300°C at a heating rate of 300 K/min to vaporize the condensed compounds and let them separate over the GC column (which was heated at 15 K/min). This method increases the concen-tration of side products and enables excellent separation. The resulting total ion current is displayed in figure 2. Comparison of the detected MS spectra for each peak with the NIST library yields a number of compounds with excellent hit quality. Identification examples are shown for the peaks with a retention time of 59.31 min and 60.89 min in figures 3 and 4. Besides acetic acid, phenol, salicylic acid, and acetylsalicylic acid, cyclic oligomers of 2-hydroxy benzoic acid as stated in literature were also found. This analysis reveals that decomposition and evaporation take place simultaneously and furthermore explains why the two mass-loss steps are not separated.
Library Search
| Retention time [min] | Name | Hit quality |
| 49.89 | Acetic acid | 91 |
| 55.58 | Phenol | 96 |
| 56.63 | Acetic acid phenyl ester | 90 |
| 59.31 | 2-Hydroxybenzoic acid (= salicylic acid) | 97 |
| 60.89 | Acetylsalicylic acid | 81 |
| 62.94 | Phenyl salicylate | 95 |
| 63.84 | Xanthone | 97 |
| 64.79 | 6H,12H-Dibenzo[b,f][1,5]dioxocin-6,12-dione (Dimer of 2-Hydroxybenzoic acid) | 64 |
| 71.02 | 2,10,18-Trioxatetracyclo[18.4.0.0(4,9).0(12,17)] tetracosa-1(24),4,6,8,12,14,16,20,22-nonaene-3,11, 19-trione (Trimer of 2-Hydroxybenzoic acid) | 90 |
Conclusion
The combination of thermogravimetry and GC-MS (gas chromatography / mass spectrometry) is a powerful tech-nique for gaining deep insights into thermal decompo-sition processes and the resulting gases released. The thermal decomposition of acetylsalicylic acid in a helium atmosphere results in a complex gas mixture of at least nine different compounds released. Previous studies by TGA-FT-IR (Fourier transform infrared spectroscopy cou-pled to a thermobalance) showed that the first mass-loss step releases acetic acid and salicylic acid, whereas the second mass-loss step is the result of a complex decompo-sition reaction. The capability of GC-MS starts where FT-IR reaches its limitations and provides much deeper insights into mixtures of simultaneously released gases. TGA-GC-MS is capable to both separate and identify them.