Introduzione
Le vernici, gli adesivi, gli inchiostri da stampa e i composti per l'invasatura sono sempre più spesso polimerizzati a temperature moderate (spesso a temperatura ambiente) mediante radiazioni ultraviolette (UV). Oltre all'aspetto del risparmio energetico - rispetto alla Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione termica - l'elevata velocità di lavorazione della reticolazione indotta dagli UV e l'ecocompatibilità dei sistemi reattivi UV sono di grande interesse per le applicazioni industriali. Poiché l'apporto di energia è breve, gli oggetti rivestiti in questo modo non vengono quasi riscaldati. Per questo motivo, questa tecnica può essere utilizzata anche per il trattamento superficiale di substrati sensibili al calore, come pellicole di plastica, legno e carta. Inoltre, le pellicole di vernice polimerizzate con raggi UV presentano generalmente un'elevata resistenza ai graffi e agli agenti chimici.
Per realizzare i suddetti vantaggi di questo metodo e generare prodotti di alta qualità, è necessario ottimizzare le formulazioni di Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione UV e determinare i tempi di irradiazione e le intensità di radiazione ottimali. I fotocalorimetri, talvolta denominati anche Photo-DSC o UV-DSC, sono ideali per lo studio delle sostanze attive alla luce e del loro comportamento di Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione.
La polimerizzazione UV è molto rapida
La Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione UV è generalmente completa in pochi secondi. I meccanismi di reazione coinvolgono tipicamente le polimerizzazioni cationic o radicali, cioè la reticolazione innescata da un iniziatore che si decompone sotto l'influenza della luce ultravioletta, provocando una reazione a catena Ionic o radicale.
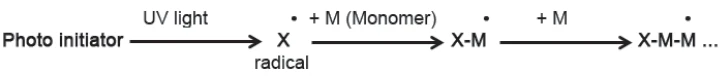
I principi di base di entrambi i tipi di reazione sono simili [1]. La maggior parte dei rivestimenti UV impiega la Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione radicalica (vedi schema in figura 1). I radicali formati durante la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione del fotoiniziatore reagiscono, ad esempio, con i doppi legami dei monomeri, generando nuovi radicali che sostengono la Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione. Con il progredire della Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione, il materiale diventa più viscoso, limitando la capacità dei radicali e dei doppi legami di diffondersi insieme, in modo che la velocità di reazione diminuisca.
Un vantaggio della Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione cationic rispetto alla Polimerizzazione (reazioni di reticolazione)Tradotto letteralmente, il termine "crosslinking" significa "reticolo incrociato". Nel contesto chimico, viene utilizzato per le reazioni in cui le molecole vengono collegate tra loro introducendo legami covalenti e formando reti tridimensionali. polimerizzazione radicale è che le polimerizzazioni cationic sono meno sensibili all'influenza dell'ossigeno.
Impostazione e modalità operativa dell'UV-DSC basato su DSC 204 F1 Phoenix®
La calorimetria differenziale a scansione (abbreviata in DSC) è un metodo termoanalitico in cui viene determinata quantitativamente la differenza di flusso di calore tra un campione e un riferimento, sottoposti a un programma di temperatura controllata (definizione basata su DIN 51 007, ISO 11357 - 1 o ASTM E 472).
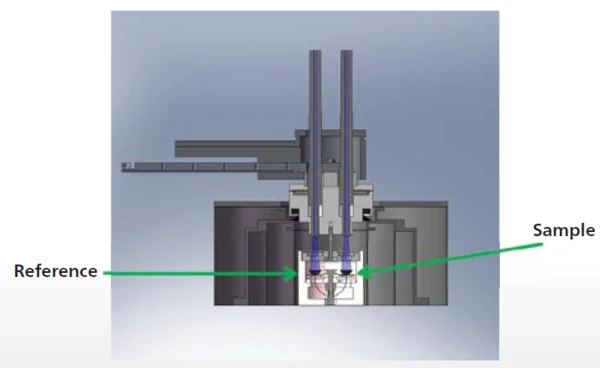
La figura 2 mostra il calorimetro a flusso di calore NETZSCH DSC 204 F1 Phoenix® (vedere anche la configurazione schematica con l'allegato UV [2], figura 3). Sia il campione che il riferimento sono collocati in un forno e vengono irradiati simultaneamente (in blu). La fibra ottica è saldamente installata nel coperchio in modo da garantire distanze riproducibili tra la fibra ottica e il campione e il riferimento. Il software di misura DSC comunica con la lampada UV, attivando gli impulsi e controllando automaticamente la lunghezza e l'intensità degli impulsi.
Durante la misurazione, i segnali rilevati sono la temperatura del campione e la differenza di flusso di calore. Integrando il segnale del flusso di calore, è possibile determinare il calore di polimerizzazione, fornendo dati significativi per lo sviluppo o l'ottimizzazione del processo.

Ottimizzazione del tempo di esposizione e del grado di polimerizzazione tramite Mezzi di UV-DSC
Durante il processo di sviluppo di adesivi, inchiostri, ecc. è importante trovare il tempo di esposizione ottimale, cioè il tempo di esposizione necessario per raggiungere il grado di polimerizzazione desiderato e, di conseguenza, le proprietà del materiale desiderate. Il grado di polimerizzazione è di primario interesse per i test in corso di lavorazione e per il controllo di qualità.
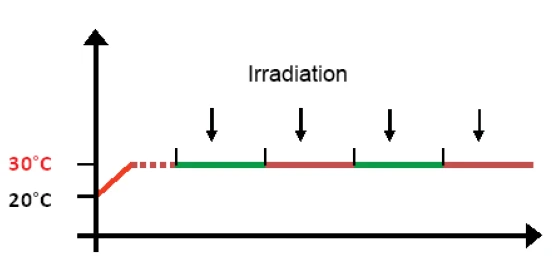
libraIn una misura UV-DSC standard, il campione viene inizialmente riscaldato alla temperatura di reazione desiderata (30°C nella figura 4) e, dopo una breve fase di equalizzazione della temperatura, viene avviata l'irradiazione. In genere si programmano più segmenti isotermici, ciascuno comprendente un singolo impulso di lampada, poiché più impulsi di lunghezza e intensità definite consentono di monitorare la polimerizzazione del campione fino al completamento. La lampada UV viene solitamente attivata pochi secondi dopo l'inizio di ogni segmento.
La Figura 5 mostra i risultati di due indagini (presentate in rosso e in blu) su un rivestimento a base di acrilato disponibile in commercio con diversi tempi di irraggiamento (0,5 s e 1 s). Come previsto, in entrambi i casi, la maggior parte della reazione esotermica avviene durante la prima fase di irraggiamento; le entalpie di reazione sono leggermente diverse per i diversi tempi di irraggiamento, tuttavia con l'impulso più lungo di 1 s che porta a un'entalpia leggermente più alta di -283,4 J/g rispetto a -236,4 J/g per l'impulso di 0,5 s. Questa differenza viene quasi compensata nei segmenti di irraggiamento successivi. Ciò significa che, a intensità di irraggiamento costante, un tempo di irraggiamento più elevato (curva blu) nel primo segmento comporta un grado di polimerizzazione parziale più elevato e smaller la post-cura nei segmenti successivi. La figura 6 mostra una rappresentazione grafica ancora più chiara dei dati.
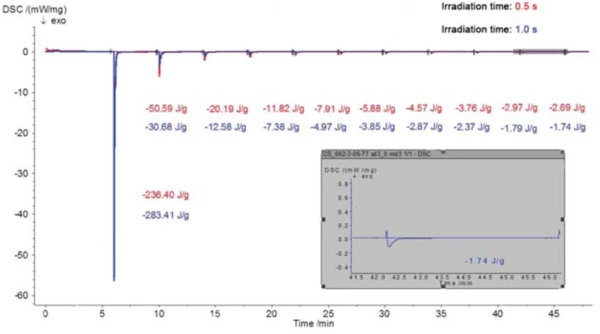
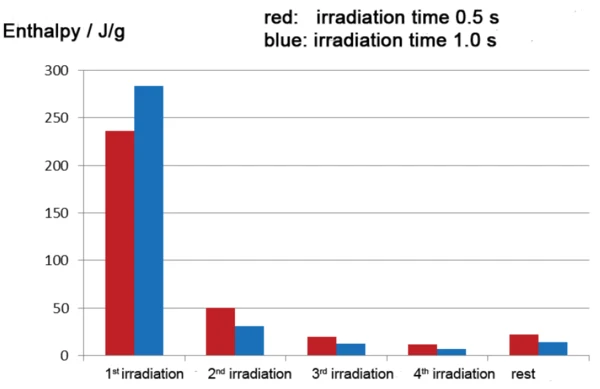
A partire da circa la decima fase di irraggiamento, le aree di picco nella misurazione DSC associate a ciascun impulso cambiano appena. L'area di picco residua costante una volta completata la polimerizzazione è dovuta al riscaldamento diff erenziale dei campioni rispetto al riferimento da parte dell'irraggiamento. Il calcolo dell'entalpia totale del processo di polimerizzazione richiede che questa entalpia residua venga sottratta dal contributo entalpico di ciascun picco incluso nel calcolo.
Se l'entalpia della prima fase di irradiazione viene rapportata all'entalpia totale, si calcola un grado di polimerizzazione di circa l'82% per il primo impulso di 1 s e un grado di polimerizzazione di circa il 67% per il primo impulso di 0,5 s. A seconda del grado di polimerizzazione desiderato per l'uso pratico, potrebbe essere sufficiente una singola fase di irradiazione della durata di 1 secondo, assumendo che lo spessore del campione di processo sia paragonabile a quello del campione DSC.
L'ossigeno come inibitore dei sistemi acrilici
Per il processo di reazione di molti sistemi di verniciatura fotopolimerizzati, l'ossigeno gassoso svolge un ruolo decisivo. Per i sistemi acrilici, l'ossigeno agisce come inibitore. Il suo meccanismo d'azione era già stato descritto da G.V. Schulz e G. Henrici [3] negli anni Cinquanta. In presenza di ossigeno, si formano radicali perossidici che portano all'incorporazione dell'ossigeno nel polimero. Ciò si traduce in catene copolimeriche relativamente corte [4].
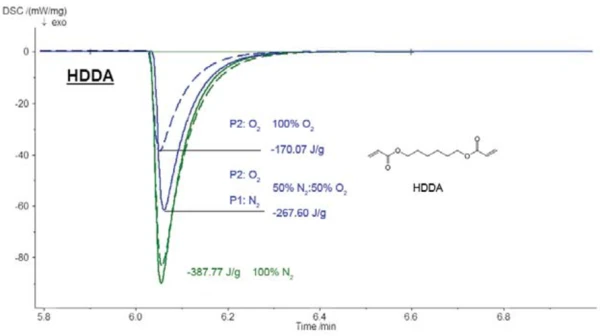
La Figura 7 mostra l'influenza dell'ossigeno sulla fotopolimerizzazione del diacrilato di esandiolo (HDDA). L'entalpia di reazione diminuisce sensibilmente con l'aumento della concentrazione di ossigeno.
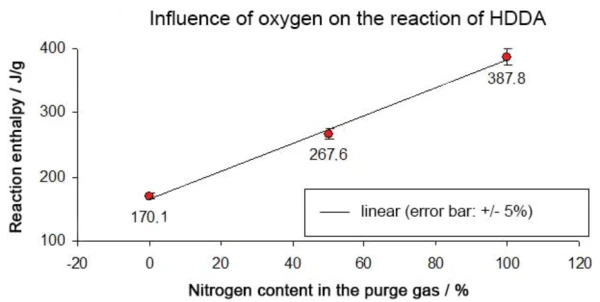
L'entalpia di reazione in un'atmosfera di azoto puro era di -388 J/g, rispetto a -268 J/g in una miscela di 50% azoto e 50% ossigeno e -170 J/g in un'atmosfera di ossigeno puro. Ne risulta una correlazione lineare tra l'entalpia di reazione e il contenuto di ossigeno (vedi figura 8).
Conclusione
Il DSC 204 NETZSCH F1 Phoenix® con accessori per lampade UV è facile da usare. La struttura a tenuta di gas consente un controllo preciso della composizione atmosferica nella camera di campionamento; ciò è di fondamentale importanza per quanto riguarda il contenuto di ossigeno residuo nel gas di lavaggio. La lampada UV è controllabile con il software di misura DSC. Parametri come il tempo di irradiazione e l'intensità possono quindi essere preselectnel programma di misura DSC. Per un numero elevato di misurazioni, è possibile utilizzare il sistema di cambio automatico dei campioni (ASC) in combinazione con l'attacco UV.
La calorimetria differenziale a scansione (DSC) in combinazione con l'irradiazione del campione con una lampada UV è ideale per la caratterizzazione semplice e rapida dei processi di polimerizzazione foto-iniziati. I risultati di queste misurazioni offrono una visione dei meccanismi di polimerizzazione e forniscono informazioni importanti per il miglioramento delle formulazioni (inibitori, fotoiniziatori, riempitivi) e per il controllo del processo.
Questo articolo è stato pubblicato nell'edizione di giugno 2013 di Laborpraxis (con un numero ridotto di figure).