Introduzione
Dalla sua scoperta nel 1965 da parte del chimico James L. Schlatter, gli scienziati litigano su questo controverso dolcificante. È giustamente consentito nelle bevande e in altri prodotti dietetici o è un pericolo per la nostra salute?
In questo caso, è stato misurato mediante DSC e TGA-FT-IR per ottenere informazioni su alcune delle sue proprietà termiche, come il Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa).punto di fusione e la temperatura di degradazione.
Risultati del test
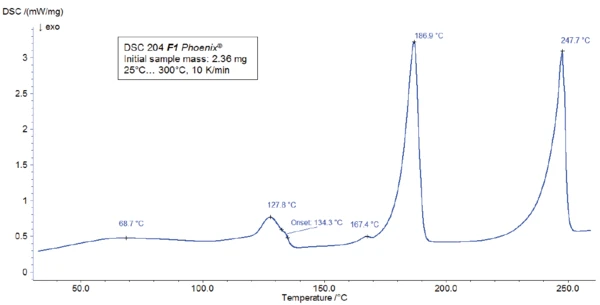
Per la misurazione DSC, un campione è stato preparato in un crogiolo Concavus® con coperchio forato e riscaldato tra la temperatura ambiente e 300°C a una velocità di riscaldamento di 10 K/min. I risultati del riscaldamento sono presentati nella Figura 2.
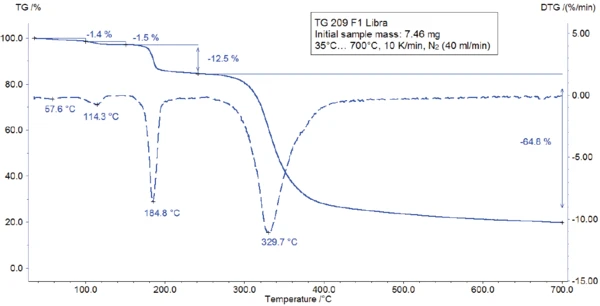
La misura TGA-FT-IR è stata eseguita su un campione di 7,46 mg preparato in un crogiolo di ossido di alluminio e riscaldato a 700°C a 10 K/min in un'atmosfera dinamica di azoto. I gas emessi durante la misurazione TGA sono stati iniettati direttamente nello spettrometro FT-IR di Bruker Optics. La curva TGA è riportata in figura 3. Un ampio effetto EndotermicoUna transizione campionaria o una reazione è endotermica se per la conversione è necessario il calore.endotermico tra 25°C e 100°C (figura 3) è associato a una perdita di massa dell'1,4%. Un secondo effetto EndotermicoUna transizione campionaria o una reazione è endotermica se per la conversione è necessario il calore.endotermico rilevato a 128°C (temperatura di picco) porta a una perdita di massa dell'1,5%. I corrispondenti spettri FT-IR a 60°C e 123°C (vedi figura 4) rivelano che la sostanza rilasciata è acqua in entrambi i casi (molto probabilmente acqua assorbita nella prima fase e acqua di idratazione nella seconda fase).
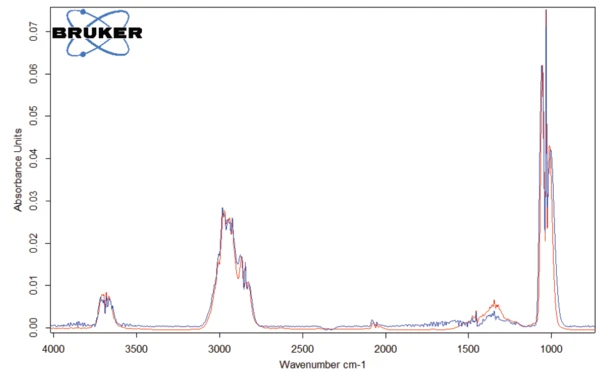
Il picco rilevato a 187°C (DSC), corrispondente alla fase TGA con una perdita di massa del 12,5%, è dovuto alla degradazione dell'aspartame. Lo spettro FT-IR rilevato a 184°C è riportato nella figura 5 (curva blu). Corrisponde molto bene allo spettro del PNNL library per il metanolo (curva rossa).
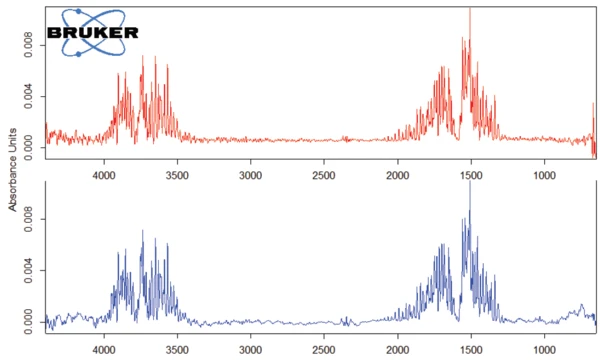
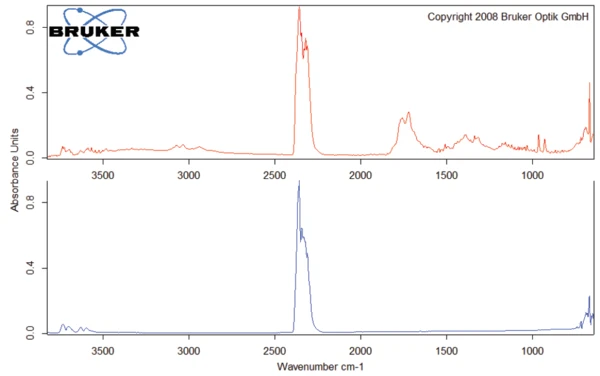
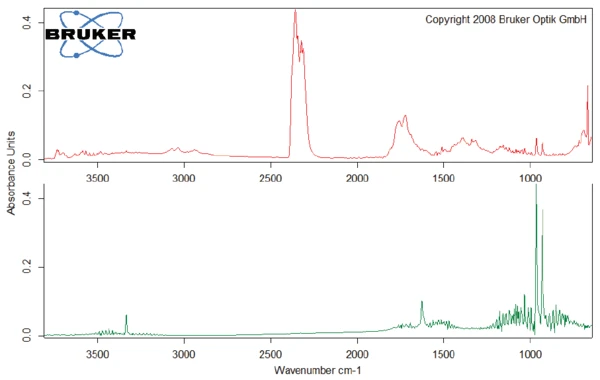
Questa degradazione termica dell'aspartame, associata al rilascio di metanolo, porta alla formazione di una nuova sostanza, presumibilmente la 2,5-diossopiperazina [2]. Il picco a 248°C nella curva DSC può essere attribuito alla Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione della sostanza formata. Segue la degradazione di questo prodotto (picco TGA a 330°C - fig. 3). Le figure 6, 7 e 8 mostrano ciascuna lo spettro FT-IR dei prodotti rilasciati a 329°C (curva rossa) in confronto con gli spettri FT-IR di diversi composti, suggeriti dal database NIST-EPA. Durante la degradazione, vengono rilasciati anidride carbonica e ammoniaca (spettro blu in figura 6; spettro verde in figura 7). Le altre bande rilevate sono probabilmente causate da gruppi funzionali contenenti legami aromatici, azoto e ossigeno.
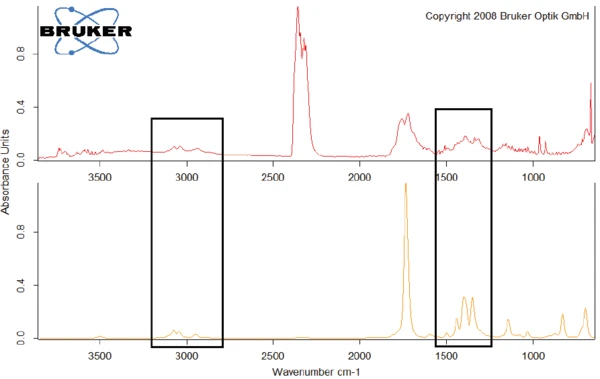
A titolo di esempio, la figura 8 confronta lo spettro FT-IR a 329,1°C con lo spettro dell'N-benzil-maleimmide, con il quale mostra conformità negli intervalli di lunghezza d'onda intorno a 3000 cm-1 e tra 1250 cm-1 e 1500 cm-1.
Conclusione
L'analisi con i metodi complementari DSC e TGA rivela la quantità di componenti volatili in un campione e le sue temperature di Temperature di fusione ed entalpieL'entalpia di fusione di una sostanza, nota anche come calore latente, è una misura dell'apporto di energia, tipicamente calore, necessario per convertire una sostanza dallo stato solido a quello liquido. Il punto di fusione di una sostanza è la temperatura alla quale essa cambia stato da solido (cristallino) a liquido (fusione isotropa). fusione e degradazione. Inoltre, l'accoppiamento FT-IR fornisce informazioni sulle sostanze rilasciate durante il riscaldamento: Nel caso dell'aspartame, l'acqua evapora per prima e la sostanza si degrada successivamente rilasciando metanolo.
Sebbene sia causata da un altro tipo di meccanismo, la degradazione dell'aspartame nel corpo dopo il suo consumo porta anche al rilascio di metanolo, tra le altre emissioni. Questo può portare a mal di testa e vertigini [3], se la sostanza viene assunta in quantità elevate. Questo è uno dei motivi per cui si raccomanda di consumare l'aspartame solo in modo sconsigliato.