過酸化水素
純粋な過酸化水素(H2O2)は淡青色の液体で、水と任意の割合で混合することができる。低濃度の水溶液は、その強い酸化作用から漂白剤として広く使用されている。過酸化水素水溶液は、木材、紙、毛髪の漂白以外にも、酸化剤として、あるいは医療用殺菌剤として使用される。過酸化水素は水と酸素に分解する性質があるため(下記式1参照)、ロケットエンジンの液体推進剤として使用されている。
(式1)
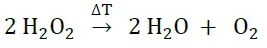
マルチモジュール熱量計(MMC)
NETZSCH マルチモジュール熱量計 マルチモジュール熱量計(MMC)ベースユニットと交換可能なモジュールからなるマルチモード熱量計。1つのモジュールは暴走反応熱量測定用(ARC )で、ARC-モジュールです。2つ目のモジュールは走査試験に使用され(走査モジュール)、3つ目と4つ目のモジュールは電池やポリマー、コインセル(コインセルモジュール)の薬学的試験に関連しています。MMC 274Nexus (図1)には、3つの異なる測定モジュールが用意されています[1]。ARC® 。コインセルモジュールは電池の調査に特化しており、走査モジュールは1回の加熱で得られた熱量データを評価するために使用できます。示差走査熱量測定(DSC)とは対照的に、マルチモジュール熱量計(MMC)ベースユニットと交換可能なモジュールからなるマルチモード熱量計。1つのモジュールは暴走反応熱量測定用(ARC )で、ARC-モジュールです。2つ目のモジュールは走査試験に使用され(走査モジュール)、3つ目と4つ目のモジュールは電池やポリマー、コインセル(コインセルモジュール)の薬学的試験に関連しています。MMCの走査モジュールは2mlまでの試料を扱うことができます。試料の加熱には、一定の加熱速度または一定の電力レベルの2つのオプションがあります。試料に供給される電力と加熱速度の両方の情報を使用することで、ヒートフロー信号を計算することができます。インジウム、スズ、ビスマスなどの金属を使用すると、装置の温度と感度の両方を決定することができます。一般的な試料質量は1000~9000mg(試料量約1ml)であり、DSCに使用される試料質量(一般的に5~10mg)よりも、マルチモジュール熱量計(MMC)ベースユニットと交換可能なモジュールからなるマルチモード熱量計。1つのモジュールは暴走反応熱量測定用(ARC )で、ARC-モジュールです。2つ目のモジュールは走査試験に使用され(走査モジュール)、3つ目と4つ目のモジュールは電池やポリマー、コインセル(コインセルモジュール)の薬学的試験に関連しています。MMCの方がかなり高くなります。それでも、マルチモジュール熱量計(MMC)ベースユニットと交換可能なモジュールからなるマルチモード熱量計。1つのモジュールは暴走反応熱量測定用(ARC )で、ARC-モジュールです。2つ目のモジュールは走査試験に使用され(走査モジュール)、3つ目と4つ目のモジュールは電池やポリマー、コインセル(コインセルモジュール)の薬学的試験に関連しています。MMCの走査モジュールの不確かさは、温度測定で約1%、エンタルピー測定で5%未満です。
この研究では、走査モジュール(図2参照)とARC® モジュール(図3参照)の2つのマルチモジュール熱量計(MMC)ベースユニットと交換可能なモジュールからなるマルチモード熱量計。1つのモジュールは暴走反応熱量測定用(ARC )で、ARC-モジュールです。2つ目のモジュールは走査試験に使用され(走査モジュール)、3つ目と4つ目のモジュールは電池やポリマー、コインセル(コインセルモジュール)の薬学的試験に関連しています。MMCモジュールを使用して、過酸化水素(35%)の熱分解挙動を研究しています。試料容器を直接囲む外部ヒーター(図4)を介して、走査モジュールは試料に一定レベルの電力を供給することができる。


測定条件
測定条件を表1にまとめた。過酸化水素(Sigma Aldrich)は水溶液(35%)として受領し、常温で保存した。
表1:測定条件
MMC 274Nexus | ||
|---|---|---|
| MMCモジュール | スキャン | ARC® |
| 容器材質 | ステンレス鋼 | ステンレス鋼 |
| 容器タイプ | 密閉式 | 密閉式 |
| 容器質量 | 7176.38 mg | 7233.59 mg |
| 加熱 | 定電力 (250 mW) | Heat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。HWS |
| 雰囲気 | 空気 | 空気 |
| パージガスレート | 静的 | 静的 |
| 温度範囲 | 室温...250 | 室温...250 |
| 試料質量 | 1031.1 mg | 1008.1 mg |
H2O2、H2Oおよび空の容器の挙動の比較
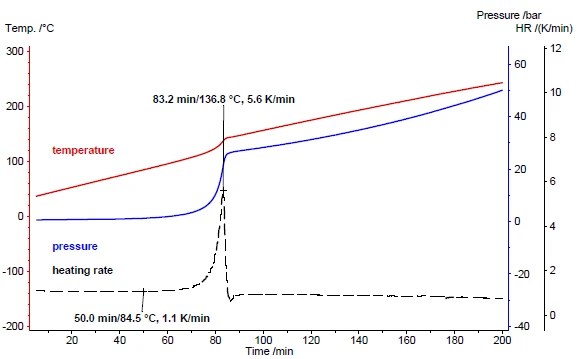
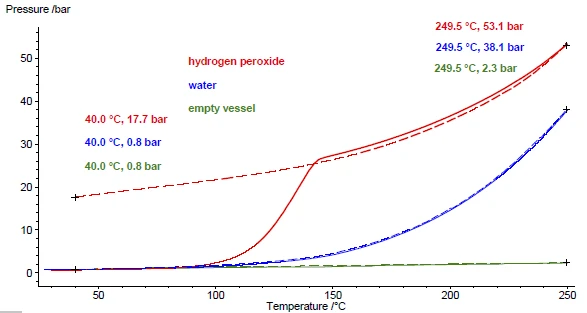
図5の結果は、もっぱら試料を加熱したものである。過酸化水素の分解反応は可逆的ではないため、発生した酸素は冷却中に再び取り込まれて最初の過酸化水素を形成することはない。その代わり、生成した水と酸素は、それぞれ液体と気体として周囲温度まで冷却される。圧力信号は40℃で17.7バールを示し、これは分解中に生成される酸素の量を反映している(図6)。代わりに同じ量の水を例にとると、圧力は加熱中に上昇するが、水は化学的に変化しないため、冷却中にすべての水蒸気が再び析出する。そのため、冷却時の水の圧力信号を示す青の破線は、加熱時(実線)とほとんど同じ値を示している。比較のために、緑色の線は空の容器の加熱時と冷却時の圧力信号の経過を示している。
走査モジュールの利点
MMC走査モジュールで得られたこれらの結果は、加熱速度の不連続な経過と圧力上昇が、分解反応や発熱(発熱性)反応の危険性を示す優れた指標であることを明確に示している。250 mWのようなsmall 出力レベルでも、約1 K/分の比較的にsmall 加熱速度となり、この例示的な測定の加熱には4時間未満しかかかりません。したがって、MMC走査モジュールはスクリーニングツールとして使用するのに適している。圧力や温度の上昇が検出された場合、次のステップとして断熱試験を行う必要があります。
暴走反応熱量測定
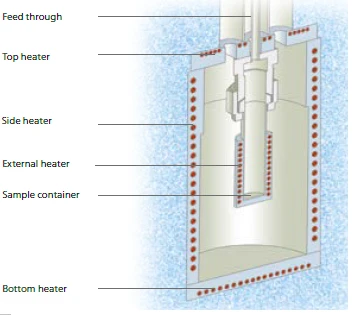
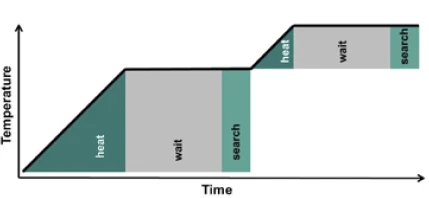
専用の熱量計により、暴走反応熱量測定法(ARC®)に従った試料調査が可能です。ARC®-タイプの装置は、熱交換を許さず、わずかな自己発熱反応も検出できるよう、断熱試料環境を提供する。一般的な測定モードは、ヒートウェイトサーチ(Heat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。HWS)と呼ばれる。加熱、平衡化、そしてわずかな自己誘導温度変化の検出という一連の流れは、分解反応の開始温度を決定するために使用される準等温アプローチである。図7にHeat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。heat-wait-search 。
MMCのARC® モジュールのセットアップの断面を図3に示す。検出期間(サーチ)中に自己発熱率0.02 K/minを超えると、測定はHeat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。heat-wait-search から断熱モードに変わる。これは、周囲のヒーター(上部、側部、下部)が上述のシーケンスに従わなくなり、むしろ試料温度に追従することを意味します。この "断熱 "モードでは、温度差がないため、試料と熱量計の環境との間で熱交換が行われません。
ARC® モジュール内のH2O2
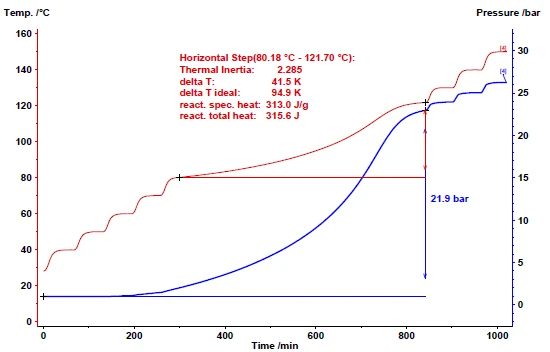
図8は、過酸化水素(35%)の分解を、Heat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。heat-wait-search モードを採用したMMCのARC® モジュールで調べた結果を示している。段階的加熱の温度増分は10 Kで、システムは待機中に30分間安定させた。10分間の探索期間中に発熱(発熱性)が検出されるかどうかの答えは、発熱開始温度(発熱しきい値)に依存する。40℃から70℃の間では、サーチ期間中の自己発熱は0.02 K/分未満であり、加熱待機-サーチのシーケンスは継続した。80℃では、検出された自己発熱がその閾値を超えたため、熱量計は断熱モードに切り替わった。熱慣性[1]を考慮すると、断熱温度上昇は94.9 K (ΔTad)と計算される。この差は、主に容器の質量倍比熱容量と試料の質量倍比熱容量の比で与えられる、いわゆるファイ係数に基づいている。分解中の試料の自己発熱による温度上昇に加え、圧力上昇も定量化できる。断熱区間の終わりでは、圧力の上昇は20bar以上であった。
結論
35%過酸化水素水溶液の分解挙動を、MMCの走査モジュールおよびARC® 。走査モジュールは、一定レベルの電力(今回のように)または一定加熱速度のいずれかを使用して操作されるため、これらの実験にかかる時間は、Heat-Wait-Search (HWS)Heat-Wait-Search は、暴走反応熱量測定(ARC)に基づく熱量計装置で使用される測定モードである。heat-wait-search の方法よりもはるかに短い。したがって、走査モジュールは、未知の試料を自己分解や有害性の可能性に関して調査するための優れたスクリーニングツールである。調査した試料がスキャニング実行中に不安定な温度挙動を示す場合、または圧力上昇によって分解反応が示される場合は、ARC®-タイプの装置を使用して試料をさらに調査する必要があります。圧力上昇や断熱温度上昇のような値は、化学物質の危険性を評価する上で非常に重要であり、NETZSCH マルチモジュール熱量計(MMC)274Nexus を使用して簡単に求めることができます。