
Perossido di idrogeno
Il perossido di idrogeno puro (H2O2) è un liquido di colore blu pallido, miscelabile in qualsiasi rapporto con l'acqua. Le soluzioni acquose a bassa percentuale sono ampiamente utilizzate come agenti sbiancanti grazie alle loro forti proprietà ossidanti. Oltre che per lo sbiancamento di legno, carta o capelli, le soluzioni di perossido di idrogeno sono utilizzate anche come agenti ossidanti o in campo medico come disinfettanti. La tendenza del perossido di idrogeno a decomporsi in acqua e ossigeno (equazione 1) è il motivo della sua applicazione come propellente liquido nei motori a razzo.
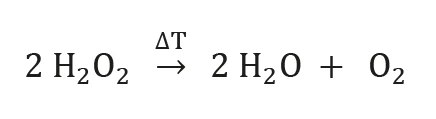
Il calorimetro a moduli multipli (MMC) a confronto con la calorimetria a scansione differenziale (DSC)
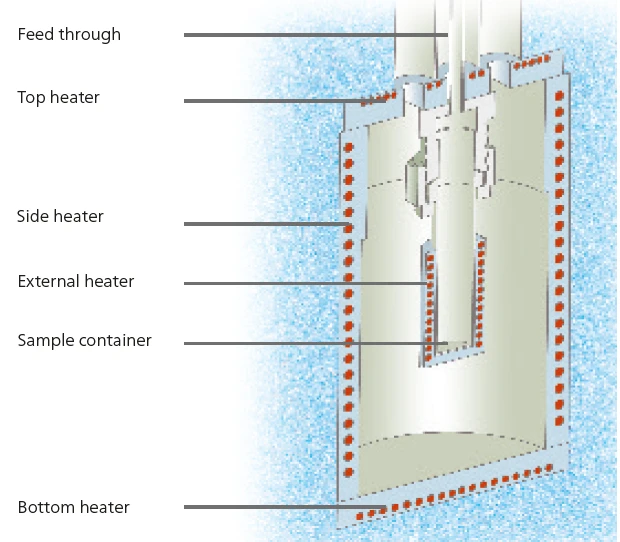
Il calorimetro a moduli multipli NETZSCH Calorimetro a moduli multipli (MMC)Un dispositivo calorimetrico a modalità multipla costituito da un'unità di base e da moduli intercambiabili. Un modulo è predisposto per la calorimetria a velocità accelerata (ARC), il modulo ARC. Un secondo modulo è utilizzato per i test di scansione (Scanning Module) e un terzo e quarto modulo sono relativi a batterie e polimeri, test farmaceutici per celle a moneta (Coin Cell Module).MMC 274 Nexus® (figura 1) offre tre diversi moduli di misurazione [1]. Il modulo ARC® può essere utilizzato per studi sui rischi termici; il modulo Coin-Cell è specializzato per lo studio delle batterie; il modulo Scanning può essere utilizzato per valutare i dati calorici di un singolo riscaldamento. A differenza della tecnica ampiamente utilizzata e conosciuta della calorimetria a scansione differenziale (DSC), il Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione dell'Calorimetro a moduli multipli (MMC)Un dispositivo calorimetrico a modalità multipla costituito da un'unità di base e da moduli intercambiabili. Un modulo è predisposto per la calorimetria a velocità accelerata (ARC), il modulo ARC. Un secondo modulo è utilizzato per i test di scansione (Scanning Module) e un terzo e quarto modulo sono relativi a batterie e polimeri, test farmaceutici per celle a moneta (Coin Cell Module).MMC può gestire campioni fino a un volume di 2 ml. Per il riscaldamento dei campioni sono disponibili due opzioni: una velocità di riscaldamento costante o un livello di potenza costante. Utilizzando le informazioni sulla potenza fornita al campione e sulla velocità di riscaldamento, è possibile calcolare un segnale di flusso di calore. Utilizzando metalli come l'indio, lo stagno e il bismuto, è possibile determinare sia la temperatura che la sensibilità dello strumento. Con 1000-9000 mg (volume del campione di circa 1 ml), le masse tipiche del campione sono notevolmente più elevate per l'Calorimetro a moduli multipli (MMC)Un dispositivo calorimetrico a modalità multipla costituito da un'unità di base e da moduli intercambiabili. Un modulo è predisposto per la calorimetria a velocità accelerata (ARC), il modulo ARC. Un secondo modulo è utilizzato per i test di scansione (Scanning Module) e un terzo e quarto modulo sono relativi a batterie e polimeri, test farmaceutici per celle a moneta (Coin Cell Module).MMC rispetto alle masse del campione utilizzate per la DSC, che sono in genere comprese tra 5 e 10 mg. Tuttavia, l'incertezza valutata per il Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione della Calorimetro a moduli multipli (MMC)Un dispositivo calorimetrico a modalità multipla costituito da un'unità di base e da moduli intercambiabili. Un modulo è predisposto per la calorimetria a velocità accelerata (ARC), il modulo ARC. Un secondo modulo è utilizzato per i test di scansione (Scanning Module) e un terzo e quarto modulo sono relativi a batterie e polimeri, test farmaceutici per celle a moneta (Coin Cell Module).MMC è di circa l'1% per le determinazioni di temperatura e meno del 5% per le determinazioni di entalpia.
Modulo di scansione e modulo ARC®
Questo lavoro studia il comportamento di Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione termica di soluzioni acquose di perossido di idrogeno a diverse concentrazioni. Per questi studi vengono impiegati due moduli MMC: il Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione (vedi figura 2) per lo screening dei campioni e il modulo ARC® (vedi figura 3) per gli studi su Heat-Wait-Search (HWS)Heat-Wait-Search è una modalità di misurazione utilizzata nei dispositivi calorimetrici secondo la calorimetria a velocità accelerata (ARC).heat-wait-search (Heat-Wait-Search (HWS)Heat-Wait-Search è una modalità di misurazione utilizzata nei dispositivi calorimetrici secondo la calorimetria a velocità accelerata (ARC).HWS). Tramite un riscaldatore esterno che circonda direttamente il recipiente del campione (figura 4), il Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione può fornire al campione un livello di potenza costante.
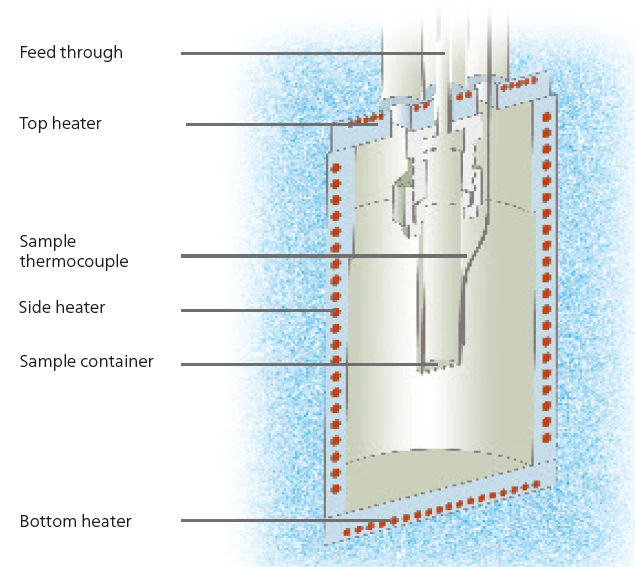

Condizioni di misura
Il perossido di idrogeno (Sigma Aldrich) è stato ricevuto in soluzione acquosa (35%) e conservato a temperatura ambiente. La soluzione di perossido di idrogeno è stata utilizzata come ricevuta ed è stata diluita con acqua purificata per osservare diverse concentrazioni inferiori. La composizione dei campioni diluiti è riassunta nelle tabelle 1 e 2. Le condizioni di misura per entrambi i moduli di scansione e ARC® sono confrontate nella tabella 3.
Tab 1: Composizioni dei campioni per lo screening (Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.Modulo di scansione)
| Numero di campioni | Concentrazione del campione/% | H2O2/g | H2O/g | Totale/g |
|---|---|---|---|---|
| 1 | 35 | 1.03106 | 0.0 | 1.03106 |
| 2 | 26 | 0.75757 | 0.25623 | 1.0138 |
| 3 | 17 | 0.5148 | 0.52494 | 1.03974 |
| 4 | 8.6 | 0.25169 | 0.7741 | 1.02579 |
| 5 | 4.3 | 0.12376 | 0.88605 | 1.00981 |
| 6 | 2.6 | 0.07316 | 0.92551 | 0.99867 |
| 7 | 1.1 | 0.03099 | 0.96707 | 0.99806 |
| 8 | 0.4 | 0.01215 | 1.00176 | 1.01391 |
Tab 2: Composizione dei campioni per le prove adiabatiche (moduloARC )
| Numero di campioni | Concentrazione del campione/% | H2O2/g | H2O/g | Totale/g |
|---|---|---|---|---|
| 9 | 35 | 1.02157 | 0.0 | 1.02157 |
| 10 | 17 | 0.74935 | 0.52494 | 1.00359 |
| 11 | 8.6 | 0.51466 | 0.50962 | 1.02428 |
| 12 | 4.3 | 0.25036 | 0.77525 | 1.02561 |
| 13 | 2.6 | 0.14776 | 0.877248 | 1.02034 |
Tab 3: Condizioni di misura
MMC 274 Nexus® | ||
|---|---|---|
| Modulo MMC | Scansione | ARC® |
| Materiale del recipiente | Acciaio inossidabile | Acciaio inossidabile |
| Tipo di recipiente | Chiuso | Chiuso |
| Massa del recipiente | 7.da 0 a 7,25 g | 7.da 0 a 7,25 g |
| Riscaldamento | Potenza costante (250 mW) | |
| Atmosfera | Aria | Aria |
| Tasso di gas di lavaggio | Statico | Statico |
| Intervallo di temperatura | RT ... 250°C | RT ... 250°C |
| Massa del campione | 998.da 67 a 1039,74 mg | 1003.da 6 a 1025,6 mg |
Risultati e discussione
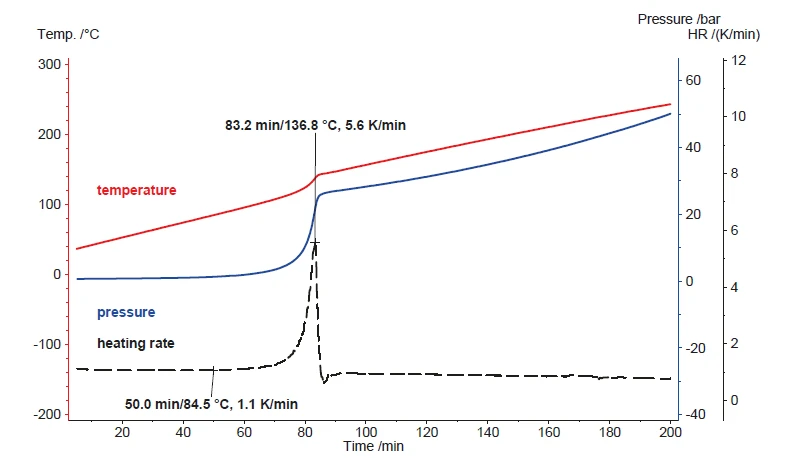
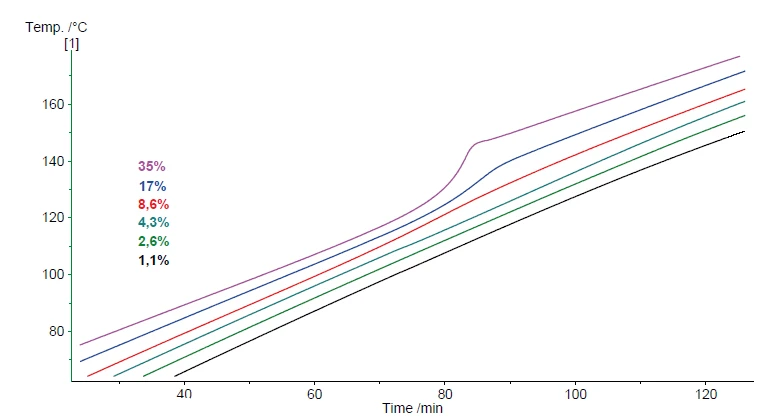
A seconda della variazione della capacità termica dei campioni, l'input di potenza costante di solito si traduce in una velocità di riscaldamento quasi costante del campione. La Figura 5 mostra il risultato del riscaldamento del perossido di idrogeno (35%) utilizzando il Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione a una potenza costante di 250 mW. La velocità di riscaldamento risultante è di circa 1 K/min per i primi 60 minuti. Dopo un'ora, inizia la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione che produce ulteriore calore. Durante la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione, la velocità di riscaldamento aumenta fino a un massimo di 5,6 K/min e anche la pressione rilevata aumenta. Secondo l'equazione 1, la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione genera ossigeno. Oltre all'evaporazione dell'acqua, questa formazione di gas è la causa principale dell'aumento di pressione durante il riscaldamento.
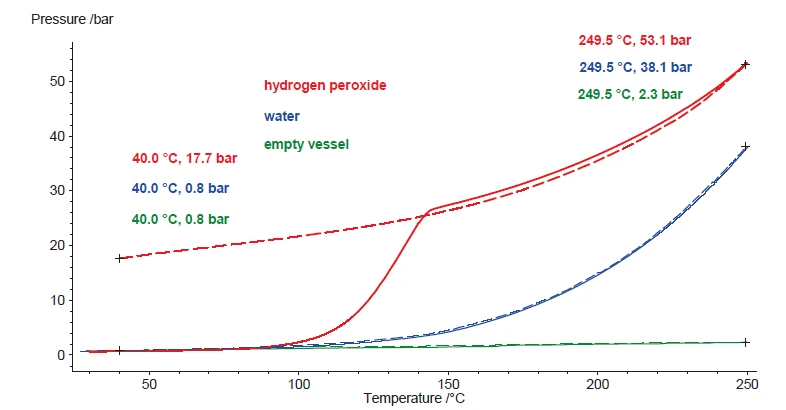
Confronto del comportamento di H2O2,H2Oe Vaso vuoto
I risultati della figura 5 presentano esclusivamente il riscaldamento del campione. Poiché la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione del perossido di idrogeno non è reversibile, l'ossigeno generato non viene ripreso per formare il perossido di idrogeno iniziale durante il raffreddamento. Invece, i prodotti formati di acqua e ossigeno si raffreddano a temperatura ambiente rispettivamente come liquido e come gas. Il segnale di pressione indica 17,7 bar a 40°C, che riflette la quantità di ossigeno che si forma durante la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione (figura 6). Prendendo invece la stessa quantità di acqua, la pressione aumenta anche durante il riscaldamento, ma poiché l'acqua rimane chimicamente invariata, tutto il vapore acqueo precipita nuovamente durante il raffreddamento. Ecco perché la linea blu tratteggiata, che indica il segnale di pressione per l'acqua durante il raffreddamento, mostra valori quasi identici a quelli del riscaldamento (linee solide). A titolo di confronto, le linee verdi mostrano l'andamento del segnale di pressione durante il riscaldamento e il raffreddamento per un recipiente vuoto.
H2O2 a varie concentrazioni
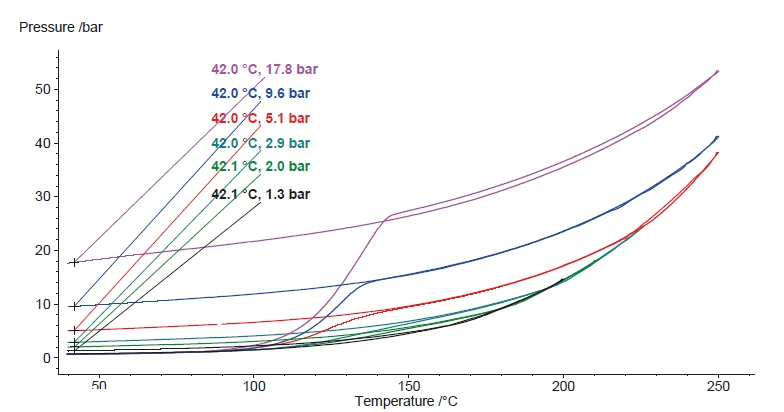
Soprattutto nel confronto con l'acqua, si può notare che l'evaporazione - che si verifica in una certa misura anche all'interno di un sistema a vaso chiuso - è sempre reversibile. Ciò è confermato dal segnale di pressione a 40°C dopo il raffreddamento. D'altra parte, la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione del perossido di idrogeno produce una quantità specifica di gas. Pertanto, il segnale di pressione dovrebbe essere proporzionale alla quantità assoluta di perossido di idrogeno all'interno della soluzione. Ripetendo questi test con campioni di varie concentrazioni di perossido di idrogeno, l'aumento di pressione durante il test dovrebbe essere proporzionale alla concentrazione di perossido di idrogeno. La Figura 7 mette a confronto i risultati del riscaldamento dei campioni da 1 a 6. Le concentrazioni di perossido di idrogeno associate sono riassunte nella Tabella 1.
Correlazione tra concentrazione di H2O2 e pressione
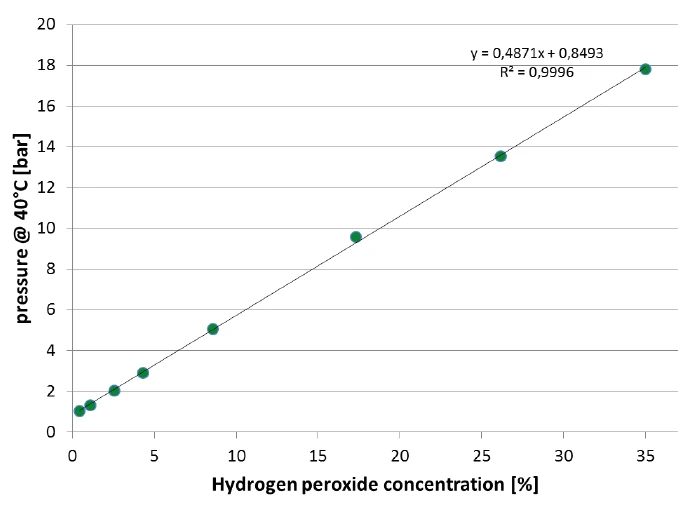
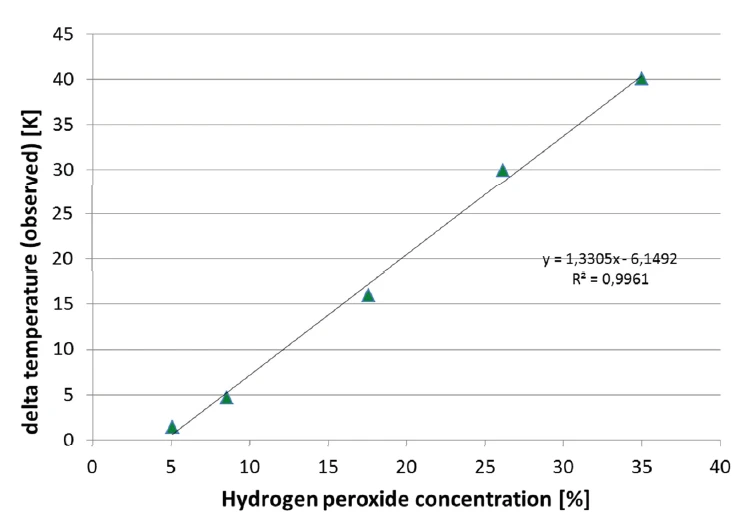
La reazione di Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione del perossido di idrogeno è indicata dall'aumento della velocità di riscaldamento misurata sul campione e dall'aumento della pressione. Nella figura 8, viene valutato il segnale della pressione rimanente dopo la reazione e dopo il raffreddamento a 42°C. Si osserva una correlazione quasi perfettamente lineare della pressione con la concentrazione di perossido di idrogeno del campione. Questa correlazione è rappresentata nella figura 9.
Varie concentrazioni di H2O2 studiate con il moduloARC®
Varie concentrazioni di H2O2 studiate con il modulo ARC® Una serie simile di concentrazioni di perossido di idrogeno acquoso è stata studiata anche con il modulo ARC® della MMC (figura 3). Le concentrazioni di perossido di idrogeno associate sono riassunte nella Tabella 2. Il modulo ARC® può essere utilizzato per determinare in modo specifico la temperatura di inizio Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. decomposizione mediante il cosiddetto programma Heat-Wait-Search (HWS)Heat-Wait-Search è una modalità di misurazione utilizzata nei dispositivi calorimetrici secondo la calorimetria a velocità accelerata (ARC).heat-wait-search (Heat-Wait-Search (HWS)Heat-Wait-Search è una modalità di misurazione utilizzata nei dispositivi calorimetrici secondo la calorimetria a velocità accelerata (ARC).HWS). Con l'aiuto della sequenza di riscaldamento, equilibratazione e rilevamento, si determina la velocità di autoriscaldamento del campione in condizioni quasi isoterme e quindi si analizza il campione in modalità adiabatica [1, 2].
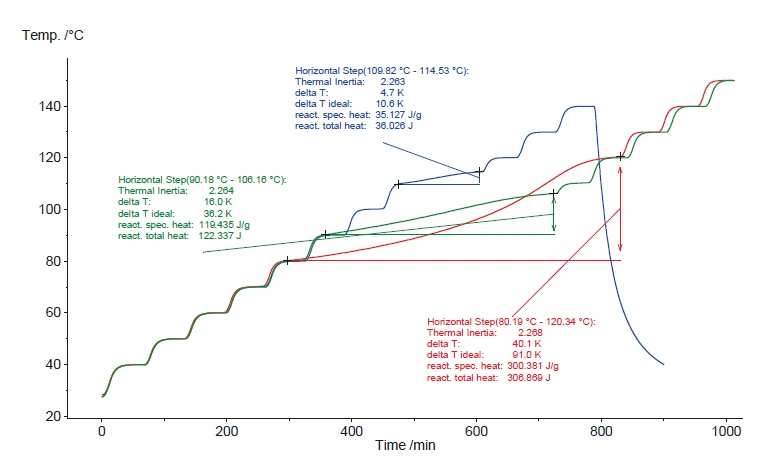
I risultati per concentrazioni di perossido di idrogeno pari al 35%, 17% e 8,6% sono presentati nella figura 10. Come previsto, i risultati confermano la presenza di un sito . Come previsto, i risultati confermano un smaller aumento della temperatura (ΔTobs) in condizioni adiabatiche per concentrazioni di perossido di idrogeno inferiori. La temperatura alla quale viene rilevata la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione (onset) aumenta per concentrazioni inferiori a causa del minor rilascio di energia (90°C e 110°C). La velocità massima di autoriscaldamento per concentrazioni di perossido di idrogeno inferiori al 5% è inferiore a 0,02 K/min. Per questo motivo non si rilevano eventi esotermici in questo caso. La figura 11 mostra le fasi di aumento della temperatura (ΔTobs) rilevate durante i segmenti adiabatici di diversi test Heat-Wait-Search (HWS)Heat-Wait-Search è una modalità di misurazione utilizzata nei dispositivi calorimetrici secondo la calorimetria a velocità accelerata (ARC).HWS.
Conclusione
Questi risultati dimostrano la capacità di screening del Modulo di scansioneUn modulo calorimetrico facente parte del Calorimetro Multiplo (MMC) che consente di eseguire un test di scansione di un campione. Questa procedura può servire come test di screening per rilevare un potenziale pericolo termico in un tempo di misurazione ragionevolmente breve.modulo di scansione MMC. Nel caso di reazioni fortemente esotermiche, la velocità di autoriscaldamento aumenterà in modo significativo, fino a superare il livello di circa 1 K/min, come risultato dell'alimentazione costante. Pertanto, quando un campione sconosciuto presenta una Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione esotermica, è possibile riconoscerlo entro alcune ore. Non appena viene riconosciuto il potenziale di pericolo, si consiglia di eseguire un test AdiabaticoL'adiabatico descrive un sistema o una modalità di misurazione senza alcuno scambio di calore con l'ambiente circostante. Questa modalità può essere realizzata utilizzando un dispositivo calorimetrico secondo il metodo della calorimetria a velocità accelerata (ARC). Lo scopo principale di tale dispositivo è quello di studiare scenari e reazioni di fuga termica. Una breve descrizione della modalità adiabatica è "nessun calore in entrata - nessun calore in uscita".adiabatico utilizzando un modulo MMC ARC® [1]. Questo test HWS può facilmente richiedere un'intera giornata, ma d'altra parte è molto più pertinente all'equilibrio termico rispetto a un test di scansione [2].
Inoltre, i risultati presentati sopra dimostrano l'utilità del segnale di pressione. La potenza costante di 250 mW consente una velocità di riscaldamento di circa 1 K/min per un campione acquoso di 1 g. I campioni con una concentrazione di perossido di idrogeno inferiore al 5% non superano questa velocità di riscaldamento grazie all'energia rilasciata durante la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione. Ciò significa che, grazie al Tasso di autoriscaldamentoPer rilevare il tasso di autoriscaldamento di una sostanza si utilizza un tipo speciale di calorimetro. Il relativo metodo è chiamato calorimetria a velocità accelerata (ARC). tasso di autoriscaldamento del campione, la Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione per le basse concentrazioni viene mascherata dalla potenza assorbita. Il segnale di pressione, invece, non è influenzato dalla potenza assorbita. Pertanto, può essere considerato un indicatore significativo del verificarsi o meno di una Reazione di decomposizioneUna reazione di decomposizione è una reazione termicamente indotta di un composto chimico che forma prodotti solidi e/o gassosi. reazione di decomposizione, soprattutto nel caso di concentrazioni inferiori.