
16.05.2022 by Aileen Sammler
DSC Helps When it Comes to Keeping Things Tight
Learn, how Differential Scanning Calorimetry can help in the case of leakage: A user report by Kirsten Hacker, chemical laboratory technician in the engineering team at KLINGER Kempchen GmbH.

Since 2008, Kirsten Hacker has been working at KLINGER Kempchen GmbH (formerly know as „Kempchen Dichtungstechnik GmbH“) as chemical laboratory technician. She now has 36 years of professional experience in the laboratory. Her responsibilities range from routine analyses and laboratory tests to material characterizations and the determination of material compatibility and resistance. In the following report she explains, how thermal analysis can help in the case of leakage.
DSC Helps When it Comes to Keeping Things Tight
Leaks and the associated undesired escape of media often result in temporary production stops which can generate high economic costs. Possible causes can include material failure or mix-up. Such incidents must be recognized in time, or even better, avoided beforehand. KLINGER Kempchen specializes in static gaskets, wrappings and expansion joints which must function particularly reliably in the process industry where criticial media are also conveyed under high pressures.

How Can Thermal Analysis Help in the Case of Leakage?
Differential scanning calorimetry (DSC), a thermal analysis method, is used to test materials for existing Phase TransitionsThe term phase transition (or phase change) is most commonly used to describe transitions between the solid, liquid and gaseous states.phase transitions or chemical reactions due to the influence of temperature (heat/cold). This way, materials with undesirable properties can be eliminated in advance. In the DSC method, the sample is subjected to a defined temperature program, i.e., the temperature at the sample is either increased or decreased. The adsorbed (ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal) or absorbed (endothermal) heat is measured by means of a heat flux. This allows conclusions to be drawn on chemical or physical processes, such as Phase TransitionsThe term phase transition (or phase change) is most commonly used to describe transitions between the solid, liquid and gaseous states.phase transitions, CrystallizationCrystallization is the physical process of hardening during the formation and growth of crystals. During this process, heat of crystallization is released.crystallization or Decomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition reactions. By recording endo- and ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermal effects, the profile of a material is established; this profile is as individual as a fingerprint. Based on this, pure and mixed materials can be qualitatively compared and material properties can be predicted.
Why Is the Seal Not Tight?
Of course, there are also recurring failures in industrial production in the field of seal joints; the causes of these must be clarified as quickly as possible. One example is the following case:
A customer of KLINGER Kempchen received a gasket from a plant engineering company which was supposed to be made of chloroprene rubber (CR), as defined for this application. A gasket made of CR was a requirement for the customer’s application due to its chemical resistance to the medium used. The gasket, however, failed during its use.
The customer contacted the Engineering Department of KLINGER Kempchen GmbH for assistance. The “alleged” CR gasket (here referred to as “unknown gasket”) was compared to a KLINGER Kempchen CR reference material by DSC analysis. The measurements were carried out in a NETZSCH DSC 200 Maja, with a sample weighing approx. 10 mg; they consisted of two heating runs at 20 K/min in the temperature range between -100°C and 100°C.
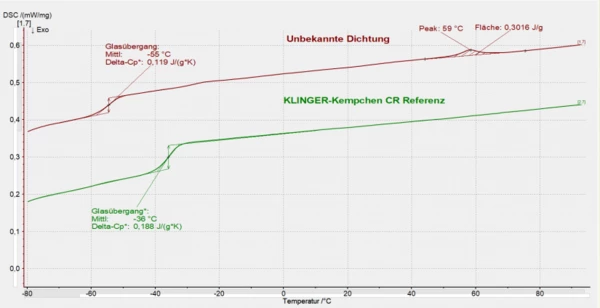
The comparison of the DSC measurements shows a significant difference in the Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition temperatures (unknown gasket -55°C; KLINGER Kempchen CR reference -36°C). The unknown gasket exhibits an additional endothermal effect at approx. 60°C.
From the DSC results, it was possible to conclude that the specifications for the gasket did not meet the quality standards and that this was the reason for the material failure. A subsequent FT-IR analysis confirmed the result.
Use of New Materials
The DSC method plays another important role in the optimization and further development of materials. Once a substitute material has been deemed to be possibly suitable, it must be tested to confirm this. In the following case, it was necessary to find a substitute material for one already in use, due to reasons of safety and health protection. The plan was to replace material A (aluminum silicate wool) with material B (biosoluble rock wool).
According to the data sheet, both materials contain rock wool, cellulose, silicate fillers and organic binders. Material A additionally contains quartz. In the case of material B, the rock wool is described as “biosoluble”.
This time, a NETZSCH STA 449 F3 was used, an instrument for combined heat-flow/mass-loss measurement. Samples of approx. 25 mg of both materials were heated from room temperature to 1200°C under air at a heating rate of 5 K/min. The combined TGA/DSC signal was evaluated.
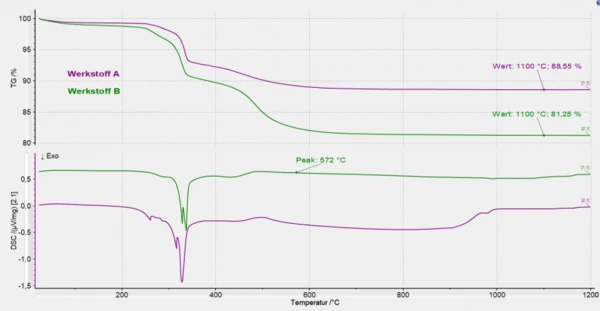
Both material types show a similar overall course over the entire temperature range (multi-step mass-loss curves) although there are substantial differences in the heights of the individual Decomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition steps.
The DSC curve shows the ExothermicA sample transition or a reaction is exothermic if heat is generated.exothermalDecomposition reactionA decomposition reaction is a thermally induced reaction of a chemical compound forming solid and/or gaseous products. decomposition of the organic binder and cellulose up to 400°C. This is followed by the endothermal release of water from the siliceous filler materials.
The presence of quartz-containing components can be confirmed by a DSC effect at 572°C for material B.
Differences in the determined residual mass at 1100°C (material A: 88.5%, material B: 81.3%) can be seen in the TGA curve. Further investigations in the area of mechanical stability would have to be carried out to ensure that the biosoluble rock wool still meets the corresponding requirements, e.g., for mechanical-technical properties and sealing behavior.
Subsequent measurements, e.g. of the leakage rates, showed that the material does still meet the corresponding requirements.
This exciting article has aroused our curiosity even more. That is why we have asked Ms. Hacker further questions:
NETZSCH: Ms. Hacker, you work in the KLINGER Kempchen laboratory. Your area of responsibility is very diverse. Customers approach the KLINGER Engineering Department and ask for support with questions such as material requirements and resistance regarding the topic of static seals or when a failure case has occurred. What are the most commonly asked questions customers approach you with and how can (thermal) analysis methods help solve such problems?
Kirsten Hacker: We create fingerprints of all our polymer materials with the help of thermal analysis methods like DSC and STA. These fingerprints of our polymer materials help monitor the quality of the production during routine comparative tests (batch testing). Furthermore, we support our customers in characterizing unknown sealing materials. It may happen, for example, that the customer used a gasket for which the specification is no longer known. Thermal analysis is very helpful in this determination/typification since, for example, certain materials can be identified by determining their thermal properties with the help of DSC. Here, among other things, the Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition temperature or Melting Temperatures and EnthalpiesThe enthalpy of fusion of a substance, also known as latent heat, is a measure of the energy input, typically heat, which is necessary to convert a substance from solid to liquid state. The melting point of a substance is the temperature at which it changes state from solid (crystalline) to liquid (isotropic melt).melting temperature provides information about the type of material involved. Additionally, STA can be employed to determine the composition of the compound or elastomer. In the event of product failure, thermal analysis can be used to rule out material mix-ups. It can also often be applied to show a change to the material occurring through the use and possible overload of the polymer.
NETZSCH: Differential Scanning Calorimetry is one of the most frequently used thermoanalytical methods. Where do you see the strength of DSC in your applications?
Kirsten Hacker: In routine analysis, i.e., in the field of quality assurance, DSC is a very fast and reliable tool. Batch tests in the laboratory immediately and easily reveal any deviation in the production of elastomer mixtures and thermoplastics as well as their compounds. In addition, STA allows for determination of the compositions, i.e., the respective proportions, so that compliance with the formulations can be tracked.
By determining melting temperatures, Glass Transition TemperatureThe glass transition is one of the most important properties of amorphous and semi-crystalline materials, e.g., inorganic glasses, amorphous metals, polymers, pharmaceuticals and food ingredients, etc., and describes the temperature region where the mechanical properties of the materials change from hard and brittle to more soft, deformable or rubbery.glass transition temperatures and thermal decompositions of the sealing materials, application fields / limits of the materials can be defined in a supportive way. However, also new materials or even a different supplier of raw materials can be put to the test beforehand with the help of DSC.
NETZSCH: Ms. Hacker, thank you very much for your comments and the interesting insight into sealing technology. We look forward to a further good cooperation.