Введение
Амоксициллин (рис. 1) - это антибиотик из группы аминопенициллинов. Он используется для лечения бактериальных инфекций, таких как инфекции среднего уха, пневмония и кожные инфекции [2]. В данном случае он был измерен с помощью ДСК и ТГ-ФТ-ИК, чтобы получить информацию о некоторых его термических свойствах, таких как температура плавления и температура разложения, а также о продуктах деградации.
Результаты испытаний
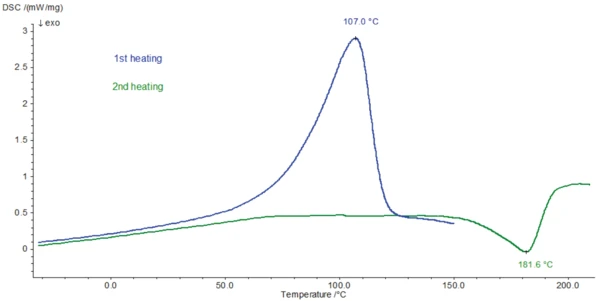
Измерение ДСК проводилось на образце тригидрата амоксициллина (1,622 мг) с помощью прибора DSC 204 F1 Phoenix® . Использовали алюминиевый тигель с пробитой вручную крышкой (3 отверстия). Образец дважды нагревали от -80°C при скорости нагрева 10 К/мин; первый раз до 150°C, второй раз до 210°C. Между двумя нагреваниями образец охлаждали с контролируемой скоростью 10 К/мин. Измерения ДСК при нагревании представлены на рисунке 2.
Для измерения ТГА образец (4,79 мг) был подготовлен в тигле из оксида алюминия и нагрет до 700°C в динамической атмосфере азота со скоростью 10 К/мин. Кривая ТГА представлена на рисунке 3.
Эндотермический пик, обнаруженный при 107°C (кривая ДСК, 1-й нагрев, рис. 2), связан с потерей массы на 12,9%. Этот процесс, который может быть связан с выделением летучих веществ, регистрируется при более высокой температуре при измерении ДСК, чем соответствующая потеря массы на кривой ТГА. Температура, при которой происходит испарение, зависит от наличия крышки на тигле и от размера отверстий в крышке тигля. Поскольку при измерении методом ТГА крышка не использовалась, улетучивание происходит при более низкой температуре, чем при измерении методом ДСК, где используется крышка с отверстиями.
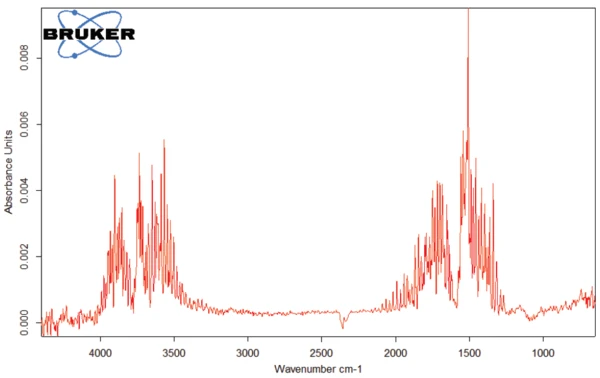
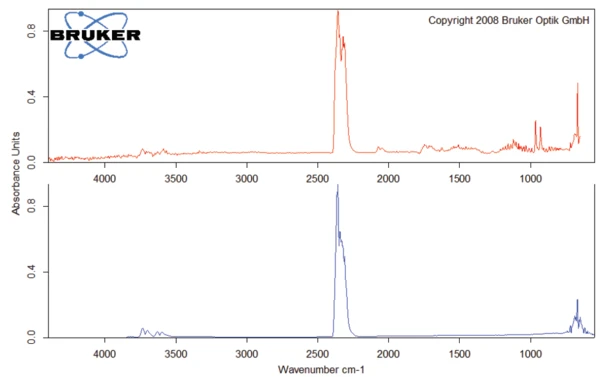
ИК-Фурье спектр продуктов, выделяющихся при 93°C (рис. 4), характерен для воды.
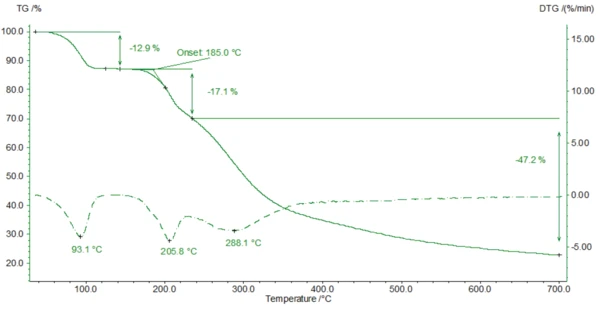
Молярная масса амоксициллина составляет 365,4 г/моль [2]. В результате молярная масса тригидрата амоксициллина составляет 419,4 г/моль. Следовательно, высвобождение всей воды из тригидрата амоксициллина теоретически привело бы к изменению массы примерно на 12,9 %. Здесь (рис. 3) результаты измерений находятся в отличном соответствии с теоретическими значениями.
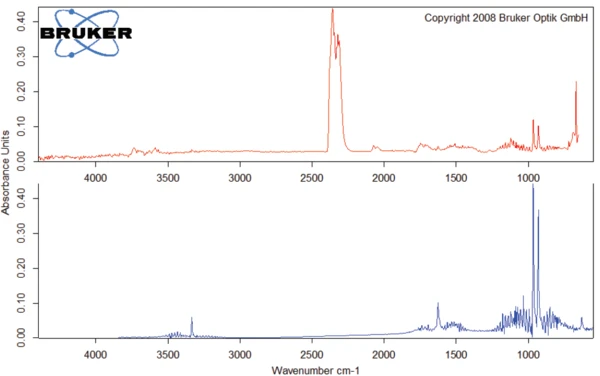
Вторая ступень потери массы происходит при 185°C (начальное значение кривой ТГА). Соответствующие ИК-Фурье спектры показывают, что деградация амоксициллина начинается с выделения диоксида углерода (рис. 5) и аммиака (рис. 6). Это связано с экзотермическим эффектом на кривой ДСК. Деградация продолжается, и при нагревании до 700 °С амоксициллин теряет более 77 % своей первоначальной массы.
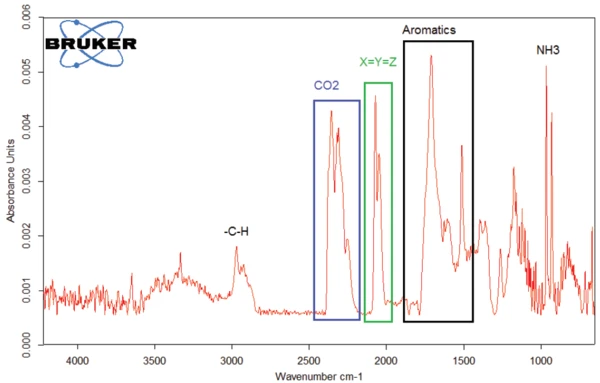
ИК-Фурье спектр для веществ, выделившихся при 294°C, приведен на рисунке 7. Характерные полосы дляCO2 и NH3, которые уже обсуждались, также были обнаружены. Однако видны дополнительные полосы поглощения: Полосы между 3000 и 2800 см-1 указывают на наличие связей -C-H, в то время как полосы между 1500 и 1800 см-1 могут быть результатом ароматики. Область между 1900 и 2300 см-1 характерна для тройных или двойных связей X_Y_Z. [3, 4] .
Резюме
Эффекты, обнаруженные с помощью измерений ДСК при термической обработке амоксициллина, не могут быть объяснены только интерпретацией результатов ДСК. Только путем дополнительных измерений с использованием комбинации методов ТГА-ФТ-ИК можно подтвердить, что эффект ДСК при 107°C обусловлен испарением воды, а не плавлением, вызывающим эндотермический пик ДСК, но без потери массы. Потеря массы, продолжающаяся после испарения воды, является результатом разложения. Продукты разложения, в том числе диоксид углерода и аммиак, могут быть четко идентифицированы с помощью ИК-Фурье.