はじめに
シュウ酸塩は、シュウ酸C2H2O4(COOH)2(エタンジカルボン酸)の塩である。シュウ酸のカルシウム塩であるシュウ酸カルシウムは、無水結晶化し、シュウ酸カルシウム一水和物CaC2O4*H2Oとして、式あたり1分子の水を含む溶媒和物として結晶化する。
発生と応用
シュウ酸カルシウム一水和物は有機酸の塩であるが、自然界では一次鉱物として見出される。図1は、ドイツのエルツゲビルゲ山脈にあるシュレマ産のウィーウェライト結晶化である。ホエウェライトに加えて、ウェッデライトも第二の鉱物種として知られている[1]。

シュウ酸カルシウムは腎臓結石の主成分でもある。
熱分析では、シュウ酸カルシウム一水和物は熱天秤の機能性を確認するために使用される。この物質は保存安定性に優れ、経時変化もなく、実験室の雰囲気から湿気を吸着する傾向もありません。このような特徴から、熱天秤の温度ベース機能のチェックに使用するリフェレン物質として理想的です。
測定条件
- 装置
- TG 209 F1 Libra®
- 試料
- CaC2O4*H2O
- 試料重量
- 8.43 mg(図2の黒い曲線)と
- 8.67 mg(図2の赤い曲線)
- 容器
- Al2O3
- 雰囲気
- 窒素
- ガス流量
- 40ml/分
- 加熱速度
- 10K/分(図2の黒曲線)および
- 200K/min(図2の赤曲線)
熱重量測定
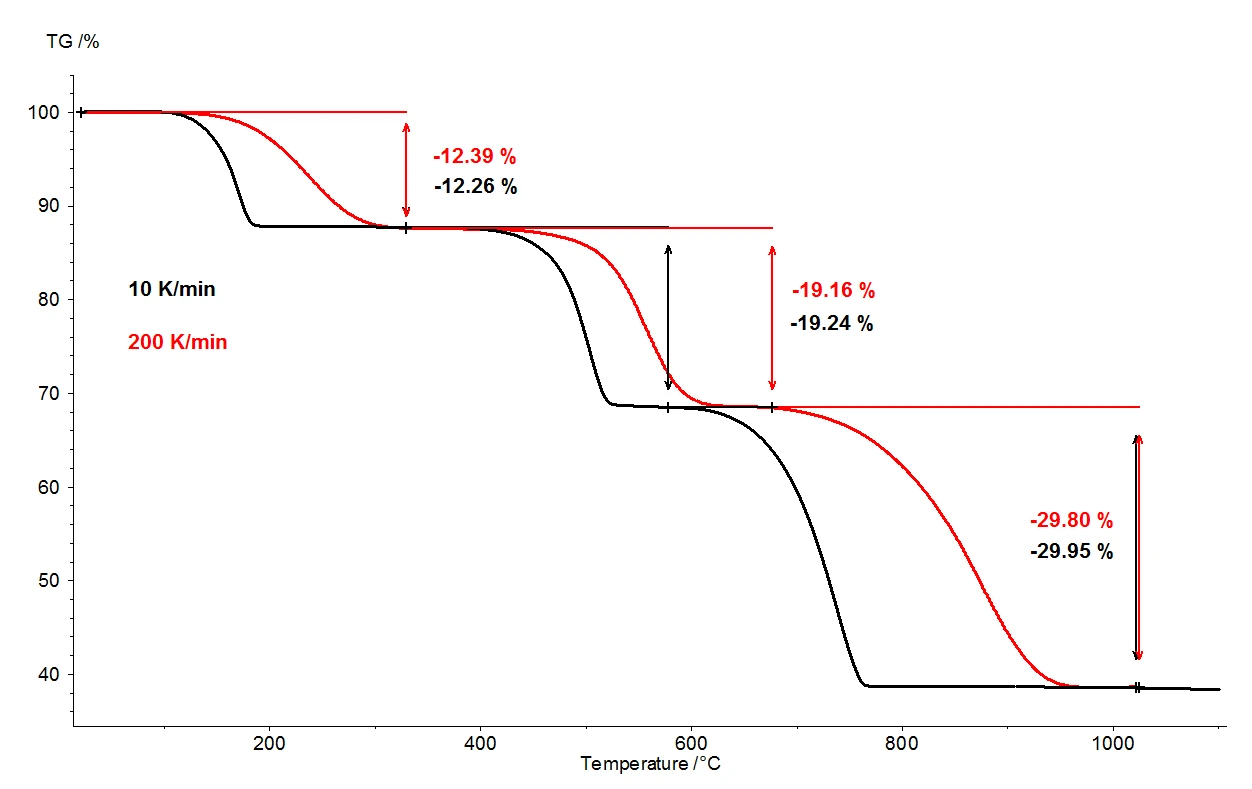
シュウ酸カルシウム一水和物を1100℃に加熱すると、熱天秤によって3つの明確に分離した質量減少ステップが検出される。図2は、シュウ酸カルシウム一水和物試料を用いた2回の熱重量測定結果の比較である。試料の相対質量変化が温度にわたって記録されている。図3は、熱重量測定結果の一次微分(DTG)について、2つの測定結果を温度の関数として比較したものである。
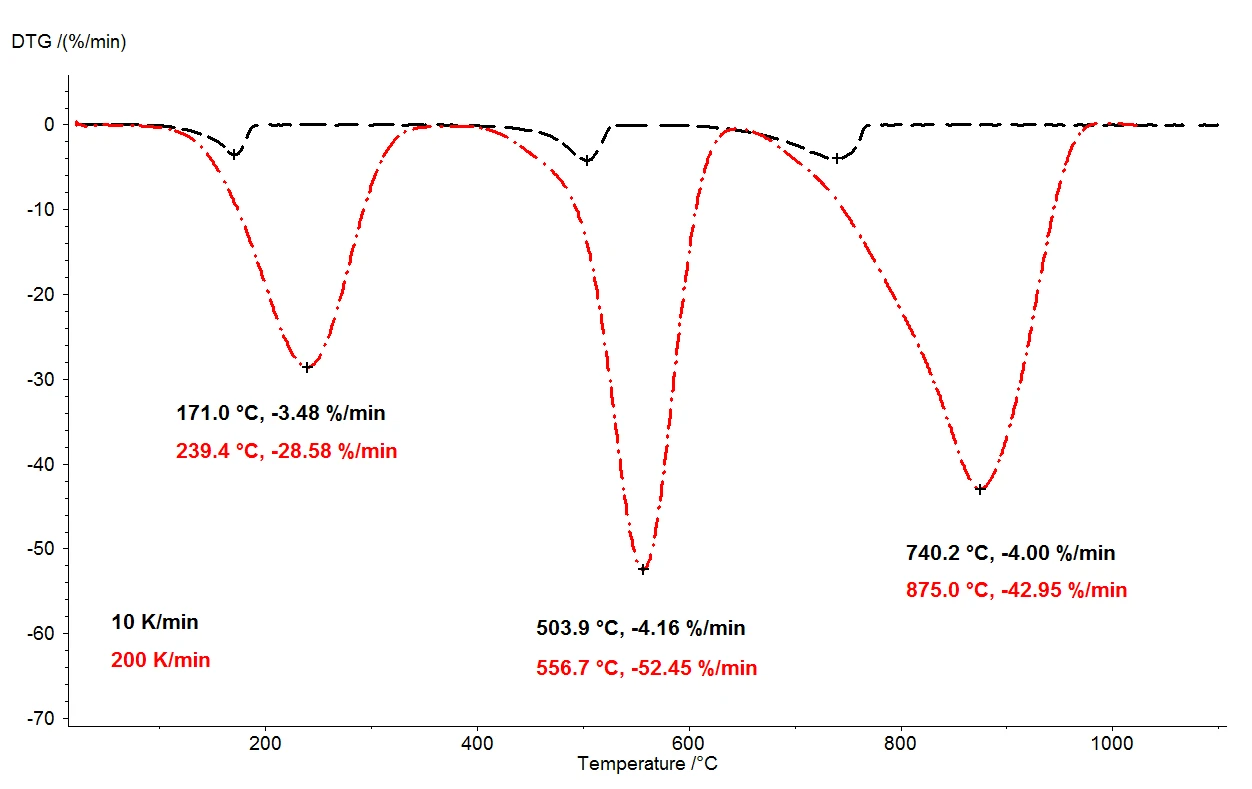
同じ条件下で、10 K/分(黒の曲線)と200 K/分(赤の曲線)の2種類の加熱速度を選択した。加熱速度を上げると、質量放出ステップの温度はより高い値にシフトし、放出速度(ガス放出の速度)は約10倍になる(DTG極小値、図3)。加熱速度を変化させたときに起こる温度のシフトは、よく理解されている現象であり、速度論データのさらなる評価に応用することができる[2]。温度シフトの他に、質量損失ステップの定量化が加熱速度に依存しないことも重要である。したがって、加熱速度200 K/分では、シュウ酸カルシウム一水和物の熱分解に関して、通常の加熱速度10 K/分と同じ情報が得られる。10K/分での測定では、室温から1100℃までの温度範囲をカバーするのに2時間近くかかるが、200K/分では5分で同じ測定が完了する。
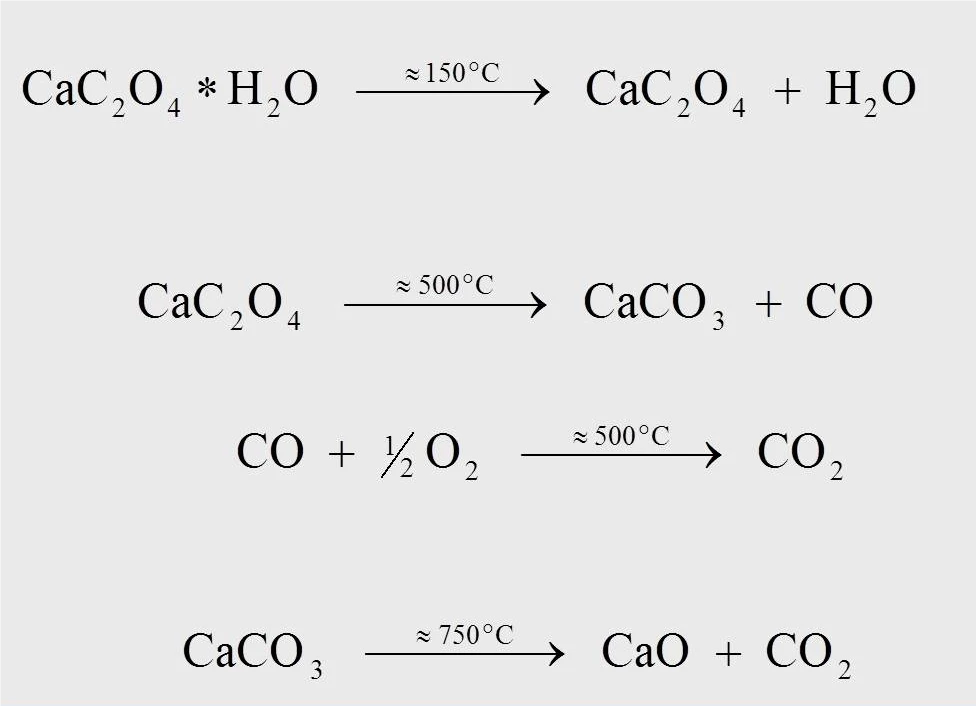
シュウ酸カルシウム一水和物の熱分解反応の反応式を図4に示す。10K/minで測定した場合、約170℃で一水和物(1)から水が分離し、無水シュウ酸カルシウムが生成する。約500℃でシュウ酸カルシウムは炭酸カルシウム(CaCO3)に変化し、一酸化炭素(CO)が分離する(2)。放出された一酸化炭素が二酸化炭素(CO2)に酸化されるその後の反応(3)は、酸素を含むパージガス流(合成空気や酸素など)の中でしか起こらない。750℃の温度で、炭酸カルシウムはCO2を放出しながら酸化カルシウムに分解する(4)。